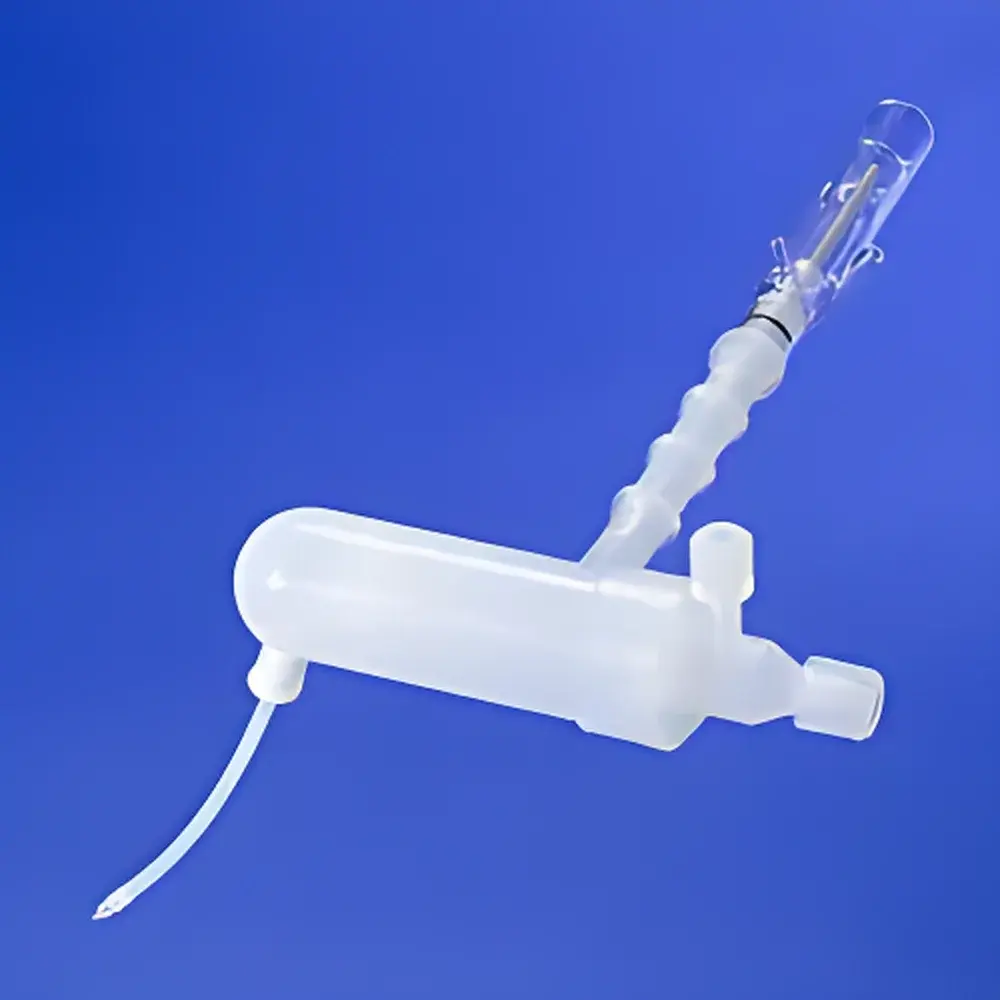
Savillex PFA Inert Sample Introduction Kit for Agilent 7500/7700/78x0x ICP-MS Systems
| Brand | Savillex |
|---|---|
| Origin | USA |
| Manufacturer Authorization | Authorized Distributor |
| Import Status | Imported |
| Model No. | 850-011-100635 |
| Pricing | Upon Request |
Overview
The Savillex PFA Inert Sample Introduction Kit (Model 850-011-100635) is a high-purity, chemically resistant consumable assembly engineered specifically for trace-level elemental analysis by inductively coupled plasma mass spectrometry (ICP-MS). Designed for seamless integration with Agilent 7500, 7700, and 78x0x series ICP-MS instruments, this kit replaces standard glass or polymer-based sample introduction components with perfluoroalkoxy alkane (PFA) materials to eliminate analyte adsorption, memory effects, and acid-induced contamination—critical concerns in ultra-trace quantification of elements such as As, Se, Hg, Tl, and rare earth elements in aggressive matrices. The system operates on a dual-channel configuration: one path accommodates a 6 mm OD nebulizer tip, while the second incorporates a dedicated gas makeup port to stabilize plasma conditions and optimize signal stability during high-solids or organic solvent introduction. All PFA components—including the spray chamber, end cap, connector, syringe, and torch support bracket—are pre-cleaned in sub-boiling HNO₃/HF solutions under Class 10 cleanroom conditions, ensuring baseline-level blank contributions (<0.1 pg/mL for most target elements).
Key Features
- Full-PFA construction eliminates metal leaching and surface adsorption, enabling reliable detection at sub-femtogram levels
- Dual-port design: integrated 6 mm outer diameter nebulizer interface + independent makeup gas inlet for plasma robustness
- Pre-cleaned in ultra-pure HNO₃/HF mixture (ASTM D5177-compliant cleaning protocol), validated via low-blank ICP-MS screening
- Direct compatibility with Agilent G3285A #102, G3285B #102, and G3285-80015 quartz torch configurations
- Thermal stability up to 260 °C supports elevated-temperature desolvation and high-boiling solvent applications
- Dimensionally matched to Agilent 7500/7700/78x0x front-end geometry—no tooling or alignment adjustments required
Sample Compatibility & Compliance
This kit maintains full compatibility with aqueous standards, hydrochloric, nitric, and hydrofluoric acid digests (up to 5% v/v HF), dilute organic solvents (e.g., methanol, ethanol), and complex environmental, geological, and biological extracts. It meets material requirements outlined in ISO/IEC 17025:2017 for trace metal analysis laboratories and supports compliance with EPA Method 1638, USP , and EU Pharmacopoeia Chapter 5.20 for elemental impurities testing. When used in conjunction with Agilent’s certified reference materials and instrument calibration protocols, the kit contributes to documented audit readiness for GLP and GMP environments requiring 21 CFR Part 11–compliant data integrity.
Software & Data Management
While the PFA kit itself contains no embedded electronics or firmware, its performance directly influences key acquisition parameters monitored and logged within Agilent MassHunter Software (v4.4 or later). Signal stability (RSD < 2% over 60 min), oxide ratio (CeO⁺/Ce⁺ < 1.5%), and doubly charged ion ratio (Ba²⁺/Ba⁺ < 2.0%) are routinely tracked during method validation and daily QC checks. MassHunter’s Audit Trail module records all user-initiated changes to acquisition methods involving this kit, satisfying FDA 21 CFR Part 11 requirements for electronic records and signatures when deployed in regulated pharmaceutical or clinical research settings.
Applications
- Ultra-trace analysis of halogens and semi-metals in semiconductor-grade reagents (SEMI F57)
- Isotopic ratio measurements (e.g., U/Pb, Sr/Nd) in geochronology labs requiring minimal procedural blanks
- Speciation studies coupling HPLC-ICP-MS where PFA tubing minimizes post-column adsorption
- High-throughput clinical metallomics using serum, urine, or CSF samples with HF-assisted digestion
- Regulatory testing of catalyst residues in active pharmaceutical ingredients (APIs) per ICH Q3D guidelines
FAQ
Is this kit compatible with Agilent 8800 or 8900 Triple Quadrupole ICP-MS systems?
No—this kit is mechanically and pneumatically optimized for the 7500/7700/78x0x series only; the 8800/8900 platforms require different spray chamber geometries and gas flow routing.
Can the PFA spray chamber be used with microflow nebulizers (e.g., 50 µL/min)?
Yes—its internal volume and flow dynamics support stable operation across 20–200 µL/min flow rates when paired with appropriate desolvation control.
Does Savillex provide certificate of analysis (CoA) with each kit?
Yes—each unit ships with a lot-specific CoA documenting blank levels for ≥12 elements (Al, Ca, Cr, Fe, Mg, Mn, Na, Ni, Pb, Si, Ti, Zn) measured by ICP-MS after 24-h acid leach.
How often should the PFA components be replaced?
Under routine use with ≤1% HF digestates, replacement is recommended every 6–12 months; frequency increases with >3% HF exposure or particulate-laden samples.
Is torch alignment affected when installing this kit?
No—the bracket design preserves original X/Y/Z positioning relative to the sampler cone; no realignment is necessary post-installation.