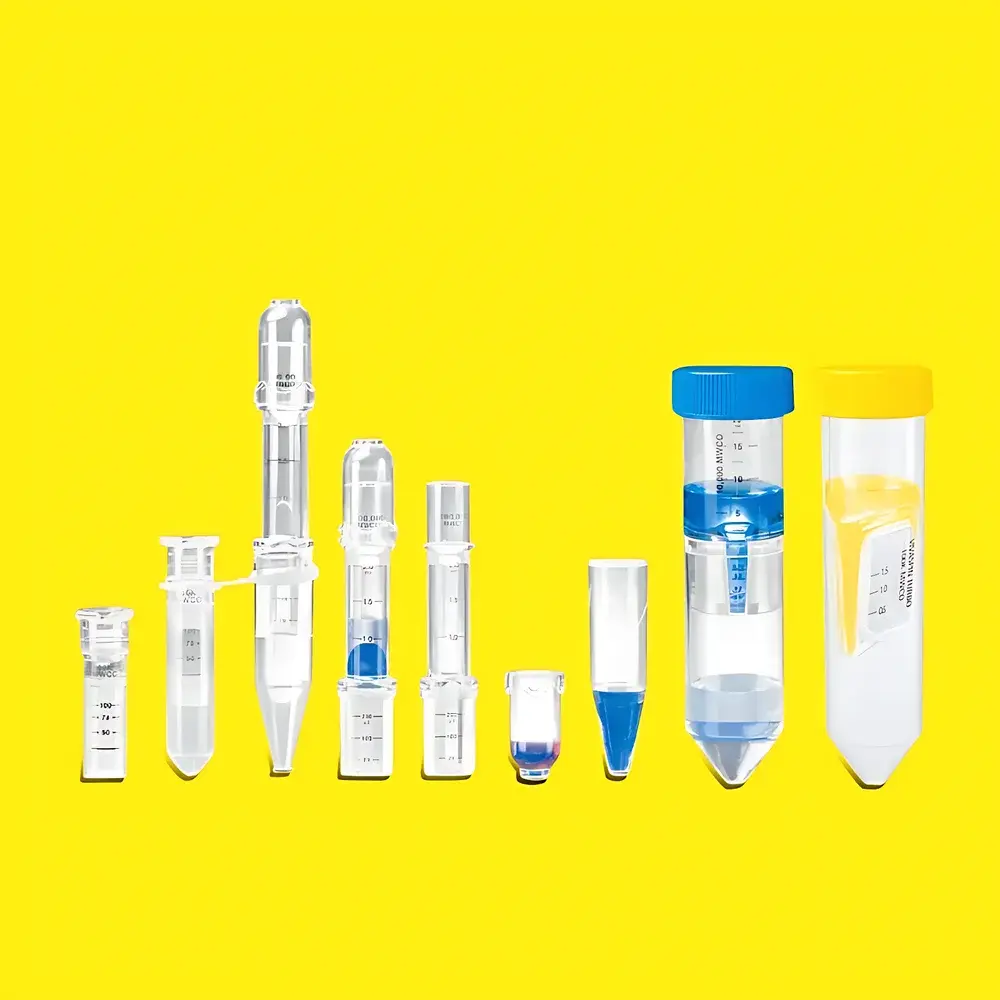
Sartorius Vivaspin Ultrafiltration Concentrator Series
| Brand | Sartorius |
|---|---|
| Origin | Germany |
| Model | Vivaspin 500 / 2 / 6 / 15R / 20 / Turbo |
| Membrane Material | Polyethersulfone (PES), Hydrosart®, Cellulose Triacetate (CTA) |
| MWCO Range | 2 kDa – 0.2 µm |
| Sample Volume Capacity | 100 µL – 20 mL |
| Sterility | Non-sterile (unless specified) |
| Compliance | Designed for GLP/GMP-aligned workflows |
Overview
The Sartorius Vivaspin Ultrafiltration Concentrator Series is a family of single-use, centrifugal ultrafiltration devices engineered for precise biomolecular concentration, buffer exchange, desalting, and size-based separation in life science research and bioprocessing applications. Based on tangential-flow-optimized vertical membrane geometry, Vivaspin units operate via centrifugal force-driven crossflow filtration—minimizing concentration polarization and membrane fouling while maximizing flux stability and solute recovery. Unlike conventional dead-end ultrafilters, the vertically oriented PES, Hydrosart®, or CTA membranes ensure uniform flow distribution across the entire active surface area, enabling reproducible performance across sample volumes ranging from 100 µL (Vivaspin 500) to 20 mL (Vivaspin 20). Each device integrates a defined retention dead volume—eliminating the need for post-concentration tube inversion and mitigating sample loss, adsorption, or degradation during recovery.
Key Features
- Vertical Membrane Architecture: Enhances convective mass transfer and reduces boundary layer formation, delivering up to 3× higher initial flux versus radial-flow designs.
- Precision Retention Dead Volume: Engineered reservoir at the bottom of the collection tube ensures complete, pipette-accessible recovery without centrifuge reversal—critical for low-abundance analytes and precious samples.
- Low-Binding Polymer Chemistries: PES membranes exhibit minimal non-specific protein adsorption (<0.5 µg IgG/cm²); Hydrosart® offers enhanced hydrophilicity for challenging samples (e.g., hydrophobic peptides, membrane proteins); CTA provides high chemical compatibility for organic solvent-containing buffers.
- Scalable MWCO Selection: Broad molecular weight cut-off range—from 2 kDa (for peptide enrichment and small-molecule removal) to 0.2 µm (for microorganism or virus particle retention)—supports diverse downstream requirements including NMR, crystallization, SEC-MALS, and ELISA preparation.
- Vivaspin Turbo Variant: Features an angled dead-volume collector and optimized rotor geometry, reducing typical concentration time by ≥40% versus standard Vivaspin formats while maintaining >95% recovery efficiency for IgG and BSA.
Sample Compatibility & Compliance
Vivaspin concentrators are validated for use with mammalian cell lysates, serum, ascites fluid, column eluates, fermentation broths, and viral suspensions. They comply with ISO 10993–5 (cytotoxicity) and EU Regulation (EC) No 1935/2004 for food-contact materials when used with appropriate membrane types. Devices are manufactured in Sartorius’ ISO 13485–certified facilities in Germany and undergo rigorous lot-release testing for integrity (bubble point), MWCO verification (dextran rejection assay), and endotoxin levels (<0.25 EU/mL for sterile variants). For regulated environments, traceability is supported via batch-specific CoA documentation, and electronic records align with FDA 21 CFR Part 11 requirements when integrated with compliant LIMS or ELN platforms.
Software & Data Management
As consumables, Vivaspin units do not incorporate embedded electronics or firmware. However, their standardized dimensions, centrifugation parameters (RCF and time), and compatibility with common benchtop centrifuges (e.g., Eppendorf 5424/5430, Thermo Scientific Sorvall Legend Micro 17/21) enable seamless integration into automated liquid handling workflows. Sartorius provides downloadable centrifugation protocols—including RCF/time matrices per model/MWCO—and digital product identifiers (GTIN/EAN) for inventory tracking in ERP systems. Sterile, individually wrapped units support audit-ready documentation for GLP studies, with full traceability to manufacturing lot and expiration date.
Applications
- Concentration of dilute antibody, antigen, or enzyme solutions post-affinity chromatography
- Buffer exchange into crystallization-compatible conditions (e.g., Tris→HEPES, removal of imidazole)
- Desalting prior to MALDI-TOF or LC-MS analysis
- Preparative purification of extracellular vesicles (EVs) and lentiviral vectors using 100 kDa or 0.2 µm MWCO variants
- Rapid cleanup of PCR reaction mixtures (Vivacon 500-PCR format)
- Removal of aggregates or degraded fragments from recombinant protein preparations
- Concentration of low-titer viral stocks for infectivity assays or cryo-EM grid preparation
FAQ
What centrifugation parameters are recommended for Vivaspin 2 with 10 kDa MWCO?
For optimal recovery and minimal shear stress, use 4,000 × g for 10–20 min at 4 °C. Maximum RCF should not exceed 5,000 × g to prevent membrane deformation.
Can Vivaspin devices be reused?
No. Vivaspin concentrators are designed as single-use, disposable devices to ensure consistency, eliminate carryover risk, and comply with regulatory expectations for sample integrity in clinical or GMP-relevant workflows.
How is MWCO verified for each lot?
Each production lot undergoes dextran-based rejection testing per ASTM D5257, measuring % retention of defined dextran standards (e.g., 10 kDa dextran for 10 kDa MWCO units), with acceptance criteria set at ≥90% retention.
Are sterile versions available?
Yes. Sterile, pyrogen-free Vivaspin units are gamma-irradiated and supplied in individually sealed, ISO Class 5 cleanroom-packaged boxes—certified to ISO 11137 and accompanied by full sterility assurance documentation.
Which membrane type is recommended for acidic or high-salt buffers?
Hydrosart® membranes demonstrate superior stability in pH 2–12 and tolerate up to 4 M NaCl, making them preferred for harsh purification conditions where PES may show reduced flux or increased protein binding.