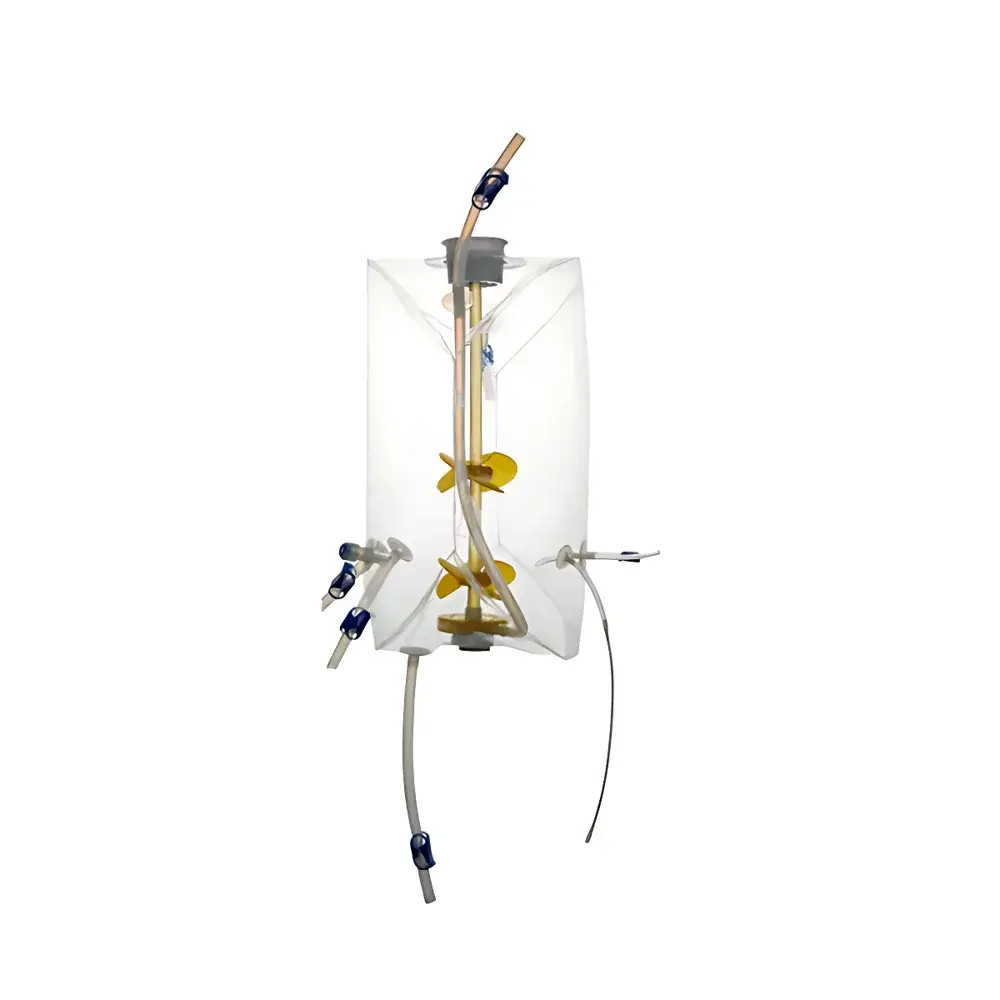
AbioBag Single-Use Bioreactor Bag
| Brand | Applitech |
|---|---|
| Origin | Guangdong, China |
| Manufacturer Type | Direct Manufacturer |
| Region of Origin | Domestic (China) |
| Model | AbioBag |
| Pricing | Upon Request |
Overview
The AbioBag Single-Use Bioreactor Bag is an engineered fluid containment system designed for scalable, aseptic, and process-integrated biomanufacturing in upstream bioprocessing. Developed by Applitech Biotechnology Co., Ltd., the AbioBag operates as a sterile, pre-validated disposable vessel liner compatible with standard stainless-steel or single-use bioreactor support systems. It leverages Couette-type mixing dynamics via marine impeller agitation and multi-point gas sparging to ensure homogeneous cell culture environments across volumes ranging from 50 L to 2000 L. The bag’s geometry—maintaining a fixed height-to-diameter ratio of 1.8—is optimized for consistent mass transfer (kLa), shear control, and scalability from lab-scale development to clinical and commercial manufacturing. Its design adheres to core principles of single-use technology: elimination of cleaning-in-place (CIP) and sterilization-in-place (SIP) validation burdens, reduction of cross-contamination risk, and accelerated campaign turnaround.
Key Features
- Engineered geometric consistency: Fixed aspect ratio (H/D = 1.8) ensures reproducible hydrodynamic behavior and facilitates scale-up from benchtop to production bioreactors.
- Agitation system compatibility: Optimized for top-driven marine impellers with dual blades, supporting controlled rotational speeds up to 60 rpm under defined gas flow conditions.
- Multi-path gas delivery: Three independent sparging routes—1 surface-level and 2 bottom-mounted—enable flexible oxygenation and CO2 stripping strategies; bubble size distribution includes macro-sparging (0.8 mm) and micro-sparging (150 µm).
- Advanced multilayer film construction: Primary contact layer composed of ethylene-vinyl acetate (EVA), laminated with polyethylene (PE)-based barrier layers (Renolit BF1400 series, grade RENOLIT 9101); free of leachable additives, antioxidants, or plasticizers.
- Enhanced mechanical compliance: Film formulation provides superior drape and conformability to rigid support vessels, minimizing air pockets and improving thermal and mixing uniformity.
- Rigorous extractables & leachables (E&L) profiling: Conducted per BPOG (BioPhorum Operations Group) guidance and USP , , and Class VI protocols to support regulatory filings for therapeutic protein, mAb, and viral vector processes.
Sample Compatibility & Compliance
The AbioBag demonstrates broad compatibility with mammalian (e.g., CHO, HEK293), microbial (e.g., E. coli, Pichia), and stem cell culture media formulations. All materials contacting process fluids are certified compliant with ISO 10993–related biological safety standards, including cytotoxicity (USP ), systemic toxicity (USP Class VI), and hemocompatibility assessments. Manufacturing occurs under a certified ISO 13485:2016 quality management system, with environmental controls aligned to ISO 14001. Each batch undergoes full release testing per internal SOPs referencing ISO 11137 (radiation sterilization dose verification), ASTM F2096 (bubble leak test), and GB/T 14233.2–2015 (bacterial endotoxin limits). Final product integrity is confirmed via pressure hold, dye ingress, and mechanical strength validation (seal peel, clamp retention, burst pressure).
Software & Data Management
While the AbioBag itself is a passive consumable, its integration into automated bioreactor platforms (e.g., Applitech’s AbioReact series or third-party controllers such as Sartorius BIOSTAT® or Thermo Fisher HyPerforma™) enables full digital traceability. Batch records—including irradiation dose logs, E&L reports, sterility certificates, and physical inspection data—are archived in secure, audit-ready formats compliant with FDA 21 CFR Part 11 requirements. Extractables datasets are structured according to PQRI and BPOG templates to support Quality-by-Design (QbD) documentation and regulatory submissions (IND/BLA/MAA). All validation documentation is provided in English and formatted for inclusion in Drug Master Files (DMFs).
Applications
- Monoclonal antibody (mAb) and recombinant protein production in suspension CHO cultures
- Viral vector manufacturing (AAV, lentivirus) requiring low-shear, high-oxygen-transfer environments
- Cell therapy expansion workflows under cGMP-compliant clinical manufacturing settings
- Process development and tech transfer studies where rapid vessel changeover and material comparability are critical
- Scale-down modeling for bioreactor qualification and platform process characterization
FAQ
Is the AbioBag suitable for GMP manufacturing?
Yes—each production lot is manufactured under ISO 13485-certified quality systems and released with full CoA, CoC, and biocompatibility documentation meeting ICH Q5A(R2), Q5B, and Q5D expectations.
What regulatory support documentation is available?
Applicability letters, DMF cross-references, USP-compliant E&L reports, ISO 11137 sterilization validation summaries, and full biocompatibility dossiers (USP , , ISO 10993-5/-10/-11) are provided upon request.
Can the AbioBag be used with non-Applitech bioreactor hardware?
Yes—the bags are dimensionally standardized for compatibility with common single-use and hybrid bioreactor support systems; mechanical interface specifications (port locations, clamp diameters, agitator shaft clearance) are supplied in technical drawings.
Are alternative film options available?
Yes—while Renolit 9101 is the default high-performance film, domestic PE-based alternatives are offered for cost-sensitive or region-specific supply chain requirements; full comparative E&L and mechanical data are available upon request.
How is sterility assurance validated?
Sterilization is performed via gamma irradiation at a minimum validated dose of 25 kGy, verified per ISO 11137–2; sterility testing follows USP and is conducted on every production lot using membrane filtration and incubation per pharmacopeial methods.