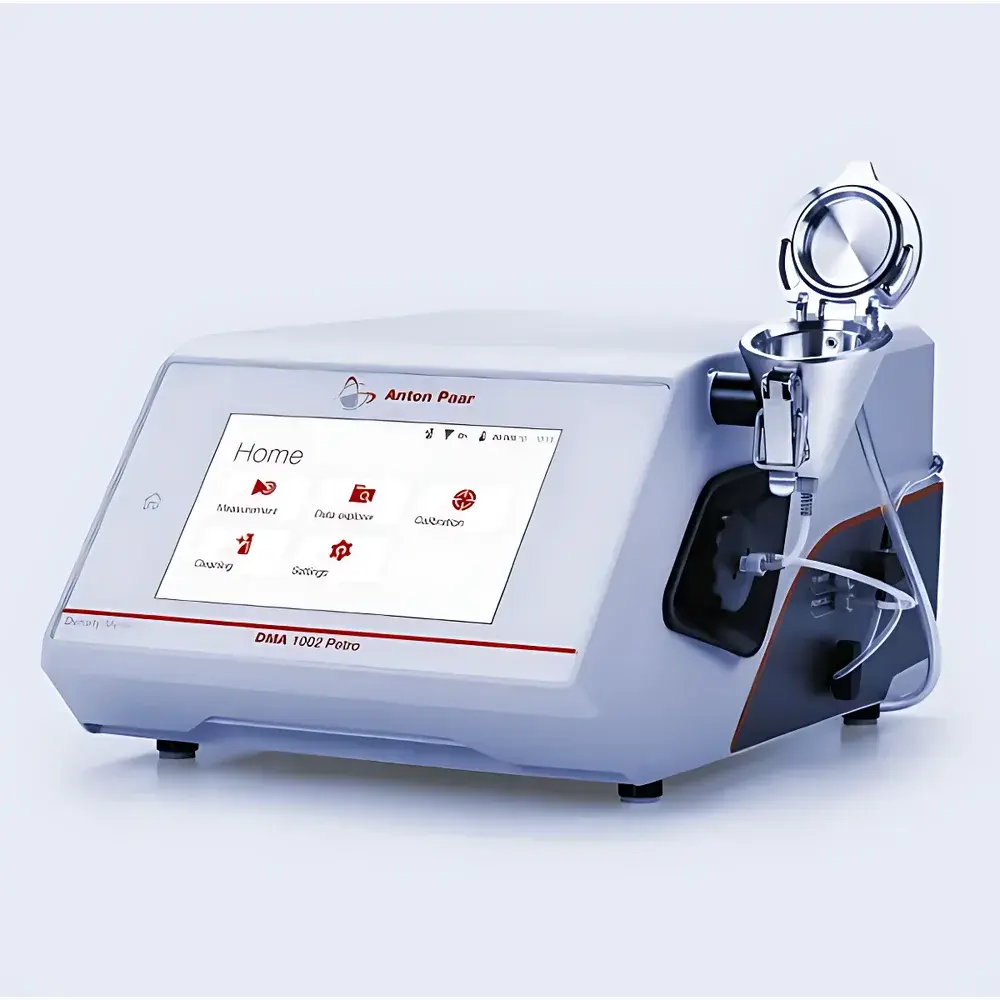
Anton Paar DMA 502/1002 Compact Digital Oscillating U-Tube Density Meter
| Brand | Anton Paar |
|---|---|
| Origin | Austria |
| Manufacturer | Anton Paar GmbH |
| Type | Oscillating U-tube Densimeter |
| Form Factor | Benchtop Laboratory Instrument |
| Sample Type | Liquids and Semi-solids (with optional kits) |
| Density Range | 0 to 3 g/cm³ |
| Density Accuracy | 0.001 g/cm³ (DMA 502), 0.0001 g/cm³ (DMA 1002) |
| Density Repeatability | 0.0002 g/cm³ (DMA 502), 0.00005 g/cm³ (DMA 1002) |
| Temperature Range | 5–35 °C (ambient), 15–40 °C (measurement, DMA 502), 15–60 °C (measurement, DMA 1002) |
| Temperature Resolution | 0.01 °C |
| Minimum Sample Volume | ~1 mL |
| Display | 7-inch TFT WVGA (800 × 480 px) PCAP capacitive touchscreen |
| Dimensions (L×W×H) | 375 × 280 × 180 mm |
| Weight | 13.5 kg |
| Connectivity | 1× Ethernet, 3× USB, 1× RS232 |
| Data Storage | 5000 measurements, 400 user-defined methods, 140 built-in conversion tables |
Overview
The Anton Paar DMA 502/1002 is a high-precision benchtop oscillating U-tube density meter engineered for traceable, ISO-compliant density and related physical property measurements in regulated and research-intensive laboratory environments. Based on the fundamental principle of oscillating tube metrology—where the resonant frequency of a precisely machined U-shaped glass tube filled with sample correlates directly with its density—the instrument delivers rapid, non-destructive, and highly reproducible results across a broad density range (0–3 g/cm³). Its dual-model architecture provides calibrated resolution options: the DMA 502 achieves three-digit precision (±0.001 g/cm³), while the DMA 1002 meets stringent pharmaceutical and petrochemical requirements with four-digit accuracy (±0.0001 g/cm³) and full compliance with USP , EP 2.2.5, ASTM D4052, ASTM D5002, and FDA 21 CFR Part 11 for electronic records and signatures. The system operates without forced ventilation or external cooling, enabling stable deployment in ambient-controlled labs, QC suites, and pilot-scale production areas.
Key Features
- Oscillating U-tube technology with borosilicate glass measurement cell and PTFE/ETFE wetted parts for chemical resistance and long-term calibration stability.
- U-View real-time imaging: A built-in optical module captures scalable, high-contrast images of the filled U-tube, enabling visual verification of meniscus position, bubble presence, and filling homogeneity prior to measurement.
- FillingCheck algorithm: Automatically detects micro-bubbles within seconds by analyzing damping behavior during initial oscillation—eliminating false readings caused by entrained air.
- Intuitive 7-inch PCAP touchscreen interface with glove-compatible operation, supporting guided workflows, multilingual UI, and context-sensitive help.
- Integrated environmental compensation: Real-time correction for ambient temperature, atmospheric pressure, and humidity ensures metrological consistency across geographic and seasonal variations.
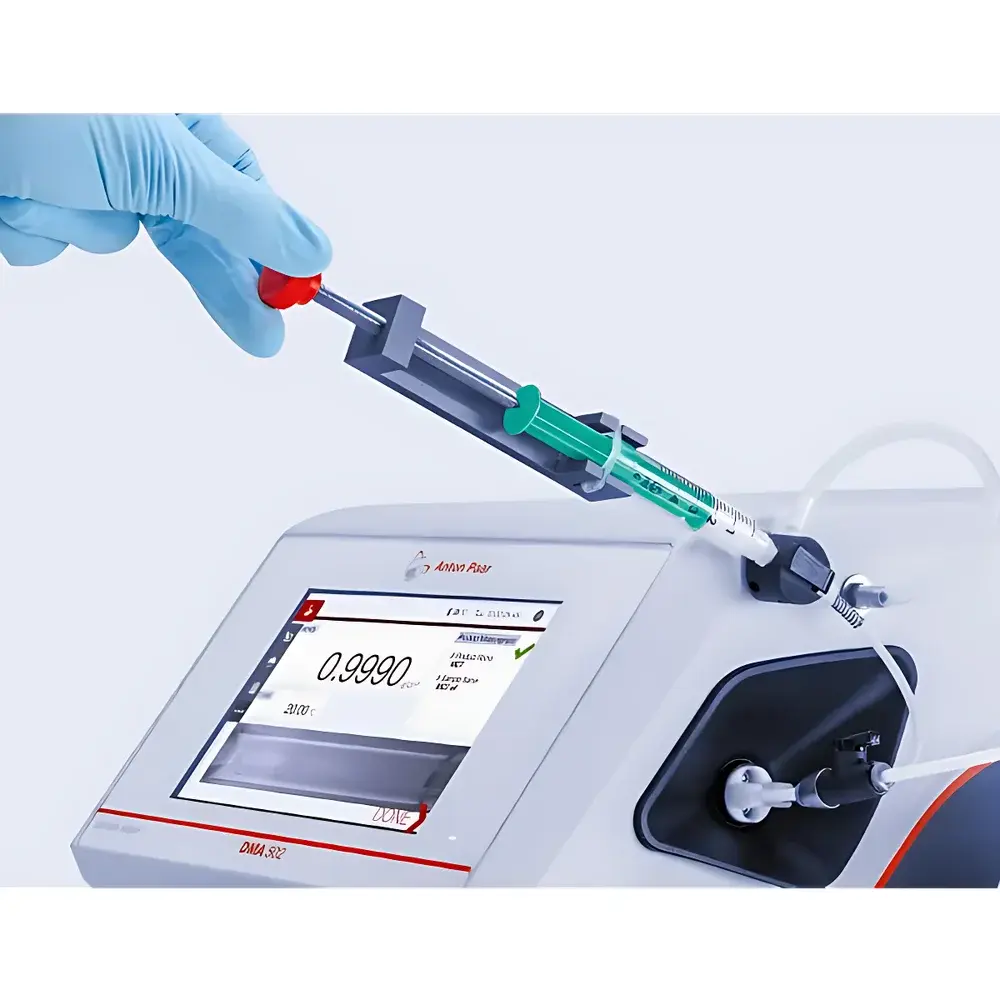
- Modular sample introduction: Supports syringe filling (manual or with integrated bracket), gravity-fed funnel, Xsample 200 automated peristaltic pump, and dedicated semi-solid kits for pastes, creams, and viscous formulations.
- Onboard temperature scanning: Enables dynamic density vs. temperature profiling over user-defined ranges (e.g., 15–60 °C) with 0.01 °C resolution and ±0.03 °C accuracy (DMA 1002).
Sample Compatibility & Compliance
The DMA 502/1002 accommodates liquids, solutions, suspensions, and semi-solids—including ethanol-water mixtures, organic solvents, acids, bases, pharmaceutical syrups, food emulsions, and polymer melts (with optional high-viscosity kit). Its <1 mL minimum sample volume reduces reagent consumption and waste generation, critical for expensive or hazardous materials. All wetted surfaces are constructed from inert borosilicate glass, PTFE, and ETFE—validated for compatibility with aggressive media per ISO 17025 and GMP Annex 15. Regulatory alignment includes full audit trail support via AP Connect integration, electronic signature capability, role-based access control, and 21 CFR Part 11–compliant data integrity features (e.g., immutable timestamps, operator ID logging, change history). Calibration certificates traceable to NIST and PTB standards are available through accredited ISO/IEC 17025 laboratories.
Software & Data Management
Data acquisition, processing, and reporting are managed through the embedded firmware and optionally extended via Anton Paar’s AP Connect platform—a secure, web-based laboratory execution system (LES) that centralizes instrument data, method templates, and audit logs. Up to 5000 measurements are stored locally with metadata (operator, timestamp, method ID, environmental conditions). The instrument hosts 400 preconfigured methods and 140 industry-specific conversion tables—including alcoholometry (OIML R22), saccharimetry (ICUMSA), API gravity, H₂SO₄/HCl concentration, and specific gravity (SG) to density transforms. Export formats include CSV, PDF reports, and XML for LIMS/SAP integration. Firmware updates and remote diagnostics are supported via Ethernet or USB.
Applications
- Pharmaceutical QC: Excipient density verification, active ingredient assay via density correlation, and formulation stability monitoring per ICH Q5C.
- Petrochemical analysis: Crude oil API gravity, biodiesel blend verification (EN 14214), and distillate density grading per ASTM D1298.
- Foods & Beverages: Alcohol content in spirits (OIML R22), Brix/sugar concentration in juices, and density-based authenticity screening for honey and olive oil.
- Chemical manufacturing: Concentration control of caustic soda, sulfuric acid, and lithium electrolytes; batch release testing per ISO 8217.
- Academic research: Thermophysical property studies, solvent mixture characterization, and validation of computational fluid models.
FAQ
What is the difference between DMA 502 and DMA 1002?
The DMA 502 provides density accuracy of ±0.001 g/cm³ and temperature accuracy of ±0.3 °C, suitable for general QC applications. The DMA 1002 offers enhanced metrological performance: ±0.0001 g/cm³ density accuracy, ±0.03 °C temperature accuracy, extended temperature range up to 60 °C, and full 21 CFR Part 11 compliance.
Can the instrument measure viscous or paste-like samples?
Yes—when equipped with the optional semi-solid sample kit, including a heated syringe holder and low-shear filling protocol, both models support reliable density determination of pastes, gels, and creams with minimal air entrapment.
Is external calibration required?
No routine external calibration is needed. The system performs automatic single-point validation using certified reference materials (e.g., air and water), and full multi-point calibration is performed offline by authorized service engineers with traceable standards.
How does FillingCheck detect bubbles?
FillingCheck analyzes the decay envelope of the initial oscillation signal; micro-bubbles increase damping, producing a measurable deviation from the expected exponential decay profile—detected before the main density measurement begins.
What data security protocols are implemented?
AP Connect enforces TLS 1.2+ encryption, password policies compliant with NIST SP 800-63B, session timeouts, and immutable audit trails meeting ALCOA+ principles (Attributable, Legible, Contemporaneous, Original, Accurate, Complete, Consistent, Enduring, Available).