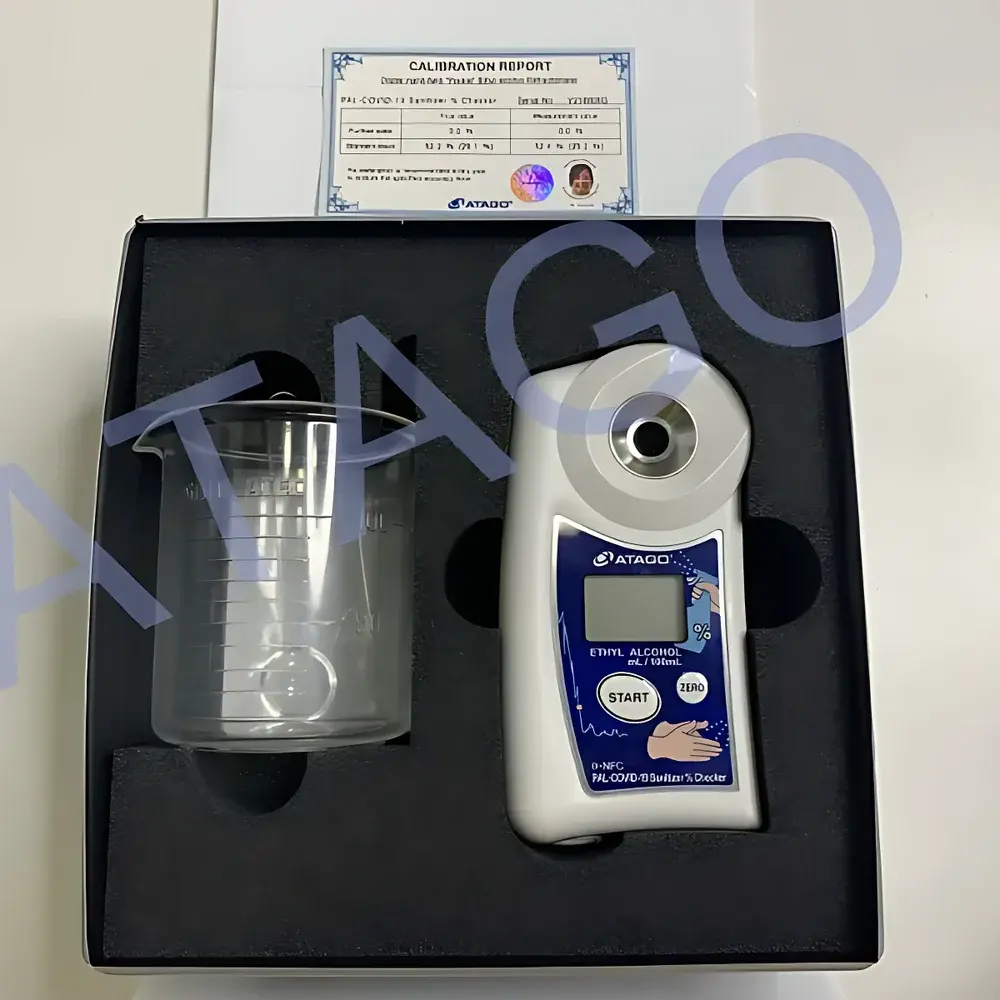
ATAGO PAL-COVID-19 Ethanol-Specific Handheld Refractometer
| Brand | ATAGO |
|---|---|
| Origin | Japan |
| Model | PAL-COVID-19 |
| Product Type | Handheld Refractometer |
| Temperature Control | None |
| Digital Display | Yes |
| Measurement Range | 0.0–100.0% (v/v) Ethanol |
| Accuracy | ±1.0% (v/v) for undiluted ethanol solutions |
Overview
The ATAGO PAL-COVID-19 is a dedicated handheld refractometer engineered for rapid, on-site quantification of ethanol concentration in aqueous disinfectant solutions. It operates on the principle of critical-angle refractometry: incident light undergoes total internal reflection at the prism-sample interface, with the resulting shadow line position correlated to the solution’s refractive index—linearly calibrated against ethanol volume fraction (v/v) across the full 0.0–100.0% range. Unlike generic alcohol refractometers, the PAL-COVID-19 employs a custom optical calibration curve optimized specifically for ethanol–water binary systems, minimizing cross-sensitivity to common excipients (e.g., glycerol, hydrogen peroxide, surfactants) found in WHO-recommended or national-standardized disinfectant formulations. Its design responds directly to pandemic-era quality assurance requirements: verifying that hand sanitizers and surface disinfectants meet the minimum 75–80% (v/v) ethanol threshold proven effective against enveloped viruses—including SARS-CoV-2—while avoiding over-concentration (>90%) that compromises virucidal efficacy due to rapid surface protein coagulation and reduced intracellular penetration.
Key Features
- Proprietary ethanol-specific calibration validated across 0.0–100.0% (v/v), traceable to NIST-traceable ethanol reference standards
- Automatic temperature compensation (ATC) algorithm compensating for sample temperature drift between 10–40 °C, referenced to 20 °C standard
- High-resolution LCD with backlight for low-light environments (e.g., production floors, field deployment)
- Robust magnesium alloy housing rated IP65 for resistance to ethanol splash, dust ingress, and mechanical shock
- Single-button operation with auto-power-off after 3 minutes of inactivity to extend battery life (CR2032, ~10,000 measurements per cell)
- No reagents, no consumables, no warm-up time—measurement completed in <2 seconds post-sample application
Sample Compatibility & Compliance
The PAL-COVID-19 is validated for use with ethanol–water mixtures containing ≤5% (w/w) non-volatile additives typical in commercial disinfectants (e.g., glycerin ≤1.45%, hydrogen peroxide ≤0.125%, carbomer, fragrance). It is not intended for methanol-, isopropanol-, or denatured-alcohol-based formulations. The instrument conforms to JIS K 0064 (Japanese Industrial Standard for refractometric analysis), and its measurement uncertainty profile supports compliance with ISO/IEC 17025:2017 clause 7.6.3 for in-house method validation. While not a regulated medical device under FDA 21 CFR Part 820 or EU MDR, its performance data aligns with WHO Technical Report Series No. 1017 Annex 5 (2020) and CDC Guideline for Disinfection and Sterilization (2023), which specify ethanol concentration verification as a critical control point in disinfectant manufacturing and hospital pharmacy compounding.
Software & Data Management
The PAL-COVID-19 operates as a standalone field instrument without embedded data logging or Bluetooth connectivity. However, its analog output (0–1 V DC proportional to % v/v) enables integration with external PLCs or analog data acquisition systems for continuous process monitoring in batch QC stations. For audit-ready documentation, users may pair the device with ATAGO’s optional RS-232 interface cable (PAL-IF-2) and PC software *RefractoLink*, enabling export of timestamped measurement records (.csv) compliant with GLP documentation requirements—including operator ID, ambient temperature, and instrument serial number. All calibration certificates issued by ATAGO Japan include uncertainty budgets calculated per GUM (JCGM 100:2008) and are valid for 12 months under normal usage conditions.
Applications
- Verification of final ethanol concentration in WHO-recommended hand rub formulations (Formulation I: 80% v/v ethanol; Formulation II: 75% v/v ethanol)
- In-process testing during industrial-scale disinfectant blending to prevent batch rejection due to out-of-spec ethanol content
- Field inspection by regulatory auditors (e.g., NMPA, PMDA, ANVISA) validating label claims on commercial antiseptic products
- Quality control in hospital pharmacies preparing compounded alcohol-based sanitizers under USP or guidelines
- Educational labs demonstrating colligative property relationships between refractive index and solute concentration in physical chemistry courses
FAQ
Does the PAL-COVID-19 require recalibration before each use?
No. Factory calibration is stable for 12 months under routine handling; verification with 0% and 75% (v/v) ethanol reference standards is recommended daily in high-frequency QC environments.
Can it measure ethanol in solutions containing >5% glycerol?
Accuracy degrades beyond ±5% (v/v) glycerol due to refractive index additivity effects; for such matrices, ATAGO recommends the digital Abbe refractometer RX-5000α with multi-component calibration.
Is temperature control built into the instrument?
No active thermostatic control is present, but ATC correction ensures measurement repeatability within ±0.3% (v/v) across 10–40 °C sample temperatures.
How does its accuracy compare to gas chromatography (GC)?
GC remains the definitive reference method (±0.1% v/v); the PAL-COVID-19 provides field-deployable screening with ±1.0% v/v accuracy—sufficient for release testing per ISO 15223-1:2021 Annex B.
Is the prism surface resistant to ethanol-induced erosion?
Yes. The sapphire prism (Mohs hardness 9) withstands repeated ethanol exposure without measurable abrasion or refractive index drift over 5+ years of daily use.