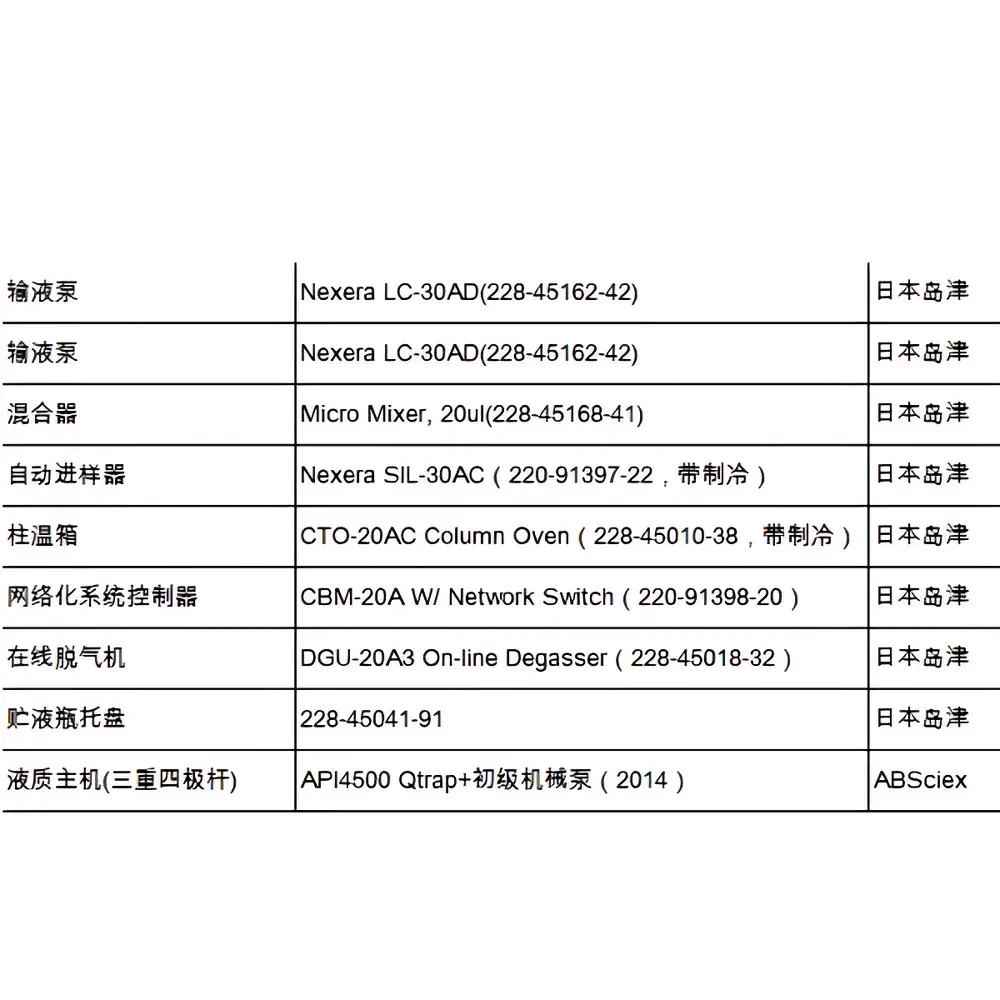
SCIEX QTRAP 4500 Hybrid Triple Quadrupole/Linear Ion Trap Mass Spectrometer with Shimadzu Nexera LC-30AD UHPLC System
| Brand | SCIEX |
|---|---|
| Origin | USA |
| Configuration | QTRAP 4500 MS/MS + Shimadzu Nexera LC-30AD UHPLC |
| Mass Analyzer Type | Hybrid Triple Quadrupole / Linear Ion Trap |
| Ion Sources | ESI & APCI |
| Mass Range | 10–10,000 Da |
| Resolution (FWHM) | Up to 10,000 |
| Detection Limit | Low-fg/mL (on-column, typical for peptide standards) |
| Scan Speed | Up to 10,000 Da/s |
| Acquisition Modes | Full Scan, SIM, MRM, Enhanced MS (EMS), Enhanced Product Ion (EPI), Information-Dependent Acquisition (IDA) |
| Software | AB SCIEX Analyst v1.7.2 or later |
| Compliance | Supports 21 CFR Part 11 audit trails (when configured with validated IT infrastructure and user access controls) |
Overview
The SCIEX QTRAP® 4500 system is a hybrid triple quadrupole / linear ion trap mass spectrometer engineered for high-sensitivity quantitative and qualitative analysis in regulated and discovery-driven laboratories. Coupled with the Shimadzu Nexera LC-30AD ultra-high-performance liquid chromatography platform, this refurbished integrated LC-MS/MS system delivers robust separation efficiency, precise gradient delivery (0.001–5.000 mL/min, ≤0.1% RSD), and consistent electrospray ionization (ESI) or atmospheric pressure chemical ionization (APCI) performance. The QTRAP architecture uniquely combines triple quadrupole quantification fidelity with linear ion trap capabilities—including enhanced MS (EMS), enhanced product ion (EPI), and information-dependent acquisition (IDA)—enabling simultaneous quantitation and structural elucidation within a single analytical run. This configuration is particularly suited for applications requiring trace-level detection, compound identification, and method transfer across QC, clinical research, and regulatory environments.
Key Features
- Hybrid mass analyzer: Triple quadrupole front end for MRM-based quantitation; linear ion trap for rapid, high-duty-cycle MS/MS scanning and library matching.
- Dual ionization capability: Switchable ESI and APCI sources mounted on a single source housing, supporting broad polarity coverage and thermally labile compound analysis.
- High-speed acquisition: Real-time scanning up to 10,000 Da/s with <10 ms dwell time per transition—critical for UHPLC peak fidelity and multi-analyte workflows.
- Dynamic range: >5 orders of magnitude linear dynamic range in MRM mode, validated per ASTM D7862 and ICH M10 guidelines for bioanalytical method validation.
- LC integration: Shimadzu Nexera LC-30AD features dual parallel pumps, low-dead-volume flow cell, and 40-position thermostatted autosampler (4–45 °C), ensuring retention time stability (<0.15% RSD) and carryover <0.005%.
- Hardware readiness: Refurbished unit includes full recalibration (mass axis, detector gain, ion optics), vacuum system integrity verification, and documented preventive maintenance history per SCIEX Service Bulletin SB-4500-02.
Sample Compatibility & Compliance
The system accommodates diverse sample matrices—including plasma, urine, tissue homogenates, environmental extracts, and food digests—without modification to standard source or interface hardware. It supports reversed-phase, HILIC, and ion-pairing chromatographic methods compliant with USP , EP 2.2.46, and ISO/IEC 17025:2017 requirements for testing laboratories. When deployed with validated Analyst software configurations (v1.7.2+), instrument control logs, audit trails, electronic signatures, and raw data archiving meet FDA 21 CFR Part 11 and EU Annex 11 expectations for GxP environments. All refurbishment procedures adhere to SCIEX’s Certified Pre-Owned Instrument Protocol, including component-level traceability, firmware version lock, and electrical safety testing (IEC 61010-1).
Software & Data Management
Data acquisition and processing are performed using AB SCIEX Analyst® software (licensed, version 1.7.2 or higher), which provides fully configurable acquisition methods, automated calibration routines, peak integration algorithms compliant with AAPS/USP recommendations, and customizable report templates. Raw data files (.wiff) are natively compatible with third-party platforms including Skyline, Compound Discoverer, and SimGlycan for retrospective analysis. Network deployment supports centralized license management, role-based access control, and integration with LIMS via ODBC or RESTful API adapters. Audit trail functionality records all parameter changes, acquisition start/stop events, and data export actions with timestamped user attribution.
Applications
This LC-QTRAP 4500 platform is routinely applied in:
- Pharmaceutical Development: Bioequivalence studies, ADME profiling, impurity identification, and forced degradation product characterization per ICH Q5A–Q5E.
- Clinical & Toxicology Research: Therapeutic drug monitoring (TDM), newborn screening metabolite panels, and forensic toxicant screening using spectral libraries (NIST, MassBank, METLIN).
- Environmental Analysis: EPA Method 1694–compliant analysis of pharmaceuticals and personal care products (PPCPs) in wastewater; PFAS quantitation using isotope dilution.
- Food Safety: Multi-residue pesticide screening (EU SANTE/11312/2021), veterinary drug confirmation (EC 2002/657), and mycotoxin quantification in cereals and dairy.
- Proteomics & Metabolomics: Label-free and TMT-labeled peptide quantitation; untargeted metabolite feature detection with IDA-triggered MS/MS.
FAQ
Is this a factory-refurbished or dealer-refurbished instrument?
This system has undergone certified pre-owned refurbishment by an AB SCIEX-authorized service partner, including full functional testing, ion optic cleaning, detector replacement if required, and documentation per SCIEX SOP-REFURB-4500.
What warranty and service support is included?
Standard offering includes 12-month comprehensive parts-and-labor warranty, remote diagnostics support, and priority access to on-site engineering assistance within 72 business hours in North America and EU regions.
Can the system be qualified for GLP or GMP use?
Yes—IQ/OQ documentation packages are available upon request; PQ execution requires site-specific protocol development aligned with your internal validation master plan and applicable regulatory guidance.
Are original consumables and spare parts still available?
All major consumables—including Turbo V™ ion sources, curtain gas orifices, and collision cell components—are actively stocked by SCIEX and authorized distributors through 2027.
Does the Shimadzu LC-30AD support 2D-LC coupling?
Yes—the system includes two independent LC modules and a 10-port/2-position switching valve, enabling comprehensive two-dimensional separations such as heart-cutting or comprehensive (LC×LC) configurations.