ELGA PURELAB Flex Series Ultra-Pure Water System
| Brand | ELGA |
|---|---|
| Origin | United Kingdom |
| Model | PURELAB Flex |
| Water Grade | Type I (Ultra-Pure) |
| Resistivity | 18.2 MΩ·cm at 25 °C |
| Heavy Metals & Soluble Silica | <0.1 ppb |
| Total Organic Carbon (TOC) | <5 ppb |
| Microbial Count | <0.001 CFU/mL |
| Endotoxins | <0.001 EU/mL |
| Particles (>0.1 µm) | <0.2 particles/mL |
| RO Production Rate | Up to 10 L/h |
| Dispensing Flow Rate | Adjustable from dropwise to 2 L/min |
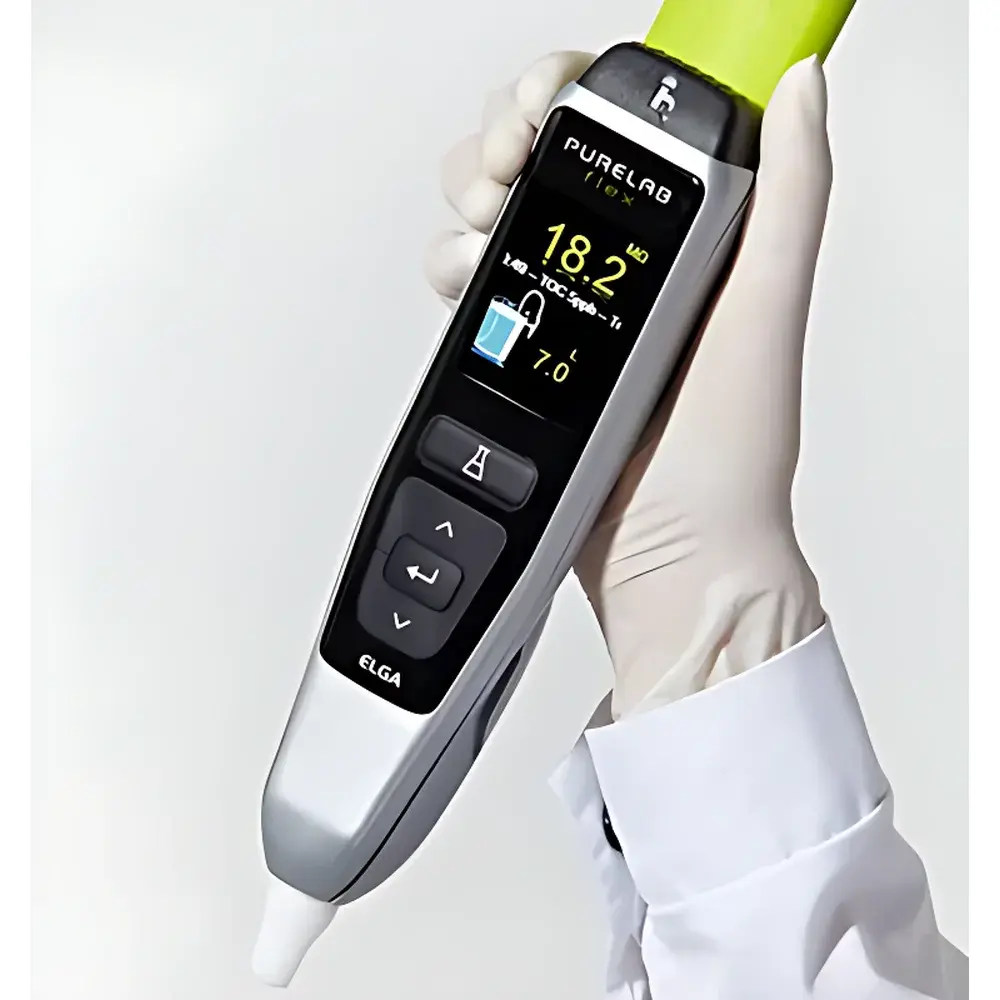
| Integrated Touchscreen Display | Real-time monitoring of resistivity, conductivity, TOC, temperature, and system status |
Overview
The ELGA PURELAB Flex Series is a modular, high-performance ultra-pure water system engineered for research-grade laboratories requiring consistent, trace-level purity across diverse analytical, biological, and pharmaceutical applications. Utilizing a multi-stage purification architecture—including reverse osmosis (RO), electrodeionization (EDI), dual-wavelength ultraviolet oxidation (185/254 nm), and final polishing via nuclear-grade mixed-bed ion exchange and 0.1 µm ultrafiltration—the system delivers Type I water compliant with ASTM D1193-20, ISO 3696:1987, and CLSI EP21-A guidelines. Its design centers on real-time, point-of-use (POU) water quality assurance: integrated sensors in the recirculating loop ensure displayed parameters—resistivity, TOC, temperature, and flow rate—precisely reflect the water delivered at the dispensing handle. This eliminates measurement lag and guarantees “what you see is what you use,” a critical requirement for GLP/GMP-regulated environments and ISO/IEC 17025-accredited testing labs.
Key Features
- Modular architecture supporting scalable configurations—from standalone RO-only (Type III) to fully integrated ultra-pure (Type I) systems—with optional add-ons including UV-TOC monitors, endotoxin-retentive filters, and DNase/RNase-free polishing cartridges.
- Ergonomic, height-adjustable dispensing handle with intuitive touchscreen interface; enables precise, programmable dispensing from 50 mL to 60 L, including dropwise, timed, or volume-triggered modes.
- Real-time, in-line TOC monitoring (standard on Flex 2–4 models) using high-sensitivity UV-persulfate oxidation and NDIR detection, meeting USP and EP 2.2.44 requirements for organic contaminant control.
- Onboard data logging with ≥12-month storage capacity; supports USB export, network-based reporting, and audit-trail generation compliant with FDA 21 CFR Part 11 for electronic records and signatures.
- Self-diagnostic firmware with predictive maintenance alerts; rapid cartridge replacement via tool-free, color-coded modules; validated sanitization protocols using integrated thermal or chemical disinfection kits (no consumable reagents required).
Sample Compatibility & Compliance
The PURELAB Flex accommodates a broad spectrum of sample-sensitive workflows without introducing interference. Its output meets or exceeds specifications for critical applications including ICP-MS, HPLC-MS, single-cell sequencing, and mammalian cell culture. The system’s ultra-low endotoxin (<0.001 EU/mL) and nuclease-free performance (DNase <0.005 ng/mL; RNase <0.001 ng/mL) are validated per USP and , enabling safe use in primary neuronal culture and CRISPR-Cas9 transfection protocols. All wetted materials comply with USP Class VI biocompatibility standards; stainless steel 316L distribution loops and PTFE/PFA fluid paths minimize leachables. Full compliance documentation—including Factory Acceptance Test (FAT) reports, IQ/OQ templates, and material traceability certificates—is provided for regulatory submissions under ISO 13485, GMP Annex 1, and EU GMP Guide Chapter 4.
Software & Data Management
The embedded PURELAB Connect software platform provides secure, role-based access to system diagnostics, historical trends, and customizable report generation. Data integrity is maintained through time-stamped, immutable logs with user authentication, electronic signatures, and automatic backup to external NAS or cloud repositories. Export formats include CSV, PDF, and XML, compatible with LIMS integration via HL7 or RESTful API. Audit trails record all parameter changes, alarm events, and maintenance actions—fully traceable to operator ID and timestamp—supporting inspections by FDA, EMA, or UK MHRA. Optional remote monitoring allows centralized fleet management across multi-site laboratory networks without compromising cybersecurity (TLS 1.2 encryption, configurable firewall rules).
Applications
- Trace elemental analysis: ICP-MS, GF-AAS, and ICP-OES where sub-ppt metal contamination must be excluded.
- Chromatography & mass spectrometry: Mobile phase preparation for UHPLC, LC-MS, and GC-MS to prevent column fouling and baseline drift.
- Molecular biology: PCR, qRT-PCR, NGS library prep, and electroporation requiring nuclease- and endotoxin-free water.
- Cell culture & regenerative medicine: Media formulation and passaging of stem cells, organoids, and primary cultures.
- Pharmaceutical QC: Buffer preparation for HPLC system suitability tests, dissolution media, and stability-indicating assays per ICH Q5C.
- Reference material calibration: Preparation of certified reference solutions for gravimetric standardization and CRM dilution.
FAQ
What water quality standards does the PURELAB Flex meet?
It consistently delivers Type I water per ASTM D1193-20, ISO 3696:1987, and CLSI EP21-A, with additional validation support for USP , EP 2.2.44, and JP 17 standards.
Can the system be integrated into an existing central water supply?
Yes—Flex units support both standalone operation and seamless integration as point-of-use polishers within centralized RO or deionized water networks, with optional pressure-regulated feed interfaces.
How is TOC monitored in real time?
A dedicated UV-persulfate oxidation module coupled with non-dispersive infrared (NDIR) detection provides continuous, in-line TOC quantification with ±0.5 ppb resolution and <30-second response time.
Is the system suitable for regulated GxP environments?
Fully—its electronic records, audit trail, user access controls, and 21 CFR Part 11-compliant signature workflow satisfy FDA, EMA, and PMDA requirements for data integrity in GLP, GMP, and clinical trial settings.
What maintenance intervals are recommended?
Pre-filters every 6 months; RO membrane every 2–3 years depending on feed water quality; polishing cartridges every 6–12 months based on usage and resistivity decay profiles—automatically tracked and alerted via the onboard system.