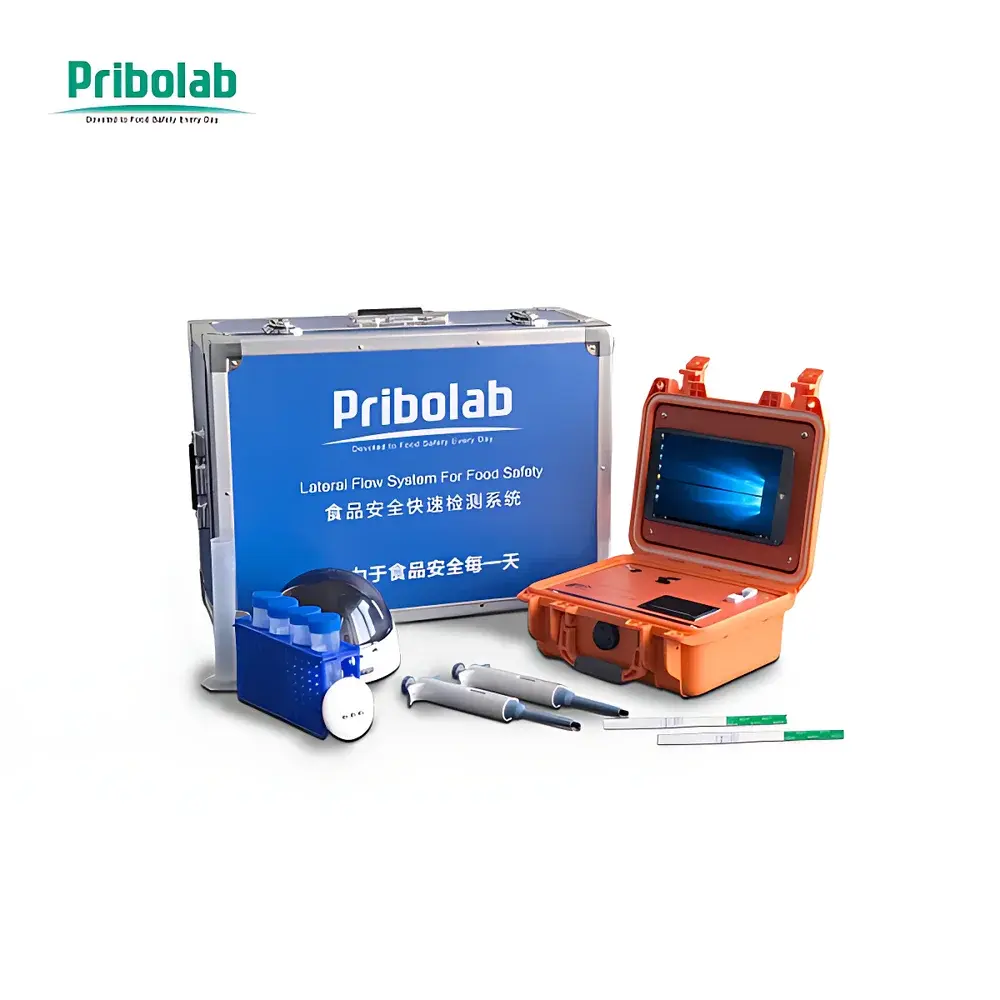
Pribolab LFR-II Food Safety Rapid Detection System
| Brand | Pribolab |
|---|---|
| Origin | Shandong, China |
| Manufacturer Type | Authorized Distributor |
| Country of Origin | China |
| Model | LFR-II |
| Pricing | Available Upon Request |
Overview
The Pribolab LFR-II Food Safety Rapid Detection System is a fully integrated, field-deployable immunoassay platform engineered for quantitative and qualitative analysis of mycotoxins, veterinary drug residues, pesticides, allergens, and pathogen antigens in complex food and agricultural matrices. Built upon lateral flow immunoassay (LFIA) principles—specifically colloidal gold–based rapid test strips—the system combines optical detection, embedded image processing, and standardized calibration algorithms to deliver laboratory-grade reproducibility in non-laboratory environments. Unlike conventional visual interpretation, the LFR-II eliminates subjective bias by digitizing strip signal intensity via high-resolution CCD imaging, applying pixel-level grayscale quantification, and correlating optical density with analyte concentration using pre-validated calibration curves. Its architecture supports full traceability, audit-ready data logging, and compliance with Good Laboratory Practice (GLP) documentation requirements for routine quality control in feed mills, grain elevators, spice processors, herbal medicine facilities, and regulatory inspection units.
Key Features
- 8.0-inch high-brightness capacitive touchscreen with native Windows 10 IoT Enterprise OS, enabling intuitive navigation, bilingual (English/Chinese) on-screen keyboard, and handwritten input support
- Integrated industrial-grade CCD sensor with auto-focus optics and adjustable LED illumination—optimized for consistent capture of colloidal gold test lines across variable ambient lighting conditions
- Real-time image correction engine: automatically compensates for lateral strip misalignment, rotational skew, and background non-uniformity using geometric transformation and adaptive thresholding
- On-device quantitative analysis: performs dual-band optical density measurement (test line vs. control line), applies matrix-specific calibration models, and outputs concentration values in ng/g or µg/kg with built-in uncertainty estimation
- Local storage capacity: 32 GB internal eMMC flash memory—retains ≥100,000 complete assay records, including raw images, processed data, operator ID, timestamp, GPS coordinates (if enabled), and environmental metadata
- Embedded diagnostics module: continuously monitors optical path integrity, sensor temperature drift, battery health, and firmware version; provides actionable error codes and self-recovery prompts
- Optional Wi-Fi 5 (802.11ac) and Bluetooth 5.0 modules enable secure TLS-encrypted data upload to cloud-based LIMS or enterprise QA dashboards
Sample Compatibility & Compliance
The LFR-II accepts all standard lateral flow test cassettes and dipstick formats compatible with colloidal gold immunochromatography—including those certified to AOAC PTM, FAPAS, and CNAS proficiency testing schemes. Validated sample types include ground cereal grains (corn, wheat, rice), animal feed pellets, dried herbs and botanicals, soy sauce and fermented condiments, and powdered dairy ingredients. Sample preparation is performed externally using Pribolab’s companion extraction kits (e.g., MycoStar™, VetStar™), ensuring adherence to ISO/IEC 17025 method validation guidelines. The system itself conforms to IEC 61000-4 electromagnetic compatibility standards and meets CE marking requirements for portable medical-grade diagnostic equipment under Directive 2017/746 (IVDR) classification as Class A non-invasive device.
Software & Data Management
Firmware v3.2+ includes FDA 21 CFR Part 11–compliant audit trail functionality: every data modification, user login, calibration event, or report generation is time-stamped, digitally signed, and immutable. Data export options include CSV (for Excel integration), PDF (print-ready reports with QR-coded result verification), and HL7-compatible JSON payloads for seamless ingestion into enterprise quality management systems (QMS). Cloud synchronization utilizes AES-256 encryption at rest and in transit; user roles (Operator, Supervisor, Administrator) enforce granular access control per ISO 27001 Annex A.8.2.3.
Applications
- Routine screening of aflatoxin B1, deoxynivalenol (DON), zearalenone (ZEN), and ochratoxin A in incoming raw materials at feed manufacturing plants
- On-site verification of antibiotic residue compliance (e.g., chloramphenicol, sulfonamides) in aquaculture and poultry supply chains
- Batch release testing of traditional Chinese medicinal herbs for illegal dye adulterants (e.g., Sudan Red, Rhodamine B)
- Field audits by national food safety regulators performing unannounced inspections at food markets and border checkpoints
- Training and competency assessment: built-in video tutorial library supports SOP-driven operator certification aligned with ISO/IEC 17025 Clause 6.2.5
FAQ
Does the LFR-II require external calibration standards for each test?
No—pre-loaded calibration curves are embedded per test strip lot number and validated against CRM reference materials (e.g., NIST SRM 2374); only annual performance verification using control strips is required.
Can the system store custom test methods developed in-house?
Yes—through the Admin Portal, users may import validated calibration files (.cal format) and associate them with proprietary strip barcodes.
Is offline operation supported without internet connectivity?
Yes—full assay execution, image capture, quantification, and local reporting function independently; cloud sync occurs automatically upon network reconnection.
What is the operating temperature range for field deployment?
The instrument is rated for continuous operation between 5 °C and 40 °C, with condensation-resistant housing tested per IP54 specifications.
How does the system ensure data integrity during power interruption?
All write operations use journaling file system (F2FS) with atomic commit; unsaved assay buffers are preserved in non-volatile RAM and recovered on reboot.