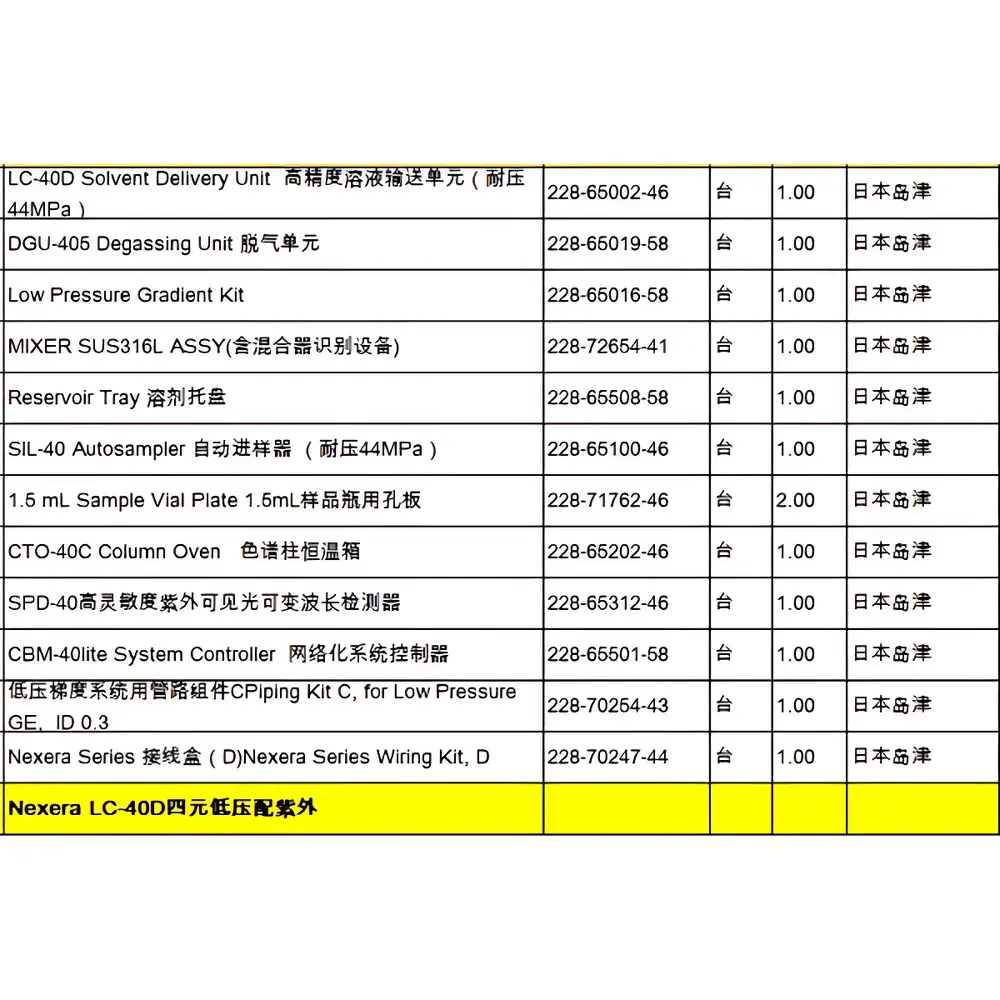
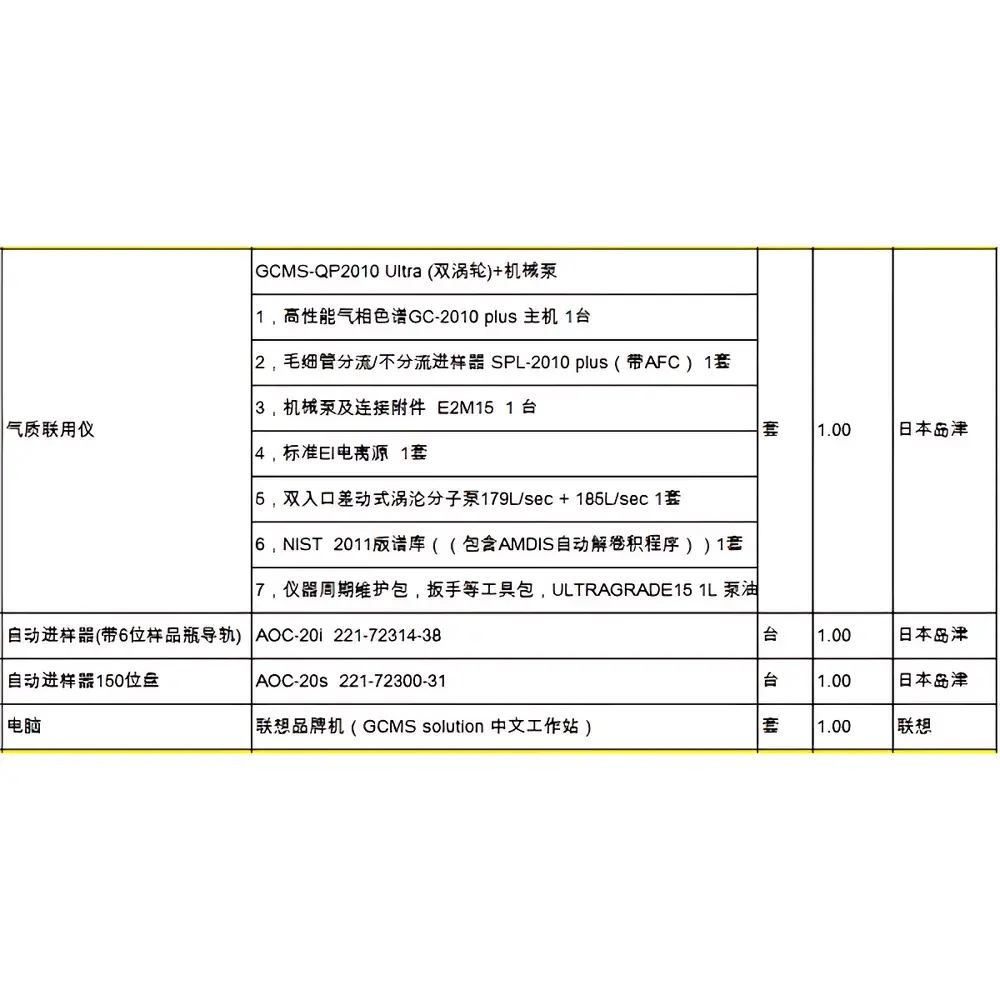
Shimadzu GCMS-TQ8050 Triple Quadrupole Gas Chromatography Mass Spectrometry System
| Brand | Shimadzu |
|---|---|
| Origin | Japan |
| Model | GCMS-TQ8050 |
| Year of Manufacture | 2018 |
| Condition | Refurbished & Certified Pre-Owned |
| Configuration | Complete Benchtop System with GC, TQMS, Autosampler, and GCMS Insight Software |
Overview
The Shimadzu GCMS-TQ8050 is a high-performance triple quadrupole gas chromatography–mass spectrometry (GC-MS/MS) system engineered for ultra-trace quantitative analysis in regulated and research-intensive environments. Utilizing tandem mass spectrometry in multiple reaction monitoring (MRM) mode, the instrument delivers exceptional selectivity, sensitivity, and reproducibility by isolating precursor ions in Q1, fragmenting them in the collision cell (q2), and detecting specific product ions in Q3. This architecture enables robust discrimination against chemical noise and matrix interferences—critical for complex sample matrices encountered in food safety testing, environmental monitoring, clinical metabolomics, and impurity profiling in pharmaceutical development. Designed and manufactured in Kyoto, Japan, the GCMS-TQ8050 incorporates Shimadzu’s proprietary ion optics, high-efficiency electron ionization (EI) source, and a newly developed high-gain detector capable of stable signal acquisition at sub-attogram (ag) levels—achieving instrument detection limits (IDLs) in the low-ag range for key analytes under optimized conditions.
Key Features
- Triple quadrupole mass analyzer with independent RF/DC voltage control for Q1 and Q3, enabling precise mass filtering and high transmission efficiency
- Advanced ion source featuring dual-mode EI/CI capability, temperature-stabilized filament, and optimized ion optics for enhanced sensitivity and long-term stability
- High-vacuum system with dual-stage turbomolecular pumps (≥300 L/s total pumping speed) ensuring rapid pump-down and sustained operational vacuum (<5 × 10⁻⁷ Pa)
- Integrated capillary flow technology with split/splitless injection and programmable temperature vaporizing (PTV) inlet compatibility
- Robust GC oven with ±0.01 °C temperature stability and ramp rates up to 120 °C/min for high-resolution separation of volatile and semi-volatile compounds
- Pre-owned unit fully refurbished to factory specifications: includes comprehensive performance verification (PV), leak testing, detector calibration, and full diagnostic report
Sample Compatibility & Compliance
The GCMS-TQ8050 supports a broad range of sample types—including aqueous extracts, organic solvent extracts, headspace volatiles, and derivatized metabolites—compatible with standard EPA, ISO, ASTM, and AOAC methods. Its analytical performance meets or exceeds requirements for regulatory workflows under FDA 21 CFR Part 11, EU Annex 11, and GLP/GMP-compliant laboratories when paired with audit-trail-enabled software configurations. The system is routinely validated for applications such as dioxin/furan congener analysis (EPA Method 1613B), pesticide residue quantification (EN 15662), persistent organic pollutant (POP) screening (Stockholm Convention), and endogenous metabolite profiling (e.g., steroids, eicosanoids, bile acids). All hardware components comply with IEC 61010-1 safety standards and electromagnetic compatibility (EMC) directives.
Software & Data Management
GCMS Insight software provides an integrated platform for method development, acquisition, processing, and reporting—designed specifically for triple quadrupole workflows. It features intelligent MRM method building with auto-optimization of collision energies and dwell times, batch processing with customizable report templates, and built-in spectral libraries (NIST, Wiley, Shimadzu Custom Libraries). Data files are stored in vendor-neutral .qgd format with embedded metadata, supporting long-term archival and reprocessing. For regulated environments, optional GCMS Insight Secure Edition adds user access controls, electronic signatures, audit trail logging, and 21 CFR Part 11 compliance modules—including secure data archiving and change history tracking.
Applications
- Quantitative residue analysis of pesticides, veterinary drugs, and mycotoxins in food and feed matrices
- Trace-level detection of dioxins, PCBs, PBDEs, and other POPs in soil, sediment, air particulates, and biota
- Targeted metabolomics and biomarker validation in plasma, urine, and tissue homogenates
- Impurity identification and quantification in active pharmaceutical ingredients (APIs) per ICH Q3 guidelines
- Forensic toxicology screening for controlled substances and novel psychoactive substances (NPS)
- Environmental fate studies including degradation product identification and kinetic modeling
FAQ
Is this a factory-refurbished unit or a dealer-refurbished system?
This GCMS-TQ8050 is a certified pre-owned system refurbished by an authorized Shimadzu service partner to original equipment manufacturer (OEM) specifications, including full mechanical inspection, detector recalibration, vacuum integrity verification, and comprehensive performance qualification (PQ) testing.
What documentation accompanies the instrument?
A complete set is provided: Certificate of Conformance, PQ report with retention time, peak area RSD, S/N, and IDL data; maintenance log; software license certificate; and OEM operation manuals in English.
Does the system include an autosampler?
Yes—the configuration includes a Shimadzu AOC-20i or AOC-6000 Plus autosampler with 108-position vial capacity and programmable injection parameters.
Can the system be configured for compliance with 21 CFR Part 11?
Yes—when deployed with GCMS Insight Secure Edition and validated on-site, it supports electronic records, role-based access control, and full audit trail functionality required for regulated laboratories.
What is the warranty coverage for this pre-owned system?
The unit carries a standard 12-month parts-and-labor warranty from date of commissioning, extendable to 24 months with optional service agreement.