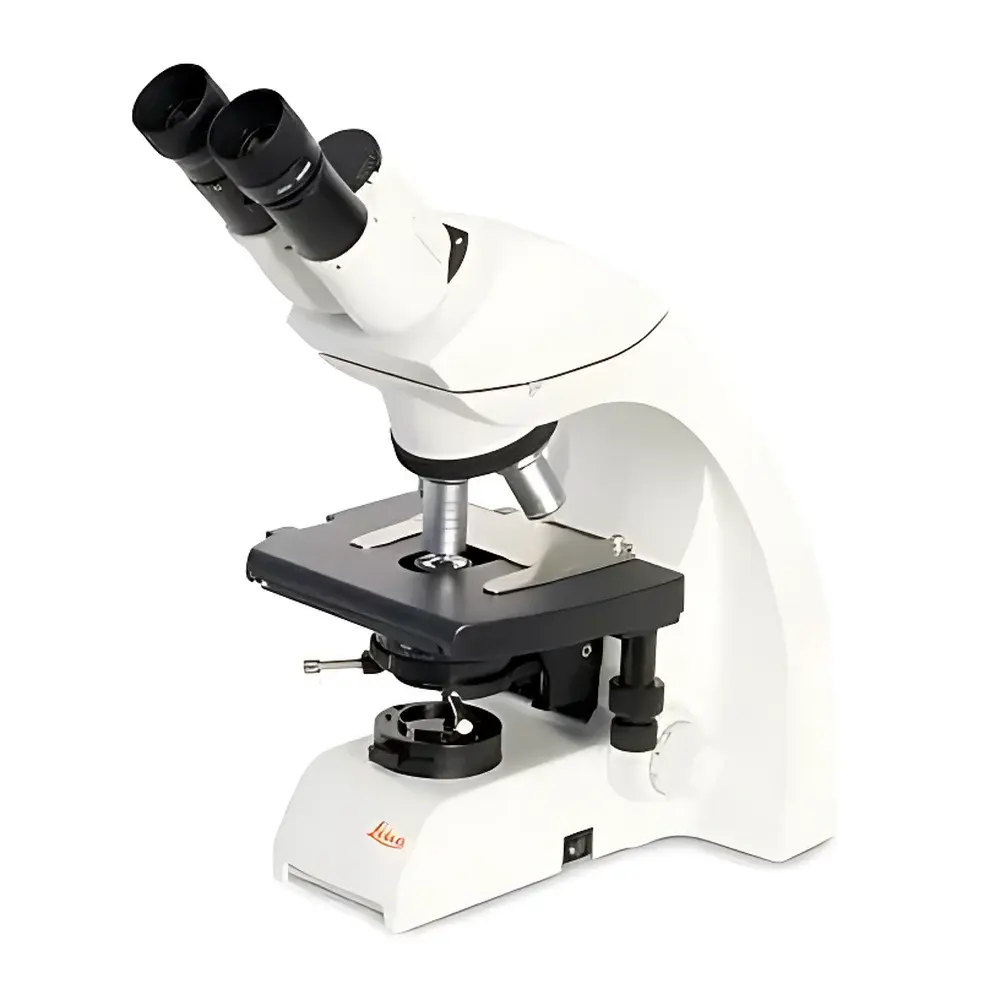
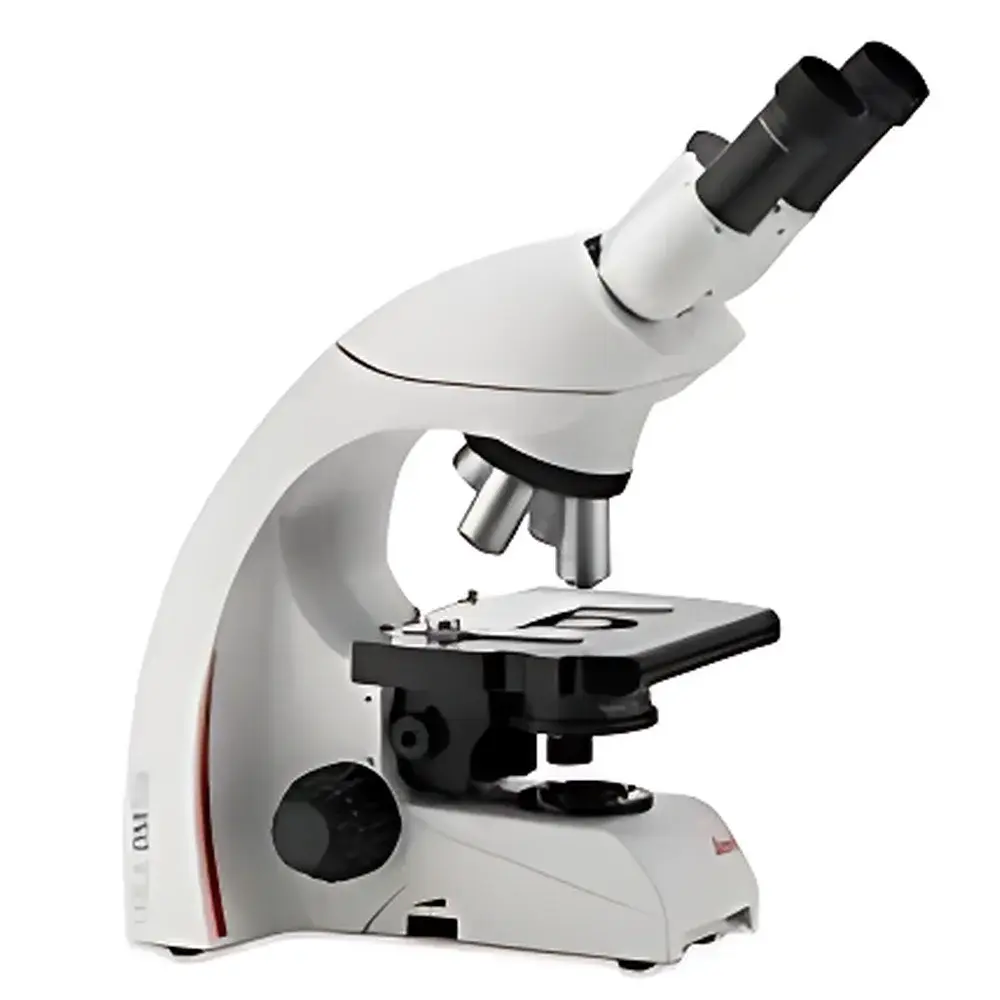
Leica DM750 Binocular Biological Microscope
| Brand | Leica |
|---|---|
| Origin | Germany |
| Model | DM750 |
| Illumination | Integrated LED (White, Cold Light) |
| Objective Lenses | Includes 100× Dry Objective (NA 0.8) |
| Stage | SafeTStage™ Self-Supporting Mechanical Stage |
| Eyepiece System | EZTube™ Integrated Eyepiece Tube with Preset Diopter Adjustment |
| Condenser | Pre-centered & Pre-focused Abbe Condenser with 4-Position Phase Contrast Slider (Brightfield/Phase Contrast Capable) |
| Ergonomic Design | EZStore™ Vertical Handle, EZGuide™ Slide Rest, EZLite™ Long-Life LED, AgTreat™ Antimicrobial Surface Treatment |
| Compliance | Designed for GLP/GMP-aligned educational and routine QC labs |
| Power | AC Adapter or Optional Battery Charging Stand |
Overview
The Leica DM750 binocular biological microscope is an entry-level yet rigorously engineered instrument designed for routine life science education, clinical training, and quality control laboratories requiring robust optical performance, long-term reliability, and user-centric ergonomics. Built on Leica’s proven optical platform shared with higher-tier research microscopes, the DM750 delivers high-contrast, color-accurate imaging across the full visible spectrum (400–700 nm) using fully corrected achromatic objectives. Its optical architecture follows standard Köhler illumination principles, ensuring uniform field illumination and optimal resolution—critical for consistent morphological assessment of unstained or lightly stained biological specimens such as blood smears, tissue sections, algae, protozoa, and cultured cells. Unlike conventional student microscopes, the DM750 incorporates design elements typically reserved for professional-grade systems—including a pre-aligned Abbe condenser, parfocal and parcentral optics, and a stable, vibration-damped base—making it suitable for extended daily use in teaching labs and diagnostic support environments.
Key Features
- EZTube™ Integrated Eyepiece System: Permanently affixed eyepiece tube eliminates accidental disconnection; factory-set diopter compensation ensures consistent visual acuity across users without manual reconfiguration.
- 100× Dry Objective (NA 0.8): Eliminates immersion oil handling while maintaining high numerical aperture—ideal for rapid screening and reducing cross-contamination risk in shared lab settings.
- EZLite™ LED Illumination: Solid-state white LED provides flicker-free, cool illumination with >15,000 hours lifetime (≈15 years at 3 hrs/day); intensity is continuously adjustable via front-panel rheostat without heat-induced specimen drying.
- SafeTStage™ Self-Supporting Mechanical Stage: Fully enclosed, low-profile stage construction removes protruding mechanical linkages—minimizing pinch hazards and improving safety during slide manipulation.
- AgTreat™ Antimicrobial Surface Treatment: Silver-ion additive integrated into all high-contact surfaces (focus knobs, stage controls, eyepiece housing) inhibits >99% of common Gram-positive and Gram-negative bacteria per ISO 22196, supporting biosafety compliance in multi-user environments.
- EZStore™ Transport & Storage Design: Vertically oriented handle and recessed front groove enable two-handed lifting; vertical power cable insertion prevents strain on internal connectors during storage or relocation.
Sample Compatibility & Compliance
The DM750 accommodates standard 1″ × 3″ (25 × 76 mm) glass slides and 0.17 mm cover slips. Its Abbe condenser (NA 1.25) supports brightfield, phase contrast (via included 4-position slider), and optional darkfield observation. The system meets IEC 61000-6-3 (EMC emissions) and IEC 61000-6-1 (immunity) standards. While not certified for ISO 13485 or FDA 21 CFR Part 11 out-of-the-box, its audit-ready mechanical stability, traceable calibration landmarks (e.g., fixed stage micrometer scale), and non-volatile focus position memory support documentation workflows required under GLP and GMP-aligned laboratory practices.
Software & Data Management
The DM750 operates as a standalone optical instrument without embedded digital capture. However, its standardized C-mount (23.2 mm) port enables seamless integration with third-party CMOS cameras (e.g., Leica ICC50 HD, Lumenera Infinity3) and image analysis platforms including ImageJ, NIS-Elements, and Olympus cellSens. All mechanical adjustments—including coarse/fine focus travel, stage X/Y coordinates, and condenser height—are fully manual and mechanically indexed, ensuring reproducibility without software dependency. For institutions requiring electronic record retention, optional USB-powered camera modules provide time-stamped image export compatible with LIMS and ELN systems.
Applications
- Undergraduate and graduate histology, microbiology, and botany instruction
- Routine hematology and urinalysis in clinical training labs
- QC inspection of cell culture morphology and confluence in biomanufacturing support labs
- Environmental monitoring of planktonic organisms and biofilm structures
- Pharmaceutical raw material identification (e.g., pollen, fungal spores, plant tissues)
- Veterinary parasitology screening (e.g., fecal floatation, skin scrapings)
FAQ
Is the DM750 compatible with digital imaging systems?
Yes—the microscope features a standardized C-mount interface (23.2 mm) and coaxial trinocular port option, enabling direct coupling to industry-standard USB or HDMI cameras.
Does the 100× dry objective require oil immersion?
No—this proprietary objective achieves NA 0.8 without immersion oil, eliminating cleaning steps and contamination risks associated with oil-based microscopy.
Can the DM750 be used for phase contrast imaging?
Yes—its Abbe condenser includes a dedicated slot for the included 4-position phase contrast slider, supporting both brightfield and multiple phase contrast configurations (e.g., Ph1, Ph2) without realignment.
What is the expected service life of the LED illumination system?
Rated for ≥15,000 operating hours under normal usage conditions (3 hours/day, 5 days/week), with no measurable lumen depreciation over the first 10,000 hours per LM-80 test data.
Is AgTreat™ treatment validated per international antimicrobial standards?
Yes—surface efficacy testing was conducted per ISO 22196:2011 (Measurement of antibacterial activity on plastics and other non-porous surfaces), demonstrating ≥99.9% reduction of Staphylococcus aureus and Escherichia coli after 24-hour contact.