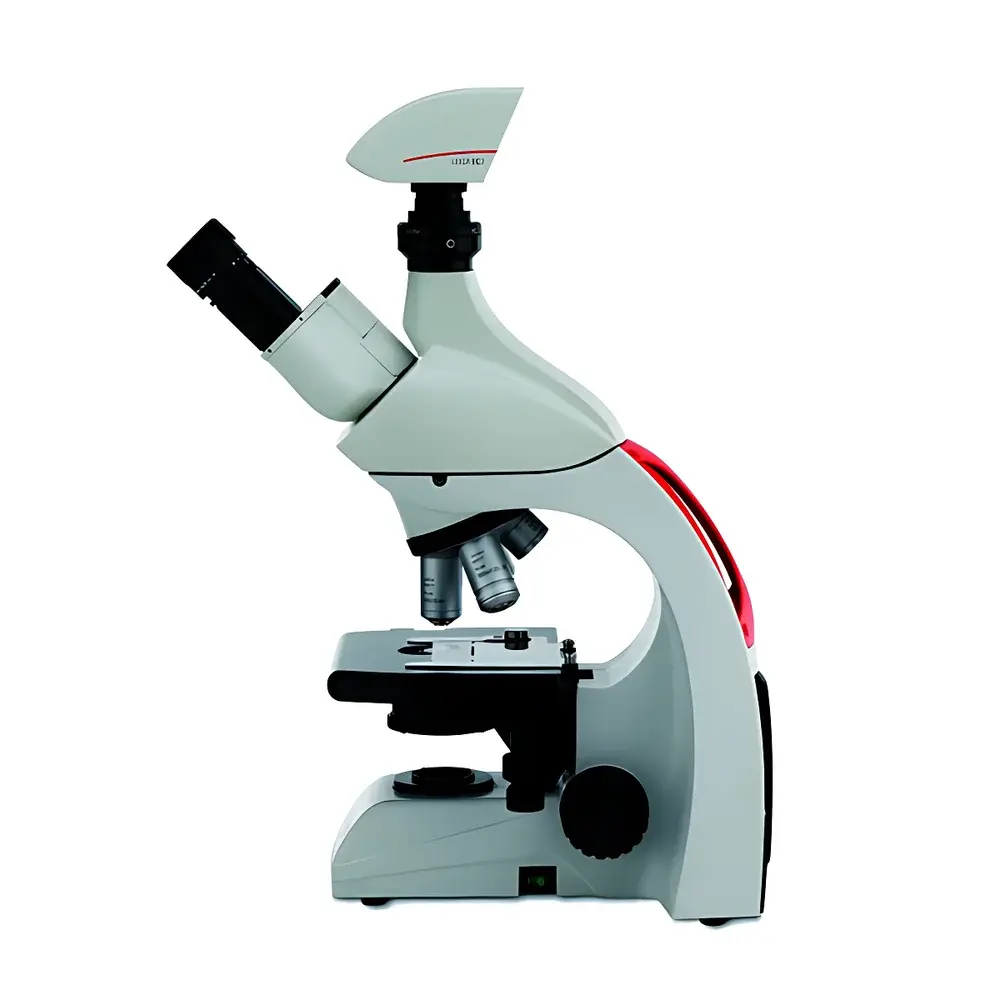
LKPT Digital Pathology Slide Scanner by LEI-TECH
| Brand | LEI-TECH |
|---|---|
| Origin | Tianjin, China |
| Manufacturer Type | Direct Manufacturer |
| Product Category | Domestic |
| Model | LKPT |
| Microscope Type | Upright Microscope |
| Scanning Modes | Brightfield, Darkfield, Phase Contrast, Polarized Light |
| Output Formats | TIFF, SVS, JPG, BMP, PNG |
| Shadow & Vignetting Correction | Active Compensation |
| Illumination Flicker Compensation | Active Real-time Compensation |
| Focus Maintenance | Real-time Z-axis Refocusing During Scan |
| Multi-Tissue Stitching | Single-Image Integration of Discrete Specimens |
| Camera Parameter Management | Preset Storage & Semi-Automatic/Full-Automatic Adjustment |
| Warranty | 24 Months |
| Service Support | 7×24 Remote & On-Site Response |
Overview
The LKPT Digital Pathology Slide Scanner, engineered by LEI-TECH Optics, is a high-fidelity upright microscope-based whole-slide imaging (WSI) system designed for clinical pathology laboratories, academic research centers, and biobanking facilities. It employs motorized precision stage translation coupled with real-time autofocus algorithms to maintain optimal focal plane integrity across large-area tissue sections—eliminating post-stitching defocus artifacts common in legacy scanners. The system integrates a scientific-grade monochrome or color CMOS camera with programmable exposure control, synchronized with LED illumination modules delivering stable, flicker-free output (±0.3% intensity variation over 10 s, per IEC 61000-4-15). Unlike conventional static-focus WSI platforms, the LKPT implements closed-loop z-height tracking during continuous scanning, enabling consistent sub-micron optical section fidelity across heterogeneous tissue morphologies—including thick sections, unevenly mounted slides, or coverslip-varied preparations.
Key Features
- Dynamic Focus Maintenance: Proprietary real-time Z-tracking algorithm continuously adjusts objective focus based on surface topography feedback, ensuring diffraction-limited resolution (≥0.22 µm lateral at 40×) throughout full-scan acquisition.
- Multi-Modal Imaging Support: Native compatibility with brightfield, darkfield, phase contrast, and polarized light microscopy—facilitating routine H&E, special stains (e.g., Masson’s trichrome), birefringence analysis (e.g., amyloid, collagen), and unstained cytology workflows.
- Optical Uniformity Assurance: Active shadow and vignetting correction applies pixel-wise gain mapping derived from flat-field calibration frames; illumination flicker compensation dynamically modulates LED drive current to counteract AC-line-induced intensity modulation.
- Flexible Tissue Integration: Multi-region stitching engine aligns and fuses non-contiguous tissue fragments (e.g., biopsy cores, excision margins, lymph node sections) into a single georeferenced SVS container without geometric distortion or chromatic shift.
- Parameter Reproducibility Framework: Up to 16 user-defined camera presets—including exposure time, gain, white balance, gamma, and binning—can be saved, recalled, and deployed across scanning sessions to enforce inter-operator and inter-day consistency.
Sample Compatibility & Compliance
The LKPT accommodates standard 1″ × 3″ (25 × 75 mm) glass slides with thicknesses ranging from 0.9 to 1.2 mm, including those with polymer-coated or charged surfaces. It supports both conventional coverslipped preparations and un-coverslipped frozen sections. All image acquisition parameters comply with DICOM Supplement 145 (Whole Slide Imaging) and are exportable in OpenSlide-compatible SVS format—enabling seamless integration with PACS, LIS, and AI-assisted diagnostic platforms. The system adheres to ISO 13485:2016 quality management requirements for medical device manufacturers and meets electromagnetic compatibility standards per GB/T 18268.1–2010 (IEC 61326-1 equivalent). For regulated environments, audit trail logging, user access controls, and electronic signature support can be enabled to satisfy FDA 21 CFR Part 11 and GLP/GMP documentation requirements.
Software & Data Management
The bundled LEI-SCAN™ acquisition suite provides intuitive workflow-driven interface navigation, supporting batch scanning, region-of-interest (ROI) marking, and metadata tagging (e.g., specimen ID, staining protocol, pathologist ID). Image data is stored in hierarchical directory structures compliant with CAP checklist ANP.42100 (digital pathology validation). Exported SVS files embed embedded pyramid layers (level 0–10), ICC profiles, and EXIF-compliant acquisition metadata—including objective magnification, numerical aperture, scan resolution (0.25–1.0 µm/pixel), and illumination settings. Raw TIFF sequences and JPEG previews are concurrently generated for rapid QA review. Data integrity is ensured via MD5 checksum generation upon file commit and optional AES-256 encryption for network transmission.
Applications
- Clinical telepathology consultation and second-opinion services
- AI model training and validation using annotated whole-slide datasets
- Longitudinal tissue archive digitization for retrospective cohort studies
- Education and collaborative case review in multi-institutional tumor boards
- Quality assurance of immunohistochemistry staining uniformity across batches
- Quantitative morphometric analysis of nuclear pleomorphism, mitotic index, or stromal-tumor ratio
FAQ
Does the LKPT support fluorescence scanning?
No—the current LKPT platform is optimized for transmitted-light modalities only. Fluorescence-capable variants (LKPT-F) are under development and scheduled for Q4 2025 release.
Can SVS files generated by the LKPT be imported into commercial digital pathology viewers?
Yes—all SVS outputs conform to Aperio’s published specification v12.1 and are natively supported by Philips IntelliSite, Hamamatsu NanoZoomer, 3DHISTECH CaseViewer, and open-source tools such as QuPath and ASAP.
Is remote diagnostic use permitted under Chinese NMPA or EU CE regulations?
The LKPT is registered as a Class I medical device under NMPA (Registration No. TJ2023XXXXX) and carries CE marking under MDR 2017/745 Annex VIII, Rule 10. Clinical diagnostic interpretation remains the responsibility of licensed pathologists; the system serves as an imaging tool, not an autonomous diagnostic device.
What is the maximum slide throughput per hour?
At 40× magnification and 0.25 µm/pixel resolution, average throughput is 8–10 slides/hour (including auto-focus, preview, and full-scan), depending on tissue area and staining density.
How is calibration traceability maintained?
Each unit ships with NIST-traceable stage calibration slides and integrated software tools for daily focus offset verification, illumination uniformity mapping, and color fidelity validation using ISO 17025-accredited reference targets.