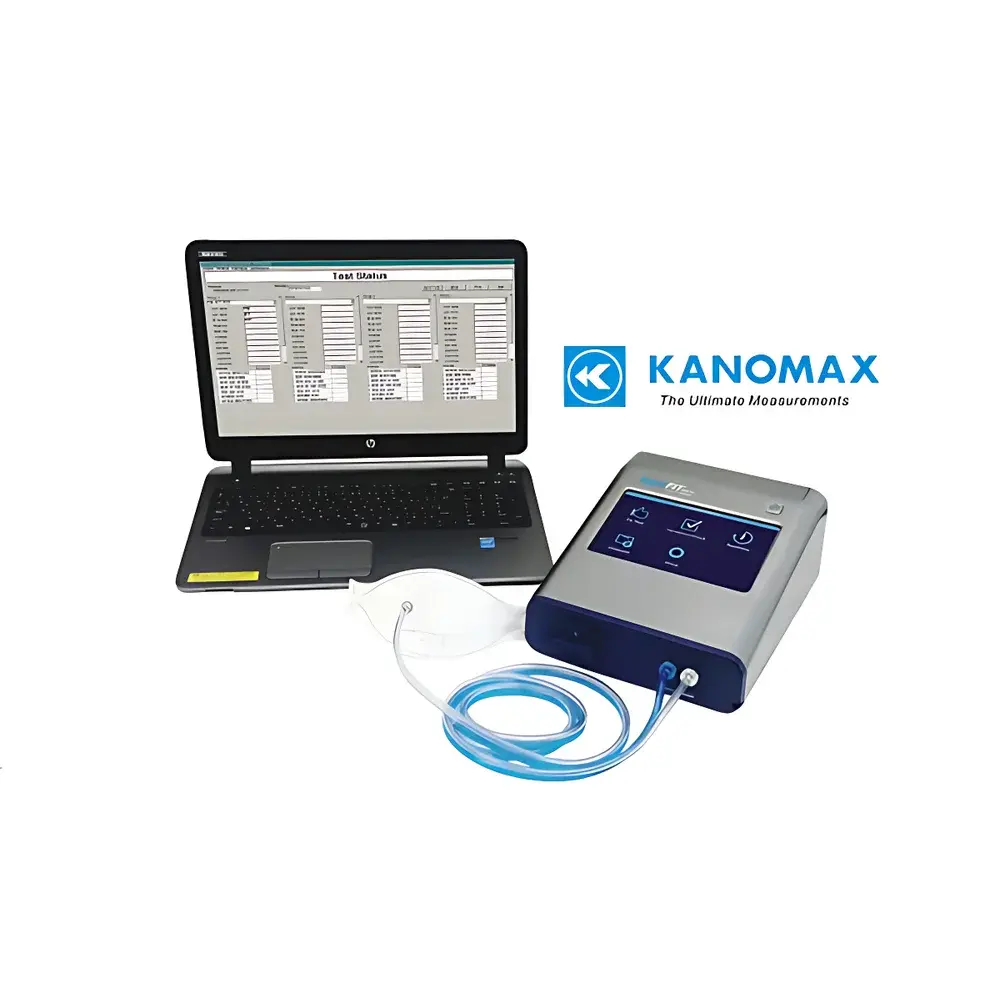
Kanomax AccuFIT 9000 Respirator Fit Testing System
| Brand | Kanomax |
|---|---|
| Origin | Japan |
| Model | 3000-0C |
| Concentration Range | 0–100,000 particles/cm³ |
| Particle Size Range | 0.02–1.0 µm |
| Sampling Flow Rate | 100 cm³/min |
| Total Flow Rate | 700 cm³/min |
| Fit Factor Measurement Method | Direct (Cout/Cin) |
| Test Aerosol | 99.5% ethanol + analytical-grade isopropanol |
| Display | 7-inch true-color touchscreen |
| Communication Interfaces | USB (Host ×2, Device ×1), Ethernet ×1, Wi-Fi enabled |
| Operating Temperature | 15–35 °C |
| Power Supply | AC 110–240 V, 50/60 Hz |
| Dimensions | 208 × 117 × 262 mm |
| Weight | 2.1 kg |
| Language Support | English, French, Spanish, Portuguese, Chinese |
| Multi-unit Control | Up to 4 units simultaneously via single PC |
| Data Export Format | Microsoft Excel |
Overview
The Kanomax AccuFIT 9000 Respirator Fit Testing System (Model 3000-0C) is a precision-engineered, quantitative respirator fit testing instrument designed to measure the actual in-use seal integrity of filtering facepiece respirators—including N95, KN95, KP95, N90, and P3/HEPA-class disposable masks—as well as half-face and full-face reusable respirators. Unlike qualitative methods relying on subjective sensory response, the AccuFIT 9000 employs a controlled, real-time particle challenge protocol based on condensation nucleus counter (CNC) technology. It quantifies the ratio of ambient aerosol concentration outside the respirator (Cout) to the concentration inside the respirator (Cin) during standardized breathing maneuvers—yielding a numerical Fit Factor (FF). This direct Cout/Cin methodology complies with internationally recognized performance criteria defined in OSHA 29 CFR 1910.134, CSA Z94.4, and ISO 16900-1:2015 for quantitative fit testing.
Key Features
- Quantitative CNC-based measurement with traceable particle detection across 0.02–1.0 µm size range, optimized for polydisperse ethanol/isopropanol challenge aerosols
- High-resolution 7-inch true-color touchscreen interface with intuitive workflow navigation and five-language support (English, French, Spanish, Portuguese, Chinese)
- Dual-flow architecture: independent 100 cm³/min sampling flow for internal respirator probe and 700 cm³/min total system flow for stable aerosol generation and chamber dynamics
- Integrated environmental and sampling ports enable simultaneous ambient background monitoring and intra-mask sampling without external manifolds
- Multi-instrument network capability: one Windows-based PC can coordinate up to four AccuFIT 9000 units via USB or Ethernet, enabling high-throughput occupational health screening
- Fully compliant with GB 2626–2019 (China’s “Respiratory Protective Devices – Self-Contained Filtering Devices Against Particles”), GB 19083–2010 (Medical Surgical Masks), and NIOSH-approved test protocols for N-series respirators
- On-device data logging and Excel-compatible export ensure audit-ready records for GLP/GMP-aligned quality assurance programs
Sample Compatibility & Compliance
The AccuFIT 9000 supports a broad spectrum of respiratory protection devices, including but not limited to: disposable N95/N99/N100, KN95/KP95/KP100, and P3-rated filtering facepieces; elastomeric half-face and full-face respirators; and powered air-purifying respirators (PAPRs) with tight-fitting hoods or facepieces. Its test protocol satisfies mandatory fit verification requirements under China’s GB 2626–2019 standard, which specifies minimum overall fit factors of ≥100 for KN95/KP95 respirators and ≥1000 for KN100/KP100 models—calculated from at least eight out of ten subjects achieving individual TIL (Total Inward Leakage) values below 8% and 2%, respectively. The system also meets OSHA’s pass/fail threshold of FF ≥100 for half-mask respirators and FF ≥500 for full-face units, with built-in validation routines traceable to NIST-traceable aerosol generators and calibrated photometers.
Software & Data Management
Data acquisition and analysis are managed through Kanomax’s proprietary AccuFIT Control Software (v3.x), compatible with Windows 10/11. Each test session automatically captures time-synchronized particle counts, flow rates, ambient temperature/humidity, operator ID, subject ID, respirator model, and test phase timestamps (e.g., normal breathing, deep breathing, head movement). All raw data and calculated Fit Factors are stored locally and exportable in native .xlsx format—with no proprietary binary dependencies. Audit trails include user login history, parameter change logs, and firmware version stamps, supporting compliance with FDA 21 CFR Part 11 requirements when deployed in regulated pharmaceutical or clinical trial environments. Optional CSV batch import enables pre-loaded subject rosters and respirator inventory mapping.
Applications
- Occupational health departments conducting annual or pre-deployment respirator fit certification per OSHA, CSA, or local regulatory mandates
- Hospital infection control units validating surgical N95 and medical-grade KN95 mask fit for frontline healthcare workers
- Industrial hygiene laboratories performing comparative fit assessments across respirator models, sizes, and facial anthropometries
- Respirator manufacturers executing design validation and iterative prototyping under ISO 16900-3:2022 (Laboratory test methods for fit testing systems)
- Academic research on facial interface leakage dynamics, beard interference, or age- and gender-related fit variability
- Government agencies administering national PPE compliance audits under GB 2626–2019 or EU EN 149:2001+A1:2009 frameworks
FAQ
What aerosol is used for challenge testing, and why is it specified?
Ethanol (99.5%) mixed with analytical-grade isopropanol generates a stable, monodisperse submicron aerosol ideal for CNC detection. Its volatility ensures rapid evaporation post-generation, minimizing residual deposition and enabling repeatable, low-background measurements.
Does the AccuFIT 9000 require annual calibration, and what standards apply?
Yes—annual calibration against NIST-traceable reference instruments is recommended. Kanomax provides certified calibration services aligned with ISO/IEC 17025:2017 and includes calibration certificates documenting flow rate accuracy (±2%), particle count linearity (R² ≥0.999), and temporal stability over 8-hour continuous operation.
Can the system be integrated into an existing LIMS or enterprise QA platform?
Via its Ethernet or USB-serial interface, the AccuFIT 9000 supports Modbus TCP and ASCII command protocols, enabling bidirectional data exchange with validated LIMS environments—subject to customer-defined API mapping and cybersecurity review.
Is the device suitable for field use, or strictly lab-based?
Its compact footprint (208 × 117 × 262 mm), lightweight chassis (2.1 kg), and AC universal input make it deployable in mobile occupational health vans or temporary cleanroom staging areas—provided ambient temperature remains within 15–35 °C and relative humidity stays below 80% non-condensing.
How does the system handle multi-user environments with shared hardware?
Role-based access control (administrator/operator/technician) is enforced via password-protected login. Each test record embeds user credentials, date/time stamps, and digital signatures—enabling full chain-of-custody documentation for regulatory inspections.