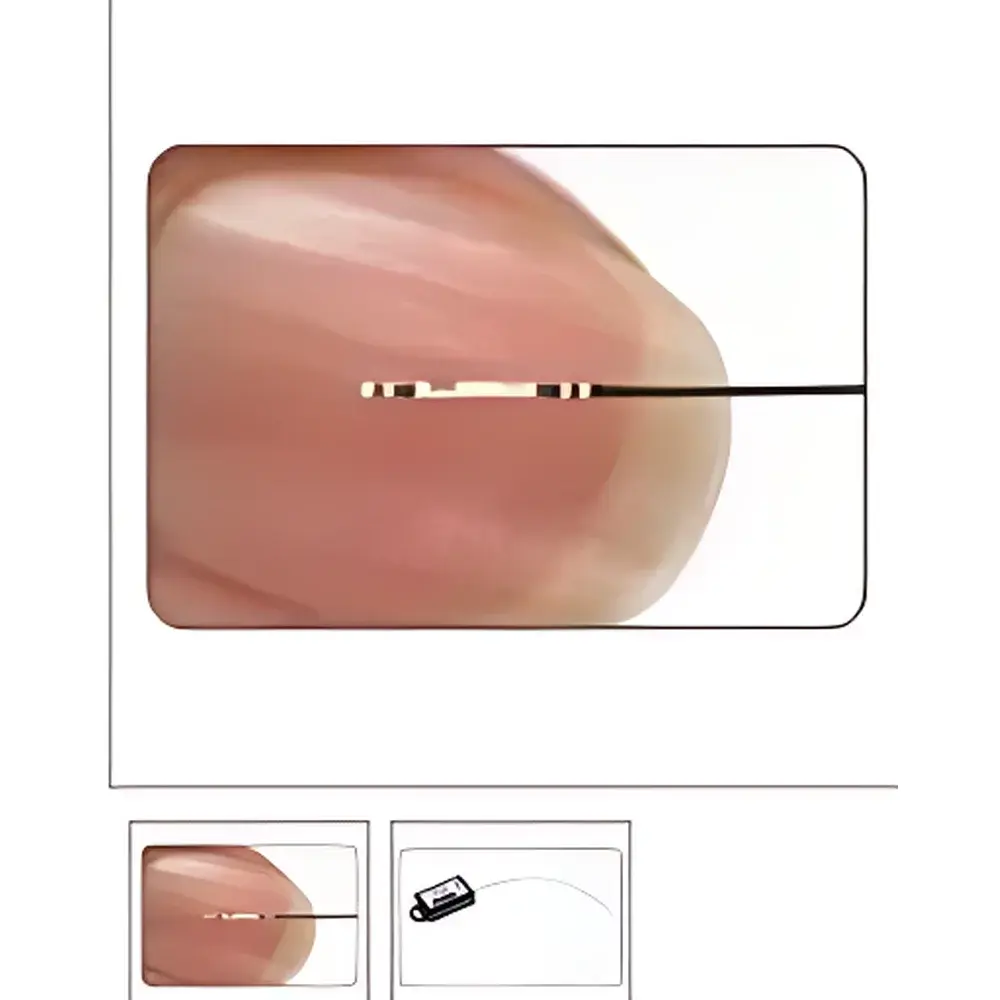
Scisense Ultra-Miniature Pressure-Volume Catheter
| Origin | USA |
|---|---|
| Manufacturer Type | Distributor |
| Origin Category | Imported |
| Model | Scisense! |
| Pricing | Upon Request |
Overview
The Scisense Ultra-Miniature Pressure-Volume (P-V) Catheter is a high-fidelity, chronically implantable intracardiac sensor system engineered for precise, simultaneous acquisition of left ventricular pressure and volume signals in small animal models—primarily transgenic mice and rats. Based on conductance catheter technology, it operates on the principle that intraventricular blood conductivity correlates linearly with chamber volume under controlled ionic conditions. The catheter integrates a miniaturized solid-state pressure transducer (0.39 mm diameter) and four precisely spaced platinum-iridium electrode rings (0.37 mm outer diameter, 0.25 mm width) along a single-axis, ultra-low-profile shaft. Its 1.2 French (0.4 mm OD) form factor enables atraumatic passage across the aortic valve without requiring pre-use flushing or air bubble removal—critical for minimizing procedural artifacts and hemodynamic perturbation during acute in vivo studies. Designed specifically for murine cardiovascular phenotyping, the system delivers real-time, beat-to-beat hemodynamic indices—including end-systolic elastance (Ees), preload recruitable stroke work (PRSW), dP/dtmax, tau (τ), and pressure-volume loop-derived contractility and compliance metrics—with minimal surgical interference and high inter-animal reproducibility.
Key Features
- Ultra-miniaturized 1.2F shaft diameter (0.4 mm OD) compatible with mouse and rat aortic valve crossing without balloon dilation or valvulotomy
- Integrated dual-modality sensing: high-stability piezoresistive pressure transducer (0.39 mm) co-located with four conductance electrodes (0.37 mm OD)
- Electrode configurations optimized per species: 4.5 mm center-to-center spacing for mice; 9 mm for rats; 0.5 mm spacing between outer and inner ring pairs
- No flush or de-airing required—enabling rapid deployment and reducing preparation time and operator variability
- Pressure sensor specifications: measurement range –50 to +300 mmHg (operating temperature 32–42°C); sensitivity ≥10 μV/V/mmHg; frequency response DC to >10 kHz; constant-current excitation; zero offset drift <6 mmHg/12 h (after 15-min saline equilibration)
- Signal conditioning unit provides analog output (±10 V full scale) compatible with third-party data acquisition systems (e.g., ADInstruments PowerLab, National Instruments DAQ, Spike2, LabChart)
Sample Compatibility & Compliance
The Scisense P-V catheter is validated for use in terminal and chronic preparations in C57BL/6, FVB/N, and other common inbred and transgenic mouse strains, as well as Sprague-Dawley and Wistar rats. Its biocompatible polyurethane shaft and platinum-iridium electrodes meet ISO 10993-5 (cytotoxicity) and ISO 10993-10 (sensitization and irritation) requirements for short-term (<24 h) and extended-duration (up to 7-day) implantation. While not FDA-cleared for human use, the system supports GLP-compliant preclinical cardiovascular safety pharmacology studies per ICH S7B and ICH E14 guidelines. Data acquisition workflows are compatible with audit-trail-enabled platforms supporting 21 CFR Part 11 compliance when paired with validated software environments.
Software & Data Management
The catheter interfaces exclusively via analog voltage output; no proprietary software is bundled. Users retain full control over signal processing, calibration (parallel conductance correction, β-coefficient determination, specific resistance [ρ] adjustment), and loop analysis using their institution’s established physiological computing environment. Standard calibration procedures follow the method of Baan et al. (1984) and subsequent refinements by Kass et al. Raw pressure and conductance signals are sampled synchronously at ≥1 kHz to preserve high-frequency components essential for accurate dP/dt derivation and diastolic stiffness assessment. Exported datasets conform to HDF5 or MATLAB .mat formats, enabling integration into custom MATLAB, Python (NumPy/SciPy), or R-based analysis pipelines for longitudinal P-V loop quantification, machine learning–enabled phenotype clustering, or statistical modeling of genotype–function relationships.
Applications
- High-resolution cardiac phenotyping of genetically modified mouse models (e.g., knockouts, knockins, Cre-lox conditional lines)
- Evaluation of acute drug effects on ventricular contractility, lusitropy, and afterload sensitivity
- Assessment of myocardial remodeling post-myocardial infarction or pressure/volume overload
- Validation of non-invasive imaging modalities (e.g., echocardiography, MRI) against gold-standard invasive metrics
- Teaching and training in advanced cardiovascular physiology laboratories
- Supporting IND-enabling safety pharmacology studies under regulatory guidance (ICH, FDA, EMA)
FAQ
Is the Scisense P-V catheter suitable for chronic implantation beyond 24 hours?
Yes—validated for up to 7 days in rodent models using sterile surgical techniques and appropriate postoperative analgesia and monitoring protocols.
Does the system require a dedicated amplifier or signal conditioner?
Yes—the Scisense Pressure Control Unit (PCU) is mandatory for transducer excitation, signal amplification, and analog output conversion; it is not compatible with generic bridge amplifiers.
Can the same catheter be used interchangeably in mice and rats?
No—electrode spacing is species-specific; separate mouse-optimized (4.5 mm) and rat-optimized (9 mm) catheters are supplied to ensure accurate volume calibration and spatial resolution.
How is parallel conductance determined during experiments?
Via hypertonic saline bolus injection (typically 10–20 µL of 15% NaCl) followed by offline correction using standard conductance catheter algorithms.
What is the recommended sampling rate for optimal P-V loop fidelity?
A minimum of 1 kHz is required; 2–5 kHz is recommended to resolve rapid pressure upstrokes and early diastolic relaxation phases without aliasing.