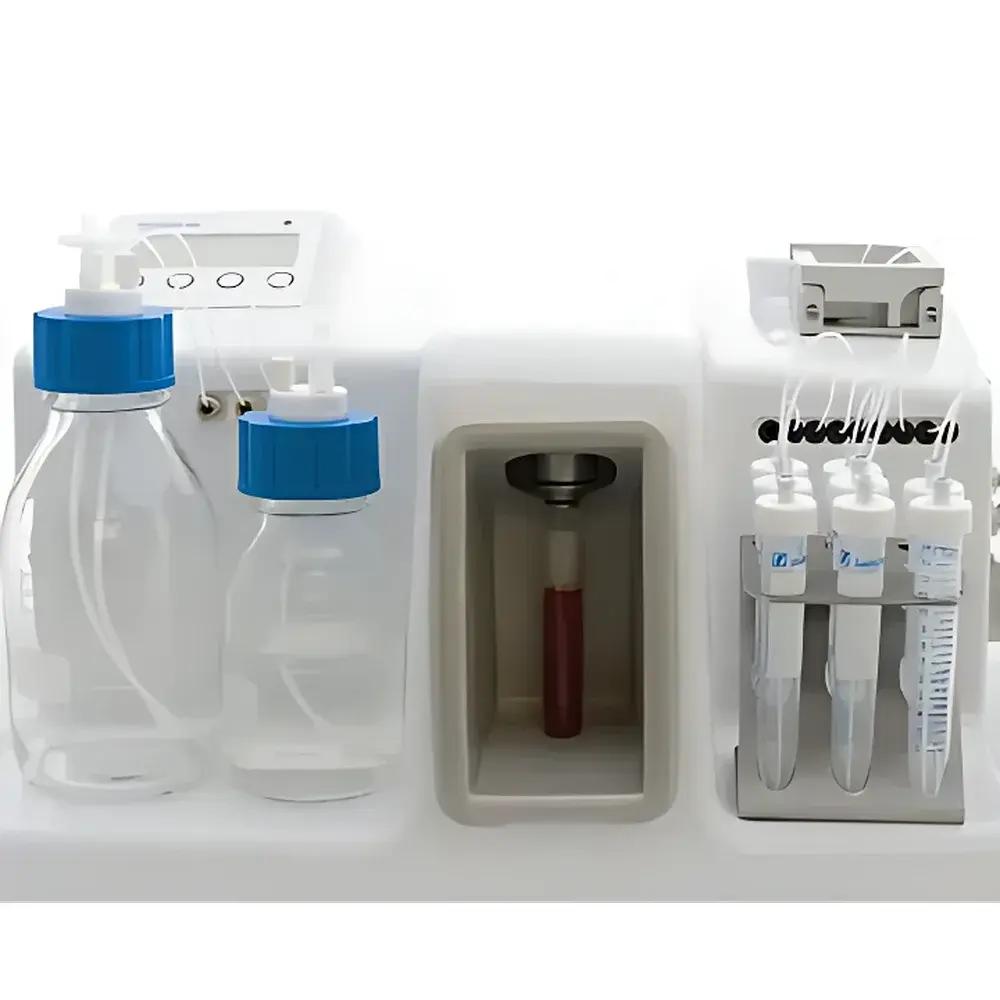
ANGLE Parsortix PR1 Automated Circulating Tumor Cell (CTC) Isolation System
| Brand | ANGLE |
|---|---|
| Origin | United Kingdom |
| Manufacturer Type | Authorized Distributor |
| Origin Category | Imported Instrument |
| Model | Parsortix PR1 |
| Pricing | Available Upon Request |
| Separation Method | Fully Automated Microfluidic-Based Capture |
Overview
The ANGLE Parsortix PR1 Automated Circulating Tumor Cell (CTC) Isolation System is an FDA-cleared, CE-marked microfluidic platform engineered for label-free, size- and deformability-based enrichment of viable circulating tumor cells from peripheral blood. Unlike antibody-dependent methods (e.g., EpCAM-based immunomagnetic capture), the Parsortix system leverages intrinsic biophysical properties—specifically larger diameter (typically >12 µm) and reduced deformability—of CTCs relative to leukocytes (<10 µm) and erythrocytes (<8 µm). Blood samples are processed under controlled laminar flow through a proprietary disposable microfluidic cassette containing precisely engineered silicon microstructures. This architecture induces deterministic lateral displacement and selective mechanical retention of CTCs while permitting unimpeded passage of smaller, more deformable hematologic cells. The result is a high-purity, functionally intact CTC population suitable for morphological assessment, molecular profiling, and functional assays without epitope masking or antibody-induced activation artifacts.
Key Features
- Fully automated sample processing: Integrated fluidics, pressure control, and real-time monitoring minimize manual intervention and operator variability.
- Label-free isolation: Eliminates reliance on surface biomarkers, enabling recovery of EpCAM-negative, mesenchymal, or stem-like CTC subpopulations often missed by immunoaffinity platforms.
- Viable cell recovery: Gentle shear stress profile (85% post-isolation metabolic activity (via Calcein-AM/PI staining) and successful in vitro expansion in multiple cancer types.
- Disposable microfluidic cassettes: Sterile, single-use silicon-glass chips with integrated microgrooves ensure batch-to-batch consistency and eliminate cross-contamination risk; each cassette is barcoded for full traceability.
- Scalable throughput: Processes 7.5–10 mL of whole blood per run in ≤2.5 hours, compatible with standard EDTA or Streck BCT collection tubes.
- Integrated imaging-ready chamber: Captured CTCs remain immobilized within the cassette’s observation zone, enabling direct on-chip fixation, permeabilization, and multiplex immunofluorescence staining (e.g., cytokeratin+/CD45−/DAPI+ phenotyping) without transfer loss.
Sample Compatibility & Compliance
The Parsortix PR1 accepts fresh or refrigerated (4 °C, up to 96 h) whole blood collected in standard anticoagulant tubes (EDTA, citrate, or Streck Cell-Free DNA BCT). It has been validated across major solid tumor indications including metastatic breast, prostate, non-small cell lung, and colorectal carcinomas. Regulatory compliance includes CE IVD marking (Class D), FDA 510(k) clearance (K183029), and alignment with ISO 13485:2016 quality management requirements. The system supports audit-ready documentation per GLP and GCP frameworks, with electronic logs capturing operator ID, timestamp, cassette lot number, pressure profiles, and run completion status—facilitating traceability in clinical trial and companion diagnostic workflows.
Software & Data Management
The Parsortix Control Software (v4.2+) provides intuitive workflow-guided operation, real-time pressure and flow monitoring, and automated cassette calibration. All acquisition parameters—including inlet pressure, flow rate, and harvest duration—are logged in encrypted SQLite databases compliant with FDA 21 CFR Part 11 requirements (electronic signatures, audit trails, role-based access control). Image data from on-chip CTC enumeration can be exported in TIFF or OME-TIFF format for integration with third-party analysis tools (e.g., HALO, Visiopharm) or LIMS systems via HL7/FHIR interfaces. Raw metadata conforms to MIAME and MIAPE standards for translational research reproducibility.
Applications
- Longitudinal CTC monitoring in metastatic disease to assess treatment response and emerging resistance mechanisms.
- Single-cell RNA sequencing and whole-genome amplification following on-chip lysis or microdissection.
- Ex vivo drug sensitivity testing using recovered CTCs cultured in collagen-Matrigel matrices or organoid co-cultures.
- Biomarker discovery through parallel proteomic (IMC, CyTOF) and genomic (targeted NGS, digital PCR) interrogation of matched CTC and ctDNA fractions.
- Clinical trial enrichment: Stratifying patients for targeted therapy based on CTC-derived HER2, AR-V7, or PD-L1 expression status.
FAQ
Does the Parsortix system require pre-enrichment or red blood cell lysis?
No—whole blood is loaded directly without centrifugation, density gradient separation, or RBC lysis, preserving CTC viability and minimizing pre-analytical bias.
Can CTCs be recovered for downstream culture?
Yes—intact, non-fixed CTCs are eluted in low-volume PBS or culture medium; published protocols report successful 7–14 day expansion of prostate and breast CTC lines.
How is system performance verified between runs?
Each cassette includes built-in hydrodynamic calibration channels; software performs real-time pressure deviation checks and flags deviations >±5% from nominal setpoints.
Is the system compatible with frozen/thawed blood samples?
No—only fresh or refrigerated (not frozen) whole blood is validated; cryopreservation compromises CTC integrity and microfluidic capture efficiency.
What regulatory documentation is provided for clinical use?
FDA 510(k) summary, CE IVD Declaration of Conformity, ISO 13485 certificate, and analytical validation reports (precision, linearity, limit of detection) are available under NDA/NDA-equivalent agreements.