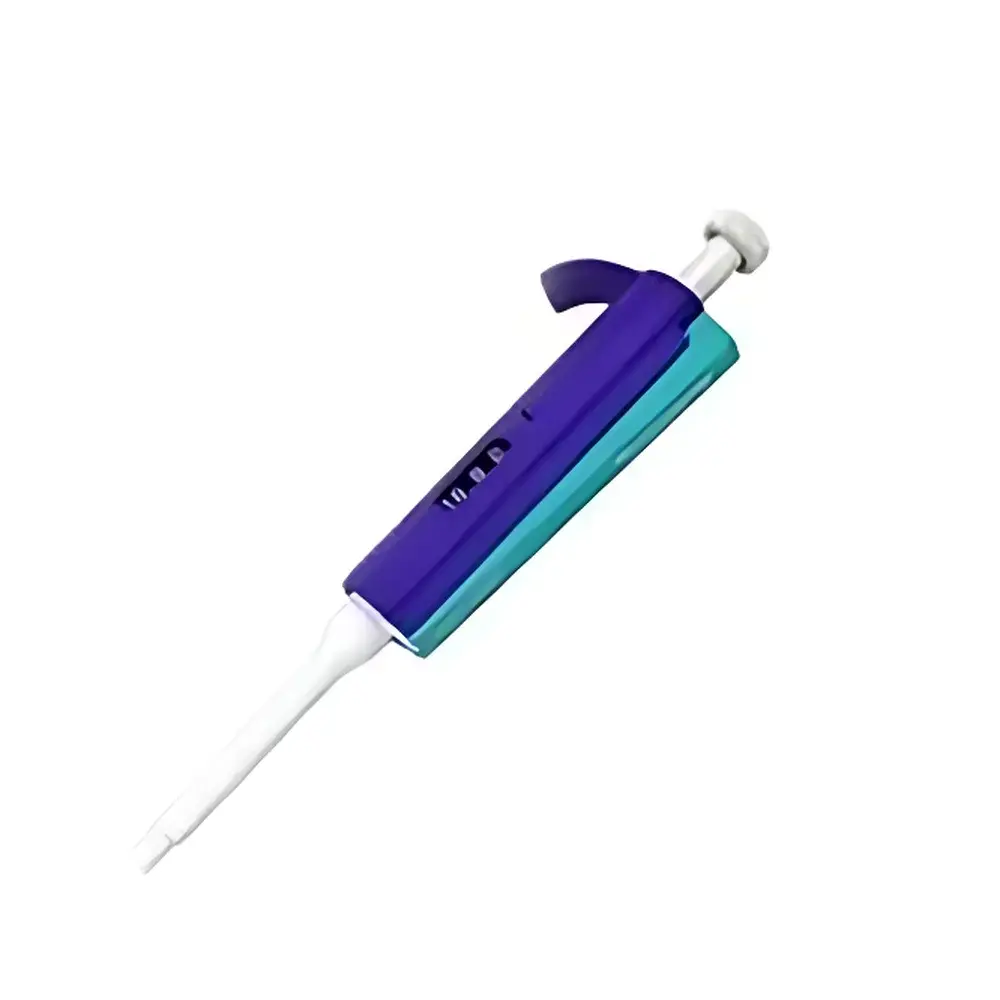
Thermo Scientific Finnpipette Color Single-Channel Manual Pipette, 100–1000 µL
| Key | Brand: Thermo Scientific Finnpipette |
|---|---|
| Type | Single-channel manual pipette |
| Volume Range | 100–1000 µL |
| Maximum Capacity | 1000 µL |
| Accuracy | ±2.5% |
| Sterilization | Half-body autoclavable |
| Origin | Manufactured in Jiangsu, China under Thermo Fisher Scientific licensing and quality oversight |
Overview
The Thermo Scientific Finnpipette Color Single-Channel Manual Pipette (100–1000 µL) is an ergonomically engineered precision liquid handling instrument designed for high-frequency, repetitive pipetting tasks in molecular biology, clinical diagnostics, pharmaceutical QC, and academic research laboratories. Based on the globally recognized Finnpipette platform—originally developed by Labconco and now fully integrated into Thermo Fisher Scientific’s portfolio—this model combines ISO 8655-compliant piston-driven air displacement technology with human-centered mechanical design. Its operating principle relies on controlled vertical movement of a stainless-steel piston within a sealed chamber to aspirate and dispense liquid volumes via disposable polypropylene tips. The device delivers traceable accuracy and repeatability across its full range, with performance validated per ISO 8655-2 (volumetric accuracy) and ISO 8655-3 (repeatability), ensuring compliance with GLP, GMP, and FDA 21 CFR Part 11 requirements when used within documented laboratory workflows.
Key Features
- Ergonomic design with low-force plunger activation (≤1.5 N for full stroke at 1000 µL), reducing operator hand fatigue and risk of repetitive strain injury (RSI) during extended use.
- Color-coded volume adjustment dial and tip ejector lever for intuitive identification and rapid setup—standardized across the Finnpipette Color series for consistent lab-wide operation.
- Patented “Super Blow-out” piston mechanism featuring a tapered, spring-loaded seal that minimizes capillary retention and ensures complete tip emptying—even with viscous or volatile liquids such as glycerol, ethanol, or serum.
- Half-body autoclavable construction (up to 121 °C, 20 min, 1 bar) without disassembly; critical O-rings and seals retain integrity after ≥20 sterilization cycles.
- Integrated tip compatibility with universal low-retention and filtered tips (e.g., Thermo Scientific Finntip, LTS, or compatible third-party options), supporting applications requiring RNase/DNase-free or sterile conditions.
- Mechanically calibrated volume lock prevents accidental setting drift; fine-tuning screw allows field recalibration using certified gravimetric standards.
Sample Compatibility & Compliance
This pipette is validated for aqueous solutions, buffers, cell culture media, diluted acids/bases (pH 2–12), and common organic solvents (e.g., ≤70% ethanol, isopropanol). It is not recommended for highly corrosive liquids (e.g., concentrated HCl, HF), halogenated hydrocarbons, or strong oxidizers without prior compatibility testing. All materials in contact with liquid pathways—including piston, seal, and tip cone—are chemically inert polyether ether ketone (PEEK), PTFE, and medical-grade stainless steel, complying with USP Class VI biocompatibility and EU Directive 2002/72/EC for food contact. Device documentation includes full ISO 8655 test reports, CE marking (Class I, Annex VII), and conformity to RoHS 2011/65/EU and REACH Regulation (EC) No. 1907/2006.
Software & Data Management
As a manually operated instrument, this pipette does not incorporate embedded electronics or firmware. However, it supports full integration into digital lab ecosystems through optional accessories: Thermo Scientific Pipette Tracker software enables barcode-based asset management, calibration scheduling, and audit trail generation for each unit. When paired with Thermo Scientific CalCheck gravimetric verification kits and automated balance interfaces, users can generate PDF reports compliant with ISO/IEC 17025 and internal SOPs. All calibration records—including date, technician ID, standard used (NIST-traceable weights), and deviation data—are exportable in CSV or XML format for LIMS ingestion and regulatory inspection readiness.
Applications
- Quantitative preparation of PCR master mixes, qRT-PCR reaction plates, and NGS library normalization steps.
- Serial dilution workflows in ELISA, cytotoxicity assays, and antimicrobial susceptibility testing (AST).
- Cell seeding and media supplementation in adherent and suspension cultures under ISO 5 cleanroom conditions.
- Standard curve generation for HPLC, GC, and mass spectrometry sample introduction protocols.
- Quality control release testing of raw materials and finished drug products per USP , , and .
- Environmental monitoring in water testing labs performing EPA Method 1603/1604 coliform enumeration.
FAQ
Is this pipette manufactured by Thermo Fisher Scientific or a licensed third party?
This instrument is produced under strict Thermo Fisher Scientific quality license agreements in Jiangsu Province, China. Final assembly, calibration, and certification are performed per Thermo’s global manufacturing specifications (QMS-THM-001), and each unit bears the Thermo Scientific Finnpipette logo and serial-numbered calibration certificate.
What is the recommended recalibration interval?
Per ISO 8655-5, recalibration is advised every 3–6 months for routine use, or immediately following impact, exposure to extreme temperatures (>40 °C), or after 10,000 actuations—whichever occurs first. Calibration must be performed using NIST-traceable analytical balances and certified distilled water at 20 °C.
Can this pipette be fully autoclaved?
No. Only half-body autoclaving (lower body including tip cone and ejector) is permitted. The upper housing contains non-autoclavable elastomeric components and must remain unexposed to steam. Full sterilization requires alternative methods such as ethylene oxide (EtO) gas or vaporized hydrogen peroxide (VHP), subject to validation.
Does it support electronic data capture for 21 CFR Part 11 compliance?
The pipette itself lacks onboard memory or connectivity. However, when used with Thermo Scientific Pipette Tracker and calibrated via Thermo-certified balances with audit trail logging, the associated calibration and usage records meet ALCOA+ data integrity principles required for FDA-regulated environments.
Are replacement parts available globally?
Yes. Genuine Thermo Scientific Finnpipette spare parts—including piston assemblies, O-ring kits (P/N 4027641), and tip cones—are distributed through authorized Thermo Fisher channels in >120 countries, with typical lead time of 3–5 business days from regional distribution centers.