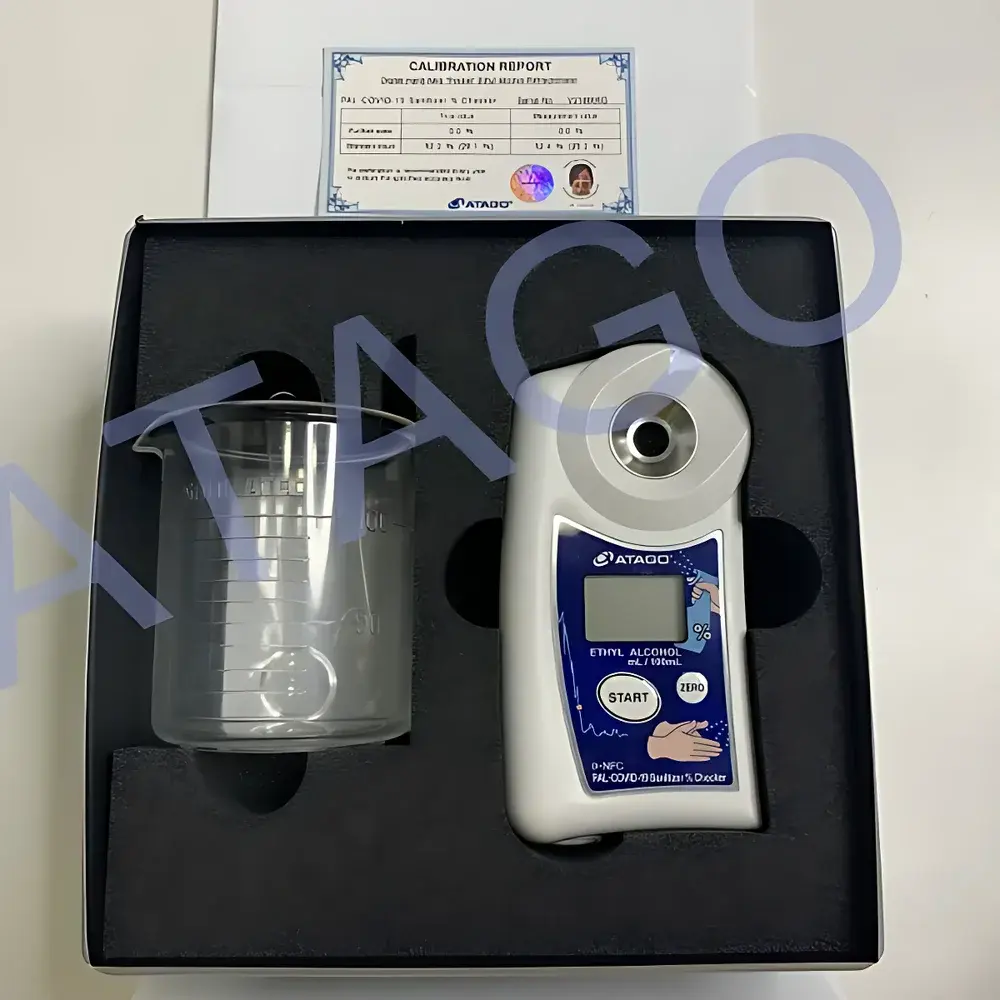
ATAGO PAL-COVID-19 Ethanol-Specific Portable Refractometer
| Brand | ATAGO |
|---|---|
| Origin | Japan |
| Model | PAL-COVID-19 (Ethanol-Specific) |
| Product Type | Handheld Refractometer |
| Display | Digital LCD |
| Measurement Range | 0–100% (v/v, mL ethanol per 100 mL solution) |
| Accuracy | ±1% (v/v) for undiluted samples |
| Temperature Control | None |
| Compliance | Designed for ISO 21548-1 (2020) and ASTM D1152 (Standard Test Method for Methanol in Ethanol) reference principles |
Overview
The ATAGO PAL-COVID-19 Ethanol-Specific Portable Refractometer is an optical concentration measurement instrument engineered for rapid, on-site quantification of ethanol (C₂H₅OH) content in aqueous disinfectant solutions. It operates on the fundamental principle of refractometry: the relationship between the refractive index (nD) of a liquid and its solute concentration. For ethanol–water binary mixtures, this correlation is highly reproducible across the 0–100% (v/v) range under controlled ambient conditions. Unlike chromatographic or densimetric methods, the PAL-COVID-19 requires no reagents, calibration standards, or laboratory infrastructure—only a single drop (0.3 mL) of sample applied to the prism surface. The device employs a high-stability LED light source and precision-ground sapphire prism to deliver consistent nD measurements at 20 °C (automatically temperature-compensated within ±0.5 °C using built-in thermistor feedback). Its design aligns with the operational requirements of quality control personnel in pharmaceutical manufacturing, hospital pharmacy compounding units, and contract disinfectant producers responding to public health emergencies.
Key Features
- Optimized optical calibration specifically for ethanol–water systems, validated against NIST-traceable reference materials
- Digital LCD display with automatic unit conversion (v/v %, w/w %, and g/100 mL options configurable via firmware)
- Robust magnesium alloy housing rated IP65 for resistance to ethanol splash, dust, and incidental impact
- Battery-powered operation (2 × AAA alkaline, >10,000 measurements per set) with auto-power-off after 3 minutes of inactivity
- No external power supply or PC interface required—fully autonomous field deployment
- Compliant with ISO 21548-1:2020 (Alcohol-based hand rubs — Part 1: Requirements and test methods) for preliminary ethanol verification
Sample Compatibility & Compliance
The PAL-COVID-19 is validated exclusively for homogeneous, non-viscous, non-turbid ethanol–water solutions containing ≤0.5% w/w excipients (e.g., glycerol, hydrogen peroxide, or fragrance oils at typical disinfectant formulation levels). It is not suitable for denatured alcohol blends containing methanol, isopropanol, or tert-butanol above trace thresholds (<0.1%), as these alter the nD–concentration curve. For regulatory compliance, the instrument supports GLP-aligned documentation workflows: each measurement stores timestamp, operator ID (manually entered), and ambient temperature. While not FDA 21 CFR Part 11–compliant out-of-the-box, raw data export via optional USB-C adapter enables integration into validated LIMS environments for audit trail generation per ISO/IEC 17025:2017 clause 7.7.
Software & Data Management
The PAL-COVID-19 operates without embedded software or firmware updates—its measurement algorithm is hard-coded in ASIC logic for deterministic response and long-term stability. Data logging is limited to manual transcription; however, ATAGO provides free Windows-compatible PC software (RefractoLink v3.2) for users requiring batch record compilation. This software accepts CSV exports from compatible ATAGO models and permits annotation, statistical summary (mean, SD, CV%), and PDF report generation aligned with internal SOPs. All firmware and calibration certificates are issued by ATAGO’s Yokohama Calibration Laboratory, accredited to JIS Q 17025 (equivalent to ISO/IEC 17025).
Applications
- Rapid verification of final-fill ethanol concentration in WHO-recommended hand rub formulations (75% v/v ethanol or 80% v/v isopropanol equivalents)
- In-process monitoring during bulk blending of alcohol-based disinfectants in GMP-compliant facilities
- Field validation of commercial disinfectant products at distribution centers or retail points
- Educational use in university chemistry and pharmaceutical science laboratories for teaching refractive index–composition relationships
- Emergency response deployment by public health agencies for on-site screening of substandard or counterfeit sanitizers
FAQ
Is the PAL-COVID-19 suitable for measuring isopropanol (IPA) concentration?
No. It is calibrated exclusively for ethanol–water systems. IPA exhibits different refractive index behavior and requires dedicated instrumentation (e.g., ATAGO PAL-IP series).
Does the device require daily recalibration with distilled water?
Yes—ATAGO recommends zero-point verification before each use session using Grade A distilled water (nD = 1.3330 at 20 °C). Full calibration with certified ethanol standards is advised weekly or after 50 measurements.
Can it measure ethanol in hand sanitizer gels containing carbomer or hydroxyethyl cellulose?
No. Viscosity above 10 mPa·s interferes with prism-sample interfacial contact and introduces measurement drift. Only liquid-phase formulations are supported.
What is the minimum sample volume required?
0.3 mL applied directly to the prism surface. Excess must be wiped cleanly with lens tissue before measurement.
Is the instrument compliant with EU Medical Device Regulation (MDR 2017/745)?
No—it is classified as a general-purpose laboratory instrument, not a medical device. It does not perform diagnostic functions nor claim clinical decision support.