Elite Elitapex Chromatography Data System
| Brand | Elite |
|---|---|
| Origin | Liaoning, China |
| Manufacturer Type | Original Equipment Manufacturer (OEM) |
| Regional Classification | Domestic (PRC) |
| Model | Elitapex |
| Price Range | USD 1,400 – 7,000 (FOB) |
| Software Type | Chromatography Data System (CDS) |
Overview
The Elite Elitapex Chromatography Data System (CDS) is a purpose-built, PC-based software platform engineered for acquisition, processing, analysis, and archival of chromatographic data generated by high-performance liquid chromatography (HPLC), ultra-high-performance liquid chromatography (UHPLC), and gas chromatography (GC) systems. Designed and developed in-house by Elite Analytical Instruments Co., Ltd., the Elitapex CDS implements a modular architecture that supports real-time bidirectional communication with Elite’s proprietary chromatographic hardware—including pumps, autosamplers, column ovens, detectors (UV-Vis, fluorescence, conductivity), and fraction collectors—via optimized low-latency instrument control protocols. Its core function is to serve as a centralized, deterministic interface between analytical instruments and laboratory personnel, enabling precise method execution, traceable data handling, and compliant reporting in regulated environments.
Key Features
- Real-time instrument control and status monitoring: Provides continuous, millisecond-level polling of module states—including flow rate, pressure, temperature, detector signal, valve position, and injection cycle status—displayed via intuitive graphical indicators and dynamic parameter gauges.
- Modular protocol stack: Implements a refined, proprietary communication layer compliant with RS-232, USB virtual COM, and Ethernet TCP/IP interfaces; supports both synchronous command-response and asynchronous event-driven messaging for enhanced system responsiveness and reduced latency.
- Streamlined user interface: Features a task-oriented layout with context-sensitive toolbars, drag-and-drop method building, one-click sequence setup, and customizable dashboard views—reducing operator training time and minimizing procedural errors.
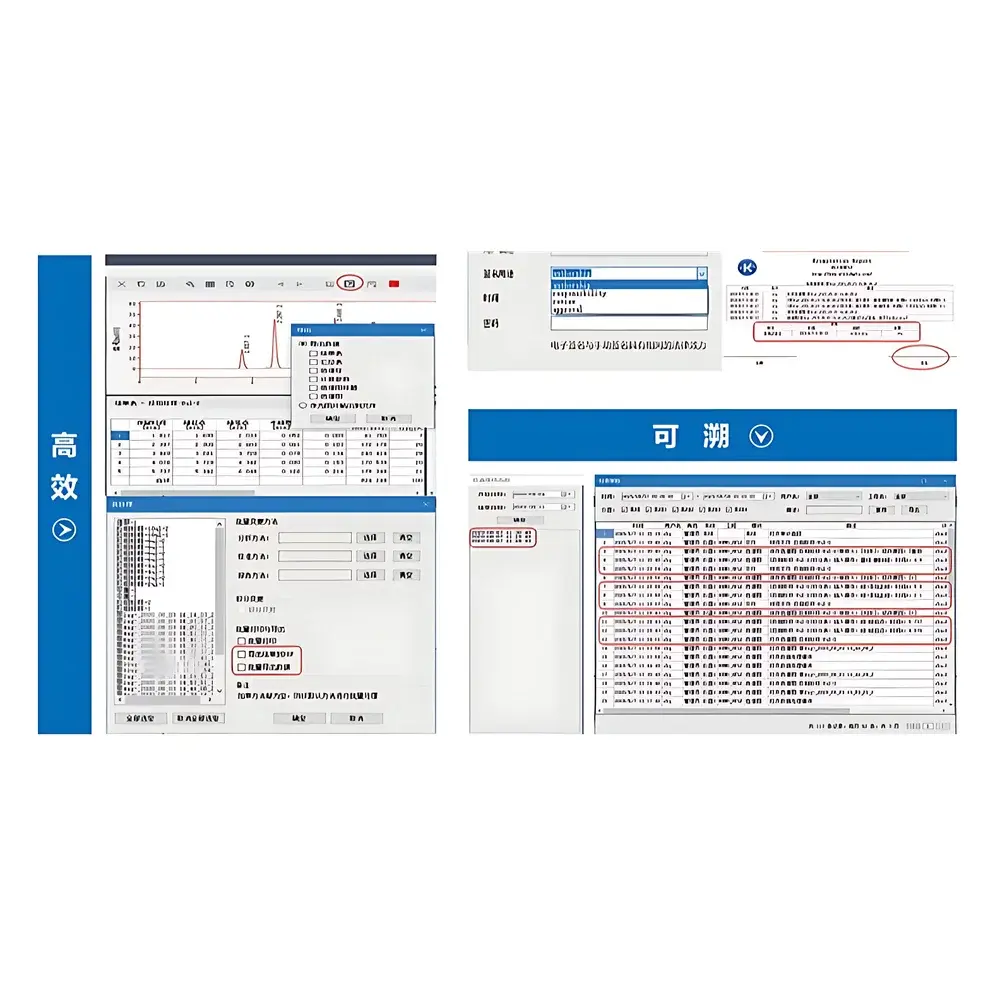
- Method validation support: Includes built-in tools for system suitability testing (SST), peak purity assessment, baseline noise/drift calculation, and resolution/symmetry evaluation per ICH Q2(R2) guidelines.
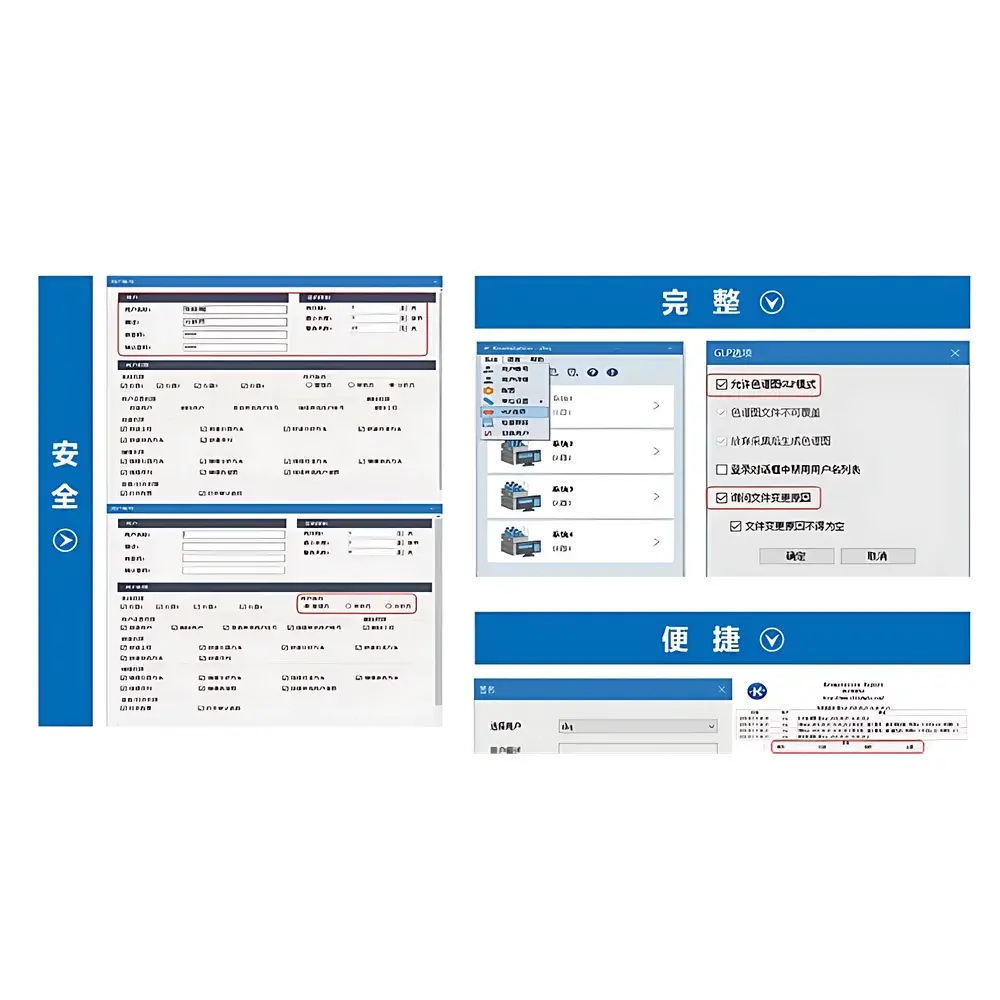
- Secure data handling: Enforces role-based access control (RBAC), electronic signature capability, and audit trail generation for all critical actions—including method modification, data reprocessing, and report export—aligned with FDA 21 CFR Part 11 and EU Annex 11 requirements.
Sample Compatibility & Compliance
The Elitapex CDS natively supports chromatographic data files from Elite HPLC systems (e.g., P230II, P3000, L-2000 series) and is interoperable with standard chromatographic file formats including .ELD, .CDF, and .RAW (via optional converters). It accommodates diverse sample types—from small-molecule pharmaceuticals and environmental contaminants to natural product extracts and polymer additives—by supporting multi-channel detection, gradient elution programming, and retention time locking. The software adheres to ISO/IEC 17025:2017 principles for data integrity and is validated for use in GLP-compliant laboratories. Full traceability is maintained through immutable audit logs recording user ID, timestamp, action type, and pre-/post-change values for every data manipulation event.
Software & Data Management
Elitapex employs a relational database backend (SQLite or Microsoft SQL Server options) for structured storage of methods, sequences, raw data, processed results, and audit trails. All chromatograms and quantitative reports are stored with embedded metadata—including instrument configuration, calibration history, and analyst credentials—to ensure full data lineage. Export functions support PDF/A-1b (for long-term archiving), CSV (for statistical analysis), and XML (for LIMS integration). Optional modules include electronic lab notebook (ELN) synchronization, automated QC pass/fail flagging against predefined acceptance criteria, and scheduled backup to network-attached storage (NAS) or cloud repositories with AES-256 encryption.
Applications
The Elitapex CDS is routinely deployed in quality control (QC) laboratories for routine release testing of active pharmaceutical ingredients (APIs) and finished dosage forms; in R&D settings for method development and stability-indicating assay optimization; and in environmental and food safety labs for pesticide residue screening, mycotoxin quantification, and adulterant detection. Its deterministic control logic ensures reproducible gradient formation and precise dwell volume compensation—critical for method transfer between Elite systems and third-party instruments. Regulatory submissions utilizing Elitapex-generated data have been accepted by NMPA, CFDA, and regional health authorities under documented validation protocols.
FAQ
Is Elitapex compatible with non-Elite chromatographic hardware?
Yes—through optional driver packages and generic SCPI-compliant interfaces, Elitapex can integrate with select third-party HPLC/GC systems; however, full feature parity (e.g., advanced detector diagnostics or fraction collector synchronization) is guaranteed only with Elite-branded modules.
Does Elitapex support 21 CFR Part 11 compliance out-of-the-box?
The system includes configurable electronic signature workflows, audit trail logging, and user authentication mechanisms; formal Part 11 compliance requires site-specific validation documentation and administrative policy implementation by the end-user laboratory.
What operating systems are supported?
Windows 10 (64-bit) and Windows 11 (64-bit) are fully supported; Windows Server 2016/2019/2022 are certified for network deployment in multi-user environments.
Can raw data be exported without proprietary software?
Yes—raw chromatographic data is stored in open binary formats with documented structure; ASCII exports (time-intensity tables) and standardized .CDF files are available without requiring Elitapex runtime.
Is remote monitoring or cloud-based access available?
Local network remote desktop access is supported; cloud-hosted deployment is not offered directly by Elite but may be implemented via customer-managed virtual desktop infrastructure (VDI) with appropriate security controls.