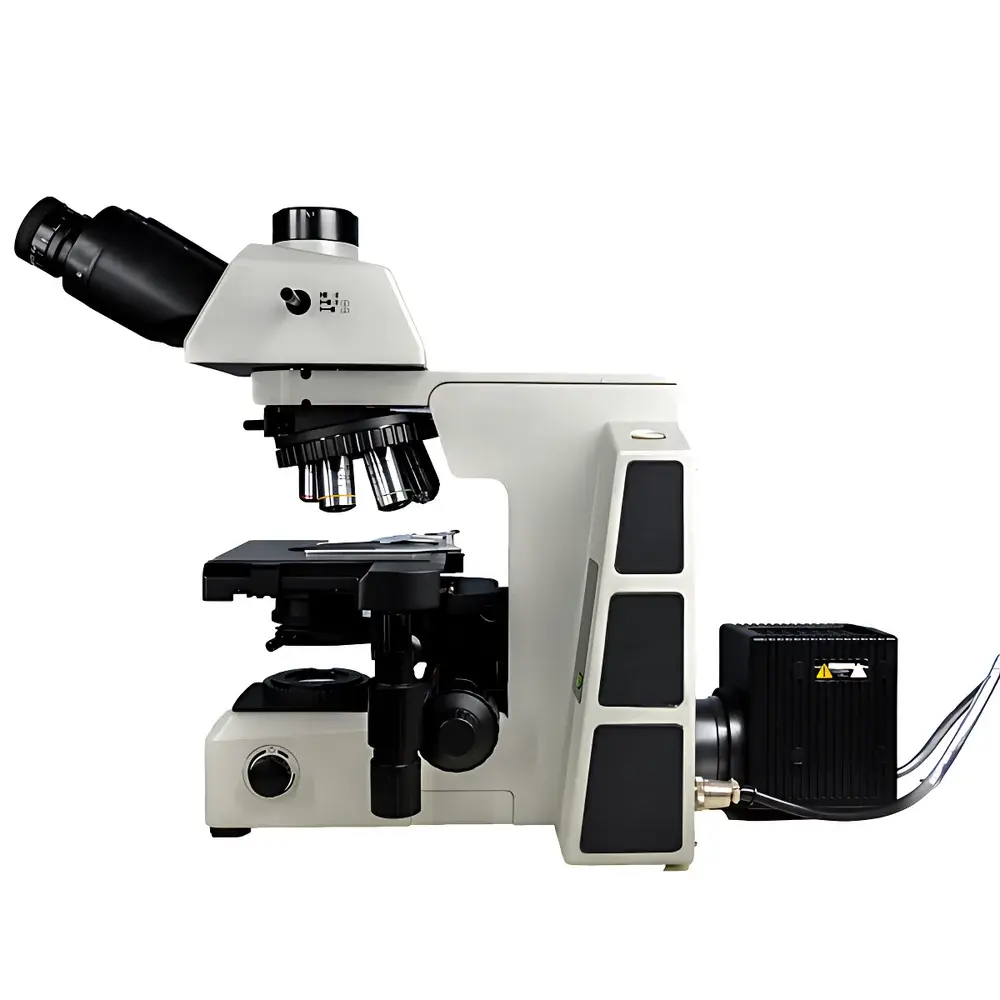
Lei-Tech LK-63 Research-Grade Upright Biological Microscope
| Brand | Lei-Tech |
|---|---|
| Origin | Tianjin, China |
| Manufacturer Type | Direct Manufacturer |
| Product Category | Upright Microscope |
| Model | LK-63 |
| Eyepiece Configuration | Trinocular |
| Optical System | Infinity-Corrected (UISC) |
| Illumination | High-Stability White LED (CCT 3700–5000 K) |
| Observation Modes | Brightfield, Phase Contrast, Fluorescence |
| Magnification Range | 40×–1000× (with 4×, 10×, 20×, 40×, 100× objectives) |
| Objective Mount | 6-Position Brightfield Turret (DIC-ready slot) |
| Condenser | Swing-out Achromatic Condenser (NA 0.9) with Adjustable Aperture Diaphragm |
| Focusing Mechanism | Coaxial Coarse/Fine Focus (25 mm coarse travel |
| Stage | Dual-layer Mechanical Stage with Bidirectional Linear Rails, 80 mm × 55 mm Travel, 0.1 mm Positioning Accuracy |
| Eyepieces | Widefield PL10×/25 mm, Adjustable Diopter, High-Eye-Point Design |
| Trinocular Port | Three-Position Beam Splitter (100:0 / 20:80 / 0:100) |
| Camera Interface | C-mount, Compatible with 20-MP (5440 × 3648) Digital Imaging Systems |
| Software Features | Real-Time Extended Depth of Field (EDF), Multi-Field Stitching, HDR Image Enhancement, Onboard Measurement Tools |
| Compliance | Designed for ISO 10993-1 pre-screening workflows |
Overview
The Lei-Tech LK-63 is a research-grade upright biological microscope engineered for precision life science applications requiring high-fidelity optical performance, multi-modal contrast flexibility, and reproducible digital documentation. Built upon an infinity-corrected optical architecture (UISC), the LK-63 eliminates inherent chromatic and spherical aberrations across its full magnification range (40×–1000×), enabling consistent resolution and flat-field imaging from low-magnification tissue screening to high-resolution subcellular observation. Its modular mechanical design separates the optical train from the structural frame—allowing seamless integration of fluorescence modules, DIC accessories, or phase contrast components without optical realignment. The system supports standardized brightfield, phase contrast, and epifluorescence observation through interchangeable condensers, turret-mounted objectives, and a fully decoupled illumination path. Unlike conventional finite-conjugate microscopes, the UISC platform ensures stable parfocality and maintains optical fidelity when adding beam-splitting optics or camera adapters—critical for quantitative image analysis and longitudinal experimental repeatability.
Key Features
- Infinity-corrected optical pathway with ultra-wide 25 mm field number eyepieces and plan semi-apochromatic objectives—optimized for minimal color fringing and maximal edge-to-edge sharpness.
- Stable white LED illumination (3700–5000 K CCT) with continuous intensity control, pre-centered lamp module, and integrated neutral density filters (ND6, ND25) and LBD daylight-balancing filter.
- Trinocular observation head with three-position beam splitter (100:0 / 20:80 / 0:100), supporting simultaneous visual inspection and high-resolution digital acquisition without compromise.
- Bidirectional linear rail stage with ergonomic left/right hand positioning, 80 × 55 mm travel range, 0.1 mm vernier scale, and dual slide夹 capable of parallel comparative analysis.
- Coaxial coarse/fine focusing mechanism featuring adjustable torque control, programmable upper limit stop, and 1 µm fine-step resolution—enabling both rapid survey scanning and nanoscale Z-stack acquisition.
- Digital imaging suite with real-time extended depth-of-field (EDF) synthesis, multi-field panoramic stitching, HDR tone mapping, and calibrated measurement tools compliant with ISO 10993-1 preliminary biocompatibility assessment protocols.
Sample Compatibility & Compliance
The LK-63 accommodates standard glass microscope slides (1 × 3 inches), petri dishes (up to 100 mm diameter), and culture flasks with optional stage adapters. Its swing-out achromatic condenser (NA 0.9) dynamically matches illumination NA to objective requirements—from low-power scanning (4×) to oil-immersion imaging (100×). For fluorescence applications, the system integrates standard excitation/emission filter cubes (340–700 nm range) and supports mercury or LED-based epi-illumination sources. All optical components are coated with anti-reflection and scratch-resistant films meeting ISO 9022-3 environmental durability standards. While not certified as medical device hardware per FDA 21 CFR Part 820, the LK-63’s optical stability, repeatable focus mechanics, and traceable illumination output make it suitable for preclinical research environments operating under GLP or ISO/IEC 17025-aligned quality frameworks. Optional software packages support 21 CFR Part 11-compliant audit trails, electronic signatures, and version-controlled image metadata logging.
Software & Data Management
The native imaging software provides vendor-agnostic TIFF/PNG export, time-lapse capture, region-of-interest (ROI) annotation, and pixel-calibrated morphometric analysis. Real-time EDF reconstruction processes Z-series stacks on-the-fly using gradient-based focus evaluation—eliminating post-acquisition computational bottlenecks. Multi-field stitching employs sub-pixel registration algorithms to generate gigapixel mosaics without seam artifacts. HDR processing applies localized luminance normalization to preserve detail in both nuclear staining and cytoplasmic background—enhancing diagnostic confidence in histopathological review. All measurements adhere to NIST-traceable calibration routines embedded in the firmware. Raw image data retains EXIF-style metadata including objective ID, magnification, exposure time, gain setting, and stage coordinates—facilitating FAIR (Findable, Accessible, Interoperable, Reusable) data principles in collaborative research infrastructures.
Applications
The LK-63 serves core functions across academic, clinical, and industrial life science domains: routine hematology and microbiology diagnostics in hospital laboratories; developmental biology time-lapse imaging in university core facilities; QC/QA of cell monolayers in biomanufacturing cleanrooms; and preclinical toxicology screening in contract research organizations (CROs). Its phase contrast capability enables label-free live-cell monitoring of mitosis, motility, and organelle dynamics; fluorescence compatibility supports immunofluorescence, FISH, and GFP-tagged protein localization studies; and brightfield EDF mode delivers publication-ready whole-slide images for pathology training repositories. The modular architecture permits future upgrades—including motorized stage control, automated filter wheels, or hyperspectral detection modules—without replacing the base optical platform.
FAQ
Is the LK-63 compliant with FDA or CE regulatory requirements for clinical diagnostics?
The LK-63 is classified as a research-use-only (RUO) instrument. It is not CE-marked as an IVD device nor cleared by the FDA for diagnostic use. However, its optical repeatability and mechanical stability meet technical prerequisites for inclusion in GLP-compliant study protocols.
Can third-party cameras be integrated with the trinocular port?
Yes—the C-mount interface adheres to ANSI/ISO 8539 specifications and supports industry-standard sensors up to 2/3″ format. Adapter rings for F-mount or M42 lenses are available upon request.
What maintenance intervals are recommended for the LED illumination system?
The LED module is rated for 50,000 hours at nominal output. No scheduled replacement is required within typical laboratory service life. Intensity drift remains within ±2% over 10,000 hours under continuous operation.
Does the system support DIC or polarization contrast?
The 6-position objective turret includes a dedicated slot for Wollaston prisms. DIC compatibility requires separate purchase of matching condenser and analyzer modules, which mount without optical recalibration.
How is calibration traceability established for measurement functions?
Each unit ships with a NIST-traceable stage micrometer and software-based calibration wizard. Users may register custom reference standards via pixel-to-micron mapping with adjustable uncertainty weighting.