BARRAQUER Automatic Eyelid Retractor R22012-45
| Brand | RWD |
|---|---|
| Origin | Shenzhen, China |
| Manufacturer Type | Direct Manufacturer |
| Product Origin | Domestic (China) |
| Model | R22012-45 |
| Pricing | Available Upon Request |
Overview
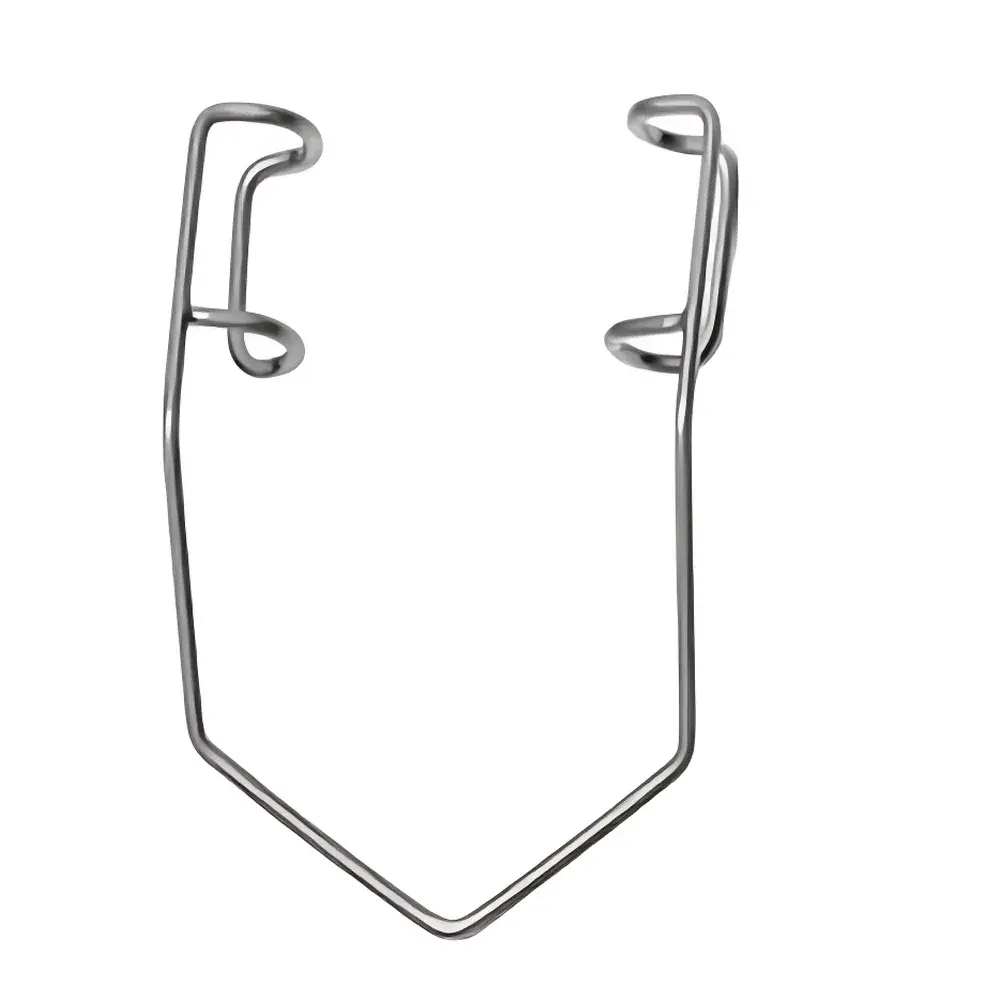
The BARRAQUER Automatic Eyelid Retractor R22012-45 is a precision-engineered ophthalmic surgical instrument designed for stable, hands-free eyelid retraction during anterior segment procedures. Based on the classic Barraguer mechanical principle—utilizing controlled spring-loaded tension and anatomically contoured stainless-steel blades—the R22012-45 integrates an automatic self-retaining mechanism that maintains consistent 4.5 cm interpalpebral aperture without manual adjustment or external fixation. This design eliminates surgeon hand fatigue, reduces intraoperative micro-movement, and preserves optimal exposure of the cornea, limbus, and conjunctival fornices. The device is intended for single-patient use in sterile environments and complies with ISO 13485–certified manufacturing protocols for Class I medical devices under regulatory frameworks applicable to ophthalmic retractors.
Key Features
- Automatic self-retaining mechanism with calibrated torsion-spring actuation ensures repeatable 4.5 cm eyelid separation without slippage or over-compression.
- Ergonomic handle with textured non-slip grip facilitates intuitive one-handed deployment and precise positioning under surgical loupes or microscope illumination.
- Blade geometry optimized for human palpebral anatomy: curved upper and lower blades minimize pressure necrosis risk while maximizing exposure of the superior and inferior fornices.
- Fully autoclavable stainless-steel construction (AISI 304) meets EN ISO 17664 requirements for cleaning, disinfection, and sterilization validation.
- Modular assembly enables rapid disassembly for thorough cleaning and inspection of hinge joints and spring components between uses.
- Compact footprint (125 mm length, 22 mm max width) allows compatibility with standard ophthalmic trays and minimizes field-of-view obstruction during phacoemulsification or gonioscopy.
Sample Compatibility & Compliance
The R22012-45 is validated for use across adult and pediatric patients with normal or mildly ptotic eyelid morphology. It is not indicated for patients with severe cicatricial entropion, acute blepharitis, or post-traumatic eyelid deformity requiring custom retraction solutions. Device labeling and packaging comply with MDR (EU 2017/745) Annex I essential requirements for mechanical safety, biocompatibility (ISO 10993-1), and labeling clarity. Sterilization instructions are aligned with AAMI ST79 and ANSI/AAMI ST55 standards. The instrument carries CE marking as a Class I non-sterile, reusable surgical retractor; final sterilization is performed by the end-user facility per local SOPs.
Software & Data Management
As a purely mechanical, non-electronic surgical instrument, the R22012-45 does not incorporate embedded software, firmware, or digital interfaces. No data acquisition, storage, or transmission capabilities are present. Consequently, it is exempt from FDA 21 CFR Part 11, IEC 62304, and EU MDR software-related conformity assessments. Instrument traceability is maintained via laser-engraved model number (R22012-45), batch code, and manufacturer logo on the handle—enabling full audit trail for GLP/GMP-aligned reprocessing logs and surgical inventory management systems.
Applications
- Corneal transplantation (penetrating keratoplasty, DMEK, DSAEK)
- Phacoemulsification cataract surgery with coaxial or micro-incision techniques
- Laser-assisted in situ keratomileusis (LASIK) flap creation and interface inspection
- Conjunctival autograft harvesting and pterygium excision
- Anterior chamber paracentesis and intracameral injection procedures
- Intraoperative gonioscopy and angle visualization in glaucoma surgery
FAQ
Is the R22012-45 compatible with magnetic eyelid speculums or integrated OR table systems?
No—it operates independently as a manual mechanical retractor and contains no ferromagnetic components or docking interfaces.
What is the recommended reprocessing cycle for this instrument?
Follow a validated sequence: pre-rinse → enzymatic soak (pH 7–8, 2–5 min) → ultrasonic cleaning (40 kHz, 5 min) → thermal disinfection (A0 ≥ 60) → steam sterilization (134°C, 3 min, gravity cycle).
Can the 4.5 cm aperture be adjusted intraoperatively?
No—the aperture is fixed by spring calibration; variable-aperture models are not offered in this series.
Does RWD provide instrument-specific training or procedural video resources?
Yes—complimentary access to IFU videos, OR integration guides, and reprocessing checklists is available through the RWD Clinical Support Portal upon device registration.
Is this device registered with the U.S. FDA or Health Canada?
It is currently distributed internationally under CE marking; FDA 510(k) clearance and Health Canada Medical Device License applications are in progress as of Q3 2024.