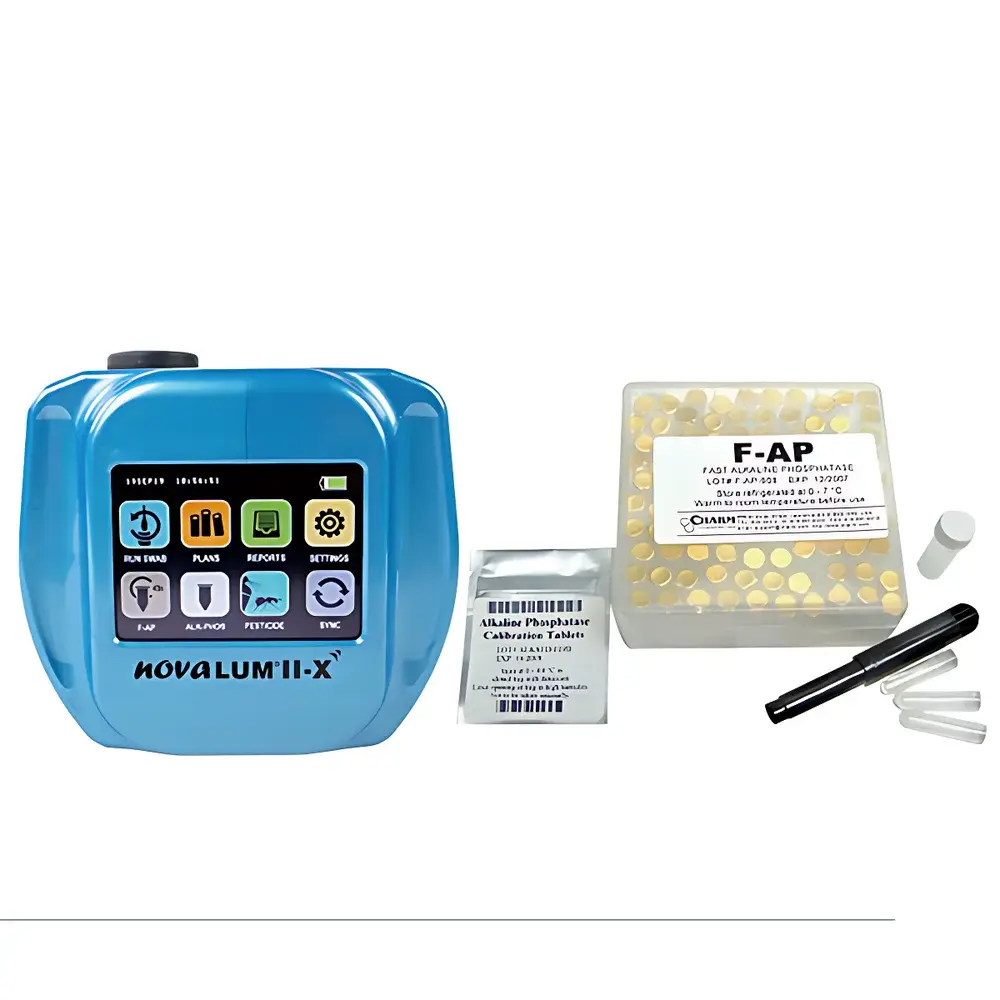
Charm novaLUM II Alkaline Phosphatase Rapid Detection System
| Brand | Charm |
|---|---|
| Model | novaLUM II |
| Origin | Beijing, China |
| Instrument Type | Dairy-Specific Analyzer |
| Automation Level | Semi-Automatic |
| Analysis Time | 45 s per sample (for alkaline phosphatase in pasteurized milk) |
| Repeatability | <0.5% RSD |
| Accuracy | 99.9% |
| Detection Limit | 20 mU/L |
| Interface | Touchscreen |
| Calibration | Fully Automatic |
| Data Storage Capacity | 16,384 test records |
| Compliance | NY/T 3799–2020 (PRC), ISO 22160 / IDF 209, NCIMS-recognized, NZFSA-verified, TDIA-validated, APHA F-AP method |
Overview
The Charm novaLUM II Alkaline Phosphatase Rapid Detection System is a semi-automatic, dairy-specific luminometric analyzer engineered for objective, quantitative assessment of alkaline phosphatase (ALP) activity in pasteurized milk and dairy products. It operates on the principle of bioluminescent enzymatic amplification: ALP catalyzes the dephosphorylation of a proprietary chemiluminescent substrate (e.g., luciferin phosphate), generating free luciferin that reacts with recombinant firefly luciferase to emit light proportional to ALP concentration. This photonic signal is captured by a high-gain, low-noise photomultiplier tube (PMT), enabling detection at the regulatory threshold of 20 mU/L—well below the maximum permissible limit (typically 350 mU/L) defined in international pasteurization verification standards. Unlike conventional colorimetric or fluorometric assays, the novaLUM II requires no incubation, no secondary reagents, and no sample pre-treatment—delivering a legally defensible, real-time pass/fail determination within 45 seconds per sample.
Key Features
- High-sensitivity PMT detection architecture with optimized quantum efficiency across 300–650 nm spectral range, delivering superior signal-to-noise ratio (>10⁴:1) and dynamic linear range spanning 20–5000 mU/L ALP
- Semi-automated workflow integrating integrated pipette actuation, auto-zeroing optics, and temperature-stabilized reaction chamber (±0.5°C)
- Full automatic calibration using built-in reference standards; eliminates manual calibration drift and operator dependency
- Industrial-grade touchscreen interface (7-inch capacitive, glove-compatible) with intuitive icon-driven navigation and multilingual OS support (English, Spanish, French, Mandarin)
- Onboard data management supporting GLP-compliant audit trails: timestamped results, user ID logging, instrument ID, calibration history, and environmental metadata (ambient temperature/humidity)
- Ruggedized chassis rated IP54 for operation in ambient dairy processing environments (10–40°C, 30–85% RH non-condensing)
Sample Compatibility & Compliance
The novaLUM II is validated exclusively for raw and heat-treated bovine milk, skim milk, standardized whole milk, and UHT-treated milk. It is not intended for use with plant-based milks, fermented dairy, or whey-based matrices without prior matrix validation. Regulatory alignment includes full conformance with ISO 22160 / IDF 209 (the internationally harmonized reference method for ALP detection in milk), NY/T 3799–2020 (People’s Republic of China agricultural industry standard), and APHA Standard Methods for the Examination of Dairy Products (F-AP protocol). The system meets NCIMS (National Conference on Interstate Milk Shipments) requirements for routine pasteurization verification and has received formal recognition from NZFSA and TDIA for compliance with national dairy safety frameworks. All firmware and assay protocols are designed to support 21 CFR Part 11–compliant electronic records when paired with Charm’s optional LIMS integration module.
Software & Data Management
The embedded firmware supports local storage of up to 16,384 test records with full traceability—including sample ID, operator ID, test date/time, ALP concentration (mU/L), pass/fail status relative to configurable thresholds (e.g., 350 mU/L), and raw luminescence counts (RLU). Data export is supported via USB 2.0 (CSV/Excel format) or optional Ethernet/Wi-Fi connectivity for secure transmission to enterprise quality management systems (QMS). Audit trail functionality logs all critical events: calibration executions, parameter modifications, user logins/logouts, and result deletions—with immutable timestamps and digital signatures. Software updates are delivered via encrypted firmware packages validated through SHA-256 checksum verification.
Applications
- Verification of pasteurization efficacy in Grade A fluid milk production lines
- QC release testing of incoming raw milk at receiving docks
- Root cause analysis during thermal process deviations (e.g., flow rate anomalies, heater fouling)
- Validation and requalification of HTST and VHT pasteurizers per FDA Pasteurized Milk Ordinance (PMO) Annex G
- Supporting HACCP prerequisite programs and FSMA Preventive Controls for dairy processors
- Third-party laboratory accreditation under ISO/IEC 17025 for ALP testing services
FAQ
Is the novaLUM II compliant with FDA Pasteurized Milk Ordinance (PMO) requirements?
Yes—the system implements the APHA F-AP method and satisfies PMO Annex G criteria for rapid ALP verification when used with Charm-certified reagents and documented SOPs.
Can the instrument be used for raw milk screening?
No—raw milk contains endogenous ALP levels far exceeding regulatory thresholds; the novaLUM II is validated only for heat-treated milk where residual ALP indicates inadequate pasteurization.
Does the system require daily maintenance beyond calibration?
Routine cleaning of the optical path and sample chamber is recommended after every 50 tests; no optical alignment or lamp replacement is required during the instrument’s 5-year service life.
How is data integrity ensured during electronic record transfer?
All exported files include embedded digital signatures and cryptographic hashes; optional QMS integration enforces role-based access control and automated backup to redundant NAS infrastructure.
What training and technical support does Charm provide?
Charm offers on-site installation qualification (IQ), operational qualification (OQ), and comprehensive operator training—including method validation guidance, troubleshooting workflows, and annual refresher courses aligned with ISO 17025 competency requirements.