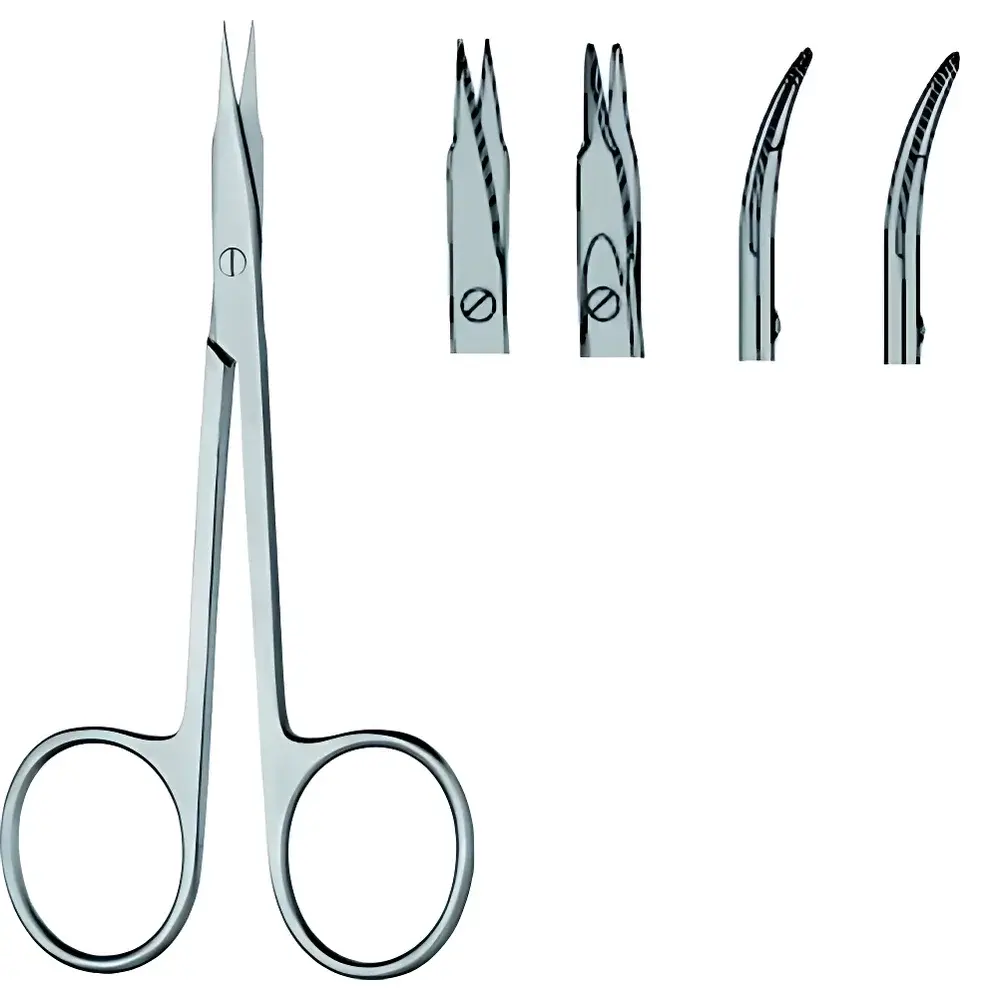
RWD STEVENS Precision Dissecting Scissors
| Brand | RWD |
|---|---|
| Origin | Shenzhen, China |
| Manufacturer Type | Direct Manufacturer |
| Country of Origin | China |
| Model | S13001-11 |
| Pricing | Available Upon Request |
Overview
RWD STEVENS Precision Dissecting Scissors (Model S13001-11) are high-precision surgical instruments engineered for delicate tissue manipulation in preclinical research, histology, and microsurgical applications. Designed in accordance with the legacy standards of STEVENS ergonomic instrumentation, these scissors feature a straight-blade/tapered-tip configuration optimized for controlled, low-force cutting of fine neural, vascular, and connective tissues. The 11.5 cm overall length balances maneuverability and control—critical for confined anatomical fields in rodent, avian, and small-mammal dissection protocols. Constructed from premium-grade stainless steel (AISI 420 or equivalent), the instrument undergoes precision grinding, cryogenic hardening, and electropolishing to ensure edge retention, corrosion resistance, and biocompatibility under repeated sterilization cycles (autoclave up to 134 °C, 2 bar).
Key Features
- Ergonomic finger-ring handles with textured matte finish for slip-resistant grip during prolonged use
- Micron-level blade alignment achieved via laser-guided assembly, ensuring consistent scissor action and minimal tissue drag
- Sharpness-certified cutting edges tested per ISO 7740 (Surgical Instruments – Requirements for Sharpness and Edge Retention)
- Non-reflective satin finish minimizes glare under stereomicroscope illumination and surgical lighting
- Compatible with standard ultrasonic cleaning systems and enzymatic decontamination solutions per AAMI ST79 guidelines
- Individually serialized for traceability; compliant with ISO 13485:2016 quality management system requirements for medical device manufacturers
Sample Compatibility & Compliance
These dissecting scissors are routinely employed in protocols involving fresh, fixed, or frozen biological specimens—including brain slices, retinal explants, embryonic tissue, and peripheral nerve segments. Their design supports compliance with Good Laboratory Practice (GLP) documentation requirements when used in regulated nonclinical safety studies (per OECD GLP Principles). While not classified as a Class II medical device for human implantation, the instrument meets ASTM F899-23 (Standard Specification for Stainless Steels for Surgical Instruments) and is suitable for use in IACUC-approved animal procedures under AAALAC-accredited facilities. Sterilization validation data (BI and process challenge devices) are available upon request for integration into facility SOPs.
Software & Data Management
As a manual, non-electronic instrument, the RWD STEVENS S13001-11 does not incorporate embedded sensors, firmware, or software interfaces. However, it is fully compatible with digital laboratory asset tracking systems (e.g., LabVantage, Quartzy, or custom LIMS) via its unique serial number. Institutions requiring audit-ready maintenance logs may utilize the included Certificate of Conformance (CoC), which documents material certification, dimensional inspection reports, and pass/fail results of functional testing. For laboratories operating under FDA 21 CFR Part 11 or EU Annex 11 environments, the physical logbook entry for instrument calibration (if sharpening is performed in-house) and sterilization cycle records remain the primary compliance artifacts.
Applications
- Microdissection of hippocampal subregions and cortical layers in rodent brain mapping studies
- Cranial window preparation and dura mater incision in in vivo optical imaging experiments
- Isolation of dorsal root ganglia and sciatic nerve branches for electrophysiology assays
- Embryonic heart tube dissection in zebrafish and chick developmental models
- Trimming of paraffin-embedded tissue blocks prior to microtome sectioning
- Preparation of explant cultures for organotypic slice viability assays
FAQ
Are these scissors autoclavable?
Yes—fully compatible with saturated steam sterilization at 134 °C, 2 bar for 18 minutes. Repeated cycling does not compromise blade hardness or handle integrity.
What sharpening service does RWD offer?
RWD provides certified re-sharpening through its ISO 13485-certified service center. Turnaround time is typically 5–7 business days; sharpening includes edge-angle verification and post-process passivation.
Is a CE mark or FDA listing available?
The instrument is CE-marked under the EU MDR 2017/745 as a Class I non-sterile, non-measuring surgical instrument. It is not FDA-listed, as it falls outside the scope of 21 CFR 878.4495 for general-purpose dissecting tools.
Can I order custom engraving (e.g., lab initials)?
Yes—laser engraving on the handle is available for orders ≥10 units. Engraving location and font comply with ISO 15223-1 labeling standards for traceability.
What is the expected service life under daily academic use?
With proper cleaning, sterilization, and storage, typical service life exceeds 5 years in high-throughput core facilities. Blade replacement is not supported; full-unit refurbishment is recommended after >2,000 autoclave cycles or visible edge degradation.