Life Science Instruments
Filter
Showing 421–450 of 2783 results
| Brand | AUCMA |
|---|---|
| Origin | Shandong, China |
| Model | YC-200 |
| Refrigeration Temperature Range | 2–8 °C |
| Effective Capacity | 200 L |
| Internal Dimensions (W×D×H) | 490 × 400 × 1016 mm |
| External Dimensions (W×D×H) | 560 × 578 × 1522 mm |
| Voltage/Frequency | ~220 V / 50 Hz |
| Net Weight | 54 kg |
| Gross Weight | 61 kg |
| Control Accuracy | ±0.1 °C |
| Cooling System | Forced-air convection with high-efficiency compressor and EBM fan motor |
| Safety Alarms | High/low temperature, door open, power failure, sensor fault |
| Door Type | Single transparent double-glazed coated glass door with anti-condensation heating strip |
| Shelving | 3 adjustable wire shelves + optional baskets |
| Compliance | Designed for GLP-compliant storage per ISO 13485 and WHO Technical Report Series No. 961 (2011), Annex 9 |
| Brand | AUCMA |
|---|---|
| Origin | Shanghai, China |
| Model | YC-80 |
| Capacity | 80 L |
| Temperature Range | 2–8°C |
| Internal Dimensions | 360 × 400 × 680 mm |
| External Dimensions | 472 × 500 × 870 mm |
| Power Consumption | 150 W |
| Rated Voltage/Frequency | 220 V / 50 Hz |
| Net Weight | 30.5 kg |
| Gross Weight | 34 kg |
| Shelving | 3 adjustable wire shelves |
| Compressor & Fan | International brand compressor + EBM high-efficiency DC fan motor |
| Temperature Display Resolution | 0.1°C |
| Alarm System | Dual-mode (audible + visual), remote alarm output, battery-backed temperature logging during power outage |
| Brand | AUCMA |
|---|---|
| Model | YCD-265 |
| Temperature Range (Refrigeration Compartment) | 2–8 °C |
| Temperature Range (Freezing Compartment) | –15 to –26 °C |
| Net Capacity | 265 L |
| Internal Dimensions (W×D×H) | 460×480×720 mm (refrigeration section) |
| External Dimensions (W×D×H) | 575×641×1687 mm |
| Power Supply | 220 V / 50 Hz |
| Control System | Microprocessor-based dual-zone digital display |
| Cooling Method | Forced-air (refrigeration), static cooling (freezing) |
| Door Lock | Mechanical safety lock |
| Insulation | CFC-free polyurethane foam |
| Refrigerant | R600a (isobutane) |
| Compliance | Designed per GB/T 20154–2014 (Chinese national standard for medical refrigerators), suitable for GLP-aligned storage environments |
| Brand | Auniontech |
|---|---|
| Origin | Shanghai, China |
| Manufacturer Type | Authorized Distributor |
| Product Category | Domestic (PRC-Made) |
| Model | OCT-3D Imaging System |
| Pricing | Available Upon Request |
| Brand | Auniontech |
|---|---|
| Origin | Shanghai, China |
| Manufacturer Type | Authorized Distributor |
| Product Category | Domestic |
| Model | SUPERNOVA-3000 Biological In Vivo Imaging System |
| Form Factor | Miniaturized Three-Photon Probe |
| Probe Weight | 2.2 g |
| Excitation NA / Collection NA | 0.55 / 0.65 |
| Immersion Medium | Water or Silicone Oil |
| Field of View | 400 × 400 µm² |
| Working Distance | 1.75 mm |
| Probe Diameter | 3.4 mm |
| Fluorescence Detection | GaAsP PMT (300–720 nm) |
| Green Channel | 520 ± 25 nm |
| Red Channel | 625 ± 25 nm |
| Controller | ≥120 MS/s Sampling Rate |
| Fiber-Coupled AOM | <250 ns Response Time |
| XYZ Stage | Bidirectional Repeatability ≤1 µm |
| Excitation Wavelength Range | 490–550 nm |
| Widefield Camera | 1920 × 1200 px, 40 Hz Full-Frame |
| Laser Options | 1300 nm / 1700 nm Femtosecond Oscillators |
| Software Suite | SUPERGIN (Acquisition), SUPERANALY (Analysis) |
| System Footprint | 595 × 400 × 668 mm³ |
| Operating Environment | 20–30 °C, RH <60% |
| Brand | Aure |
|---|---|
| Model | AF100 |
| Origin | Jiangsu, China |
| Instrument Type | Automated Time-Lapse Microscopy System for Long-Term Culture Monitoring |
| Optical Resolution | 5 MP Monochrome CMOS Sensor |
| Compatible Well Plates | 6–384-well |
| Objective Lenses Supported | 4×, 10×, 20× (Standard, UP, and LWD) |
| Imaging Mode | Brightfield Only |
| Maximum Scan Speed | Full 96-well Plate in ≤4 min |
| Environmental Integration | Designed for In-CO₂ Incubator Deployment (Temperature/Humidity/CO₂ Stable Operation) |
| Software | Integrated Time-Lapse Acquisition, Image Stitching, Video Synthesis, and Multi-Point Retrospective Analysis Engine |
| Brand | Aure |
|---|---|
| Model | BP4000 |
| Origin | Beijing, China |
| Temperature Control Range | 2–8 °C |
| Category | Cell Preparation System for 3D Culture |
| Core Technology | CMOS-MEMS Microfluidic Digital Droplet Chip |
| Operating Principle | Precision Droplet-Based Cell Patterning on ECM Substrates |
| Brand | Aure |
|---|---|
| Origin | Shanghai, China |
| Manufacturer Type | Authorized Distributor |
| Country of Origin | China |
| Model | SCP4000 |
| Pricing | Available Upon Request |
| Single-Cell Recognition Accuracy | ≥98% |
| Monoclonal Guarantee Rate | >99.5% |
| Imaging Modality | Brightfield |
| Objective NA | 0.3 |
| Plate Throughput | ≤2 min/96-well plate, ≤8 min/384-well plate |
| Nozzle Count | 640 independently addressable nozzles |
| Nozzle Diameter | 30 µm |
| Droplet Volume | 24 pL |
| Compatible Substrates | 6/12/24/48/96/384/1536-well plates, 8-tube strips, glass slides, custom arrays |
| Compatible Buffers | Serum-containing media, PBS, H₂O, ethanol |
| Cell Viability Post-Printing | >90% |
| Operating Temperature | 15–30 °C |
| Relative Humidity | 20–80% RH |
| Core Chip | B150 CMOS-MEMS Integrated Microfluidic Cartridge |
| Recognition Algorithm | Convolutional Neural Network (CNN) trained on cell diameter, circularity, and morphological features |
| Sample Compatibility | CHO, HEK, tumor cell lines, iPSCs, primary cells, nuclei, protoplasts |
| Brand | Aure |
|---|---|
| Origin | Beijing, China |
| Model | SCP4000 |
| Sample Throughput | 16 samples per run |
| Chip Configuration | 640 parallel nozzles per disposable B150 print chip |
| Compliance | Designed for GLP-compliant cell line development workflows |
| Software | Embedded AI-powered image recognition engine for real-time single-cell identification and deposition targeting |
| Brand | Aurora |
|---|---|
| Origin | USA |
| Model | 1300A |
| Force Range | Up to 10 N |
| Temperature Control | Precision-regulated platform |
| Stimulation | Biphasic current/voltage neuromuscular stimulator (701C) |
| Software | Dynamic Muscle Control & Analysis (DMC/DMA) |
| Compliance | Designed for ISO/IEC 17025-aligned laboratory workflows, supports GLP-compliant audit trails and FDA 21 CFR Part 11–ready data export |
| Brand | Aurora Scientific |
|---|---|
| Origin | USA |
| Model | A1300 |
| Application Scope | Rodent (mouse/rat) skeletal muscle functional assessment |
| Core Sensor | Dual-Mode Muscle Lever (force + length transduction & actuation) |
| Temperature Control | Integrated thermostatic platform (±0.1 °C stability) |
| Stimulator | High-energy biphasic stimulator (0–200 V, 0.01–100 ms pulse width) |
| Data Acquisition | Up to 100 kHz sampling rate |
| Software Platform | DMC/DMA v6.x (Windows/Linux compatible) |
| Compliance | Designed for GLP-compliant workflows |
| Brand | Autobio |
|---|---|
| Origin | Henan, China |
| Manufacturer Type | Authorized Distributor |
| Country of Manufacture | China |
| Model | AutoChem B861 |
| Price Range | USD 150–15,000,000 (FOB) |
| Detection Principle | Post-Dispensing Spectrophotometry with 16-Wavelength Optical System |
| Sample Throughput | Up to 800 tests/hour (typical configuration) |
| HbA1c Capability | Whole Blood Direct Measurement |
| Mechanical Durability | >5,000,000 cycles validated under continuous-load fatigue testing |
| Key Components | Imported precision optical modules, high-accuracy peristaltic pumps, and temperature-stabilized reaction cuvettes |
| Brand | Autobio |
|---|---|
| Model | Autof MS |
| Origin | Henan, China |
| Laser Lifetime | >100 million shots |
| Mass Analyzer | Titanium Alloy Linear TOF (1.05 m) |
| Vacuum System | High-Throughput Turbo-Molecular Pump |
| Identification Speed | ≤0.1 s per sample (database search) |
| Microbial Database | 5,000+ species across 1,000+ genera, including filamentous fungi, anaerobic gut bacteria, and marine isolates |
| Nucleic Acid Detection Capacity | Up to 40-plex per well |
| Regulatory Certification | NMPA-approved calibration and control materials |
| Software Compliance | Audit trail, user access control, and electronic signature support per FDA 21 CFR Part 11 requirements |
| Brand | Autobio |
|---|---|
| Model | Autof ms2000 |
| Origin | Henan, China |
| Laser Source | Solid-state, ≥10⁹ shots lifetime, 1–1000 Hz tunable frequency |
| Ion Source Vacuum | ≤10⁻⁷ mbar |
| Ion Source Maintenance | Auto-cleaning cycle ≤10 min |
| Mass Analyzer | Titanium alloy flight tube with high thermal stability |
| Vacuum System | High-throughput turbomolecular pump |
| Signal Acquisition | High-precision FPGA-controlled real-time digitizer |
| Software Platform | AI-accelerated microbial identification engine (96 samples in <5 min) |
| Database Coverage | >1,000 genera, >5,000 species, >16,000 strains |
| Regulatory Status | CE-IVD compliant |
| Data Security | On-premise and cloud-deployable database options |
| Brand | Autobio |
|---|---|
| Origin | Henan, China |
| Manufacturer Type | Authorized Distributor |
| Country of Manufacture | China |
| Model | Autostreak S1800 |
| Price Range | USD 140,000 – 210,000 (FOB) |
| Instrument Type | Fully Automated |
| Sample Type | Liquid (including sputum, broth cultures, urine supernatants) |
| Throughput | 180 Petri Dishes per Hour |
| Liquefaction Time | ≤5 min (sputum) |
| Streaking Time | ≤60 sec per 3 plates |
| Biosafety Level | Integrated Class II Biological Safety Cabinet Architecture |
| Filtration | HEPA H14 (99.995% @ 0.3 µm) |
| Decontamination | Automated UV-C (254 nm) cycle with timer-based scheduling |
| Consumables | Disposable sterile tips and single-use inoculation brushes |
| LIS Integration | ASTM E1384 / HL7 v2.x compliant interface |
| Software Compliance | Audit trail, user access control, electronic signature support per FDA 21 CFR Part 11 requirements |
| Brand | Autobio |
|---|---|
| Origin | Henan, China |
| Manufacturer Type | Authorized Distributor |
| Product Category | Domestic |
| Model | iWO-960 |
| Price Range | USD 7,000–14,000 (FOB) |
| Instrument Type | Multifunctional Plate Washer |
| Wash Head Configuration | 12-channel |
| Rinse Head Design | Integrated Anti-Clogging Mesh Filter |
| Fluid Channels | One wash solution channel + one waste aspiration channel |
| Soak Time Range | 0–10 seconds (adjustable in 1-s increments) |
| Wash Cycles | Manual or programmable auto-mode (1–10 cycles) |
| Residual Volume per Well | ≤1 µL |
| Orbital Shaking Function | Yes, with acoustic-dampened motor and vibration-isolated platform |
| Waste Bottle Capacity | Standard 2 L, with full-level optical sensor and audible/visual alarm |
| Brand | Autobio |
|---|---|
| Model | LUMO |
| Detection Method | Chemiluminescence |
| Detector Type | Imported Photomultiplier Tube (PMT) |
| Luminescence Range | 10⁰–10⁹ RLU |
| Plate Format | 96-well and 48-well microplates |
| Read Time per Well | 0.1–10 s |
| Full 96-Well Plate Read Time | <32 s |
| Sensitivity | 10⁻² mol/plate |
| Precision (CV) | <2% |
| Inter-well Cross-Talk | <5×10⁻⁷ |
| Plate Shaking Modes | 3 programmable patterns (speed & duration adjustable) |
| Power Input | 220 V ± 22 V AC |
| Power Consumption | <32.4 VA |
| Dimensions (L×W×H) | 470 × 335 × 205 mm |
| Weight | 13 kg |
| Safety Compliance | CE-marked, 24 V DC internal supply, ISO 13485 certified |
| Brand | Autobio |
|---|---|
| Model | PHOMO |
| Origin | Henan, China |
| Instrument Type | Multifunctional Automated Microplate Reader |
| Assay Mode | Endpoint |
| Calibration Curve | Linear |
| Detector | Dual-LED Laser-Based Photodiode Array |
| Wavelength Range | 200–1000 nm |
| Absorbance Range | 0–4.5 OD |
| Absorbance Linearity | 0.000–2.000 Abs ±0.01% |
| Measurement Speed | 12 s (single wavelength), 20 s (dual wavelength), 6 s (kinetic single-wavelength) |
| Precision (CV) | <0.5% |
| Repeatability (CV) | ≤0.15% |
| Light Source | Dual-LED Cold Light Source (Lifetime ≥100,000 h) |
| Optical Architecture | RSE (Real-time Spectral Enhancement) Silicon Photodetector Platform |
| Scan Mode | Multi-point per well (up to 9 points) |
| Brand | Autobio |
|---|---|
| Model | Sikun1000 |
| Instrument Type | Second-Generation Sequencing Platform |
| Regulatory Status | CE-IVD & NMPA-Certified In Vitro Diagnostic Device |
| Form Factor | Benchtop Sequencer |
| Read Length | 50–300 bp |
| Throughput | 110M–330M Reads per Run |
| Data Output | 5–100 Gb per Run |
| Run Time | 3.5–24 h |
| Base Call Accuracy (Q30) | >85%–90% |
| Optical Detection Method | Fluorescence-Based Sequencing-by-Synthesis on Solid-Phase Amplified Flow Cell |
| Compliance | ISO 13485, IEC 62304, FDA 21 CFR Part 820, GLP/GMP-Aligned Data Integrity Framework |
| Brand | Autobio |
|---|---|
| Model | Sikun2000 |
| Instrument Type | Second-Generation Sequencing Platform |
| Regulatory Status | CE-IVDR and NMPA-Certified In Vitro Diagnostic Device |
| Form Factor | Benchtop Sequencer |
| Read Length Range | 50–300 bp |
| Data Quality | Q30 ≥ 85%–90% |
| Accuracy | >99.9% Base Call Accuracy (per-cycle consensus) |
| Run Time | As fast as 3.2 hours (dependent on read length and chip configuration) |
| Throughput | Mid-Throughput Chip: 220 million reads (11–66 Gb) |
| High-Throughput Chip | 660 million reads (33–200 Gb) |
| Chemistry | Surface-Based Bridge Amplification with Reversible Dye-Terminator Fluorescence Detection |
| Compliance | Designed and validated per ISO 13485, IEC 62304, and aligned with FDA 21 CFR Part 820 and EU IVDR Annex I essential requirements |
| Brand | Autobio |
|---|---|
| Model | Sikun500 |
| Instrument Type | Second-Generation Sequencing Platform |
| Medical Device Classification | Class III (China NMPA) |
| Form Factor | Benchtop Sequencer |
| Read Length | 50–300 bp |
| Q30 Score | >85–90% |
| Sequencing Accuracy | >99.9% |
| Run Time | 3.5–24 hours |
| Throughput | 60M–170M Reads per Run |
| Data Output | 5–50 Gb per Run |
| Origin | Henan, China |
| Regulatory Status | CE-IVD compliant (pending), NMPA registered |
| Brand | Autobio |
|---|---|
| Model | AutoMolec 3000 |
| Origin | Henan, China |
| Sample Throughput | 3000 tests/day |
| Heating Rate | 4 °C/s |
| Temperature Accuracy | ±0.1 °C |
| Well-to-Well Temperature Uniformity | ±0.5 °C |
| Detection Limit | <100 copies/mL |
| Cycle Time per Sample | ~100 min |
| Compliance | ISO 13485, CE-IVD, NMPA Registration No. 20223400123 |
| Brand | D.C. Labware |
|---|---|
| Origin | Guangdong, China |
| Model | Autopet-CP1 |
| Instrument Type | Single-needle colony picking workstation |
| Compatible Microorganisms | *Escherichia coli*, yeast, pathogenic bacteria, anaerobes, phage plaques, microalgae |
| Inoculation Mode | Solid-to-liquid transfer |
| Picking Throughput | 200–300 colonies/hour |
| Picking Accuracy | >99.5% |
| Positional Precision | ≤0.2 mm |
| Target Plate Compatibility | 24-, 48-, 96-, and 384-well plates |
| Source Plate Compatibility | Petri dishes with diameters of 6–9 cm |
| Dimensions (L×W×H) | 85 × 62 × 72 cm |
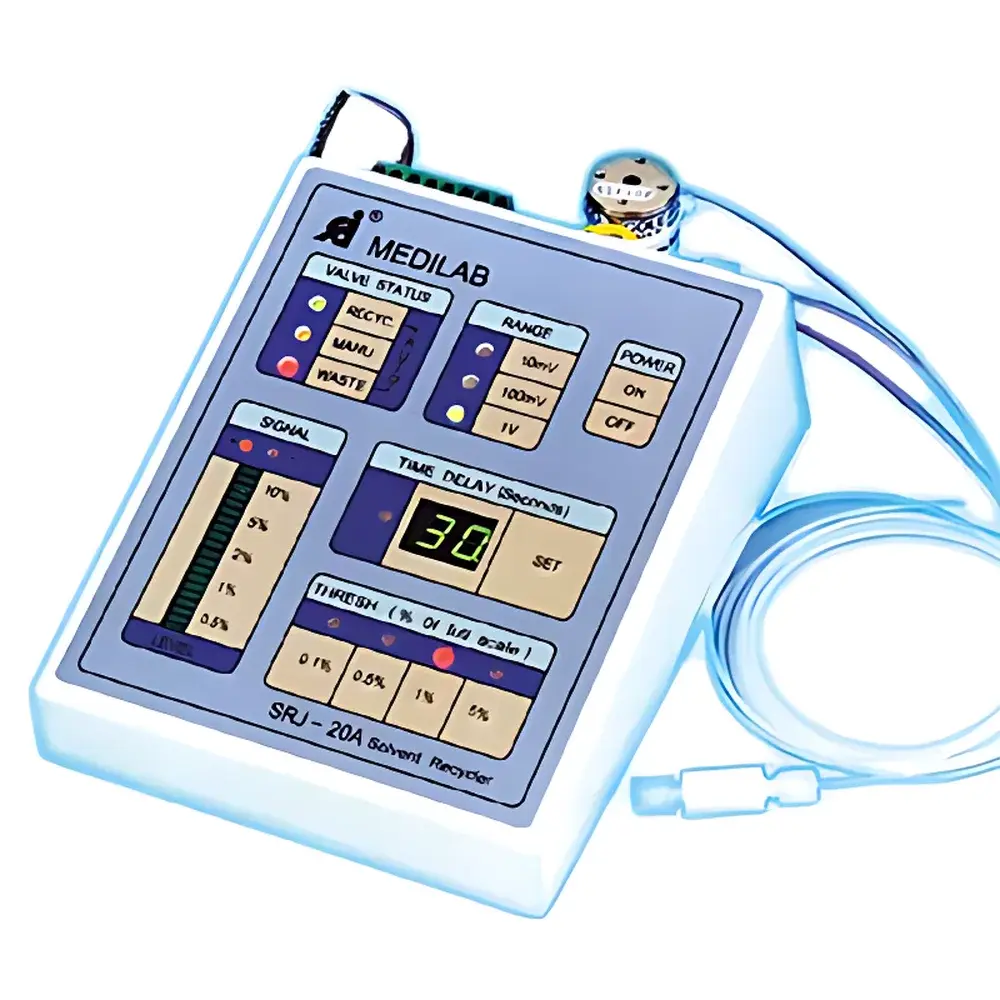
| Brand | Autoscience |
|---|---|
| Origin | Tianjin, China |
| Manufacturer Type | Authorized Distributor |
| Country of Origin | China |
| Model | SRJ-20A |
| Input Signal Range | 10 mV / 100 mV / 1 V |
| Valve Delay Time | 10–90 s |
| Maximum Flow Rate | 50 mL/min (through 1/16" tubing) |
| Display | Dual-color (red/green) LCD |
| Zero Adjustment | Auto-zero tracking baseline drift |
| Power Supply | 13.5 V / 500 mA |
| Dimensions (L×W×H) | 14.2 × 18.5 × 6.28 cm |
| Weight | 1.0 kg |
| Brand | Autosense |
|---|---|
| Membrane Materials | Mixed Cellulose Ester (MCE) & Polyvinylidene Fluoride (PVDF) |
| Available Diameters | 13 mm, 25 mm, 50 mm |
| Pore Sizes | 0.18 µm, 0.22 µm, 0.45 µm, 0.8 µm, 1.2 µm |
| Pack Size | 100 units per pack |
| Compliance | ISO 9001-manufactured |
| Brand | Autosense |
|---|---|
| Origin | Tianjin, China |
| Manufacturer Type | Authorized Distributor |
| Country of Origin | China |
| Model | FB-04T |
| Capacity | 30 mL |
| Filter Diameter | 25 mm |
| Components | Filter Funnel (30 mL), Central Filter Holder, Vacuum Filtration Flask, Aluminum Clamp Set |
| Price Range | USD 140 – 1,400 |
| Brand | Autosience |
|---|---|
| Model | AL-01 |
| Construction | Borosilicate glass filtration assembly (FB-10T, 1000 mL capacity) with sintered glass frit (porosity: 10–16 µm) |
| Vacuum source | AP-01P oil-free diaphragm vacuum pump (ultimate vacuum: ≤70 mbar, flow rate: 25 L/min) |
| Optional upgrade | FB-20T 2000 mL filtration kit |
| Filtration volume per cycle | 300 mL (funnel capacity) |
| Compatibility | Standard 29/32 and 45/50 joint sizes for interchangeability with major international brands (e.g., Whatman, Millipore, Thermo Fisher) |
| Regulatory alignment | Designed for GLP-compliant lab environments |
| Brand | AVESTIN |
|---|---|
| Origin | Canada |
| Manufacturer | Avestin Inc., Ottawa, Canada |
| Model | EmulsiFlex-C50 |
| Max Operating Pressure | 30,000 psi (207 MPa) |
| Flow Rate | 15–50 L/h |
| Minimum Sample Volume | 25 mL |
| Drive Type | Pneumatic Dual-Piston |
| Safety | Auto-circuit cutoff with 20% transient overpressure tolerance |
| Construction Material | ASTM-certified 316L stainless steel |
| Sterilization | Direct steam-in-place (SIP), O-ring-free fluid path |
| Compliance | FDA 21 CFR Part 11-ready architecture, GMP-compliant design |
| Optional Accessories | Extrusion filter, heat exchanger |
| Instrument Category | High-Pressure Cell Disruptor |
| Brand | Avestin |
|---|---|
| Origin | Canada |
| Manufacturer | Avestin Inc., Ottawa, Canada |
| Model | EmulsiFlex-C55 |
| Pressure Range | 0–30,000 psi (0–207 MPa) |
| Flow Rate | 15–50 L/h |
| Minimum Sample Volume | 7 mL |
| Drive Type | Pneumatic Dual-Piston |
| Compliance | GMP, FDA 21 CFR Part 11–ready architecture, ASME BPE–compatible materials |
| Construction Material | Electropolished 316L stainless steel |
| Sterilization | SIP/CIP-capable, O-ring–free fluid path |
| Safety | Auto-cutoff circuit protection, transient overpressure tolerance up to 20% |
| Brand | Avestin |
|---|---|
| Origin | Canada |
| Manufacturer | Avestin Inc., Ottawa, ON |
| Model | EmulsiFlex-D20 |
| Operating Pressure | 35,000 psi (2,413 bar) / Max Continuous: 30,000 psi (2,070 bar) |
| Flow Rate | 20 L/h |
| Minimum Sample Volume | 50 mL |
| Sample Hold-up Volume | <1 mL |
| Drive Type | Electric Single-Plunger Pump |
| Dimensions (W×D×H) | 730 × 600 × 640 mm |
| Weight | 120 kg |
| Power Supply | 3-phase, 50/60 Hz, 230/400 V |
| Construction Material | ASTM A240 316L stainless steel + ceramic wear components |
| Sterilization | Direct steam-in-place (SIP) compatible |
| Compliance | GMP-compliant design, FDA 21 CFR Part 11–ready software architecture, USP <1043> and ISO 22442-1 applicable for bioprocess equipment qualification |