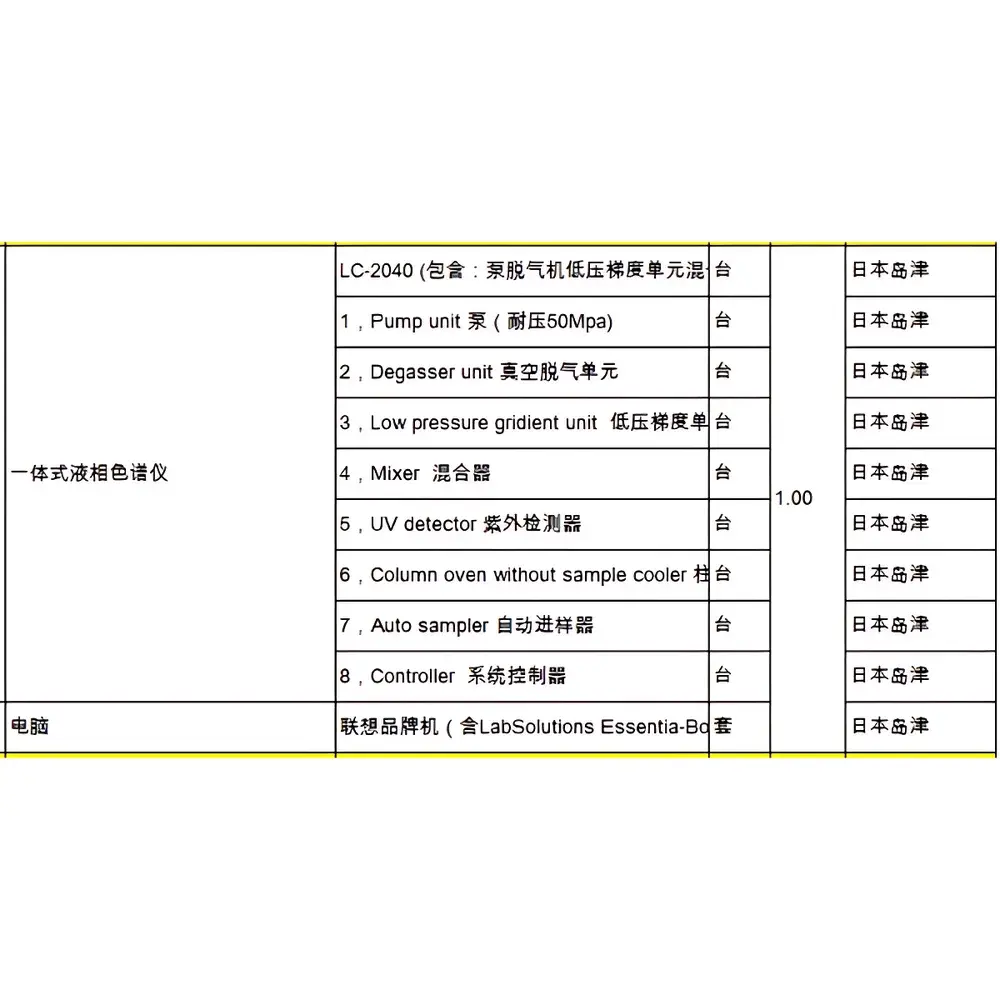
Shimadzu LC-2040 Nexera-i Integrated UHPLC System (Refurbished)
| Brand | Shimadzu |
|---|---|
| Origin | Japan |
| Model | LC-2040 |
| Maximum System Pressure | 66 MPa (9,500 psi) |
| Column Compatibility | Sub-2.5 µm and Core-Shell Packings |
| Detector | UV-Vis (Standard), Optional PDA or Fluorescence |
| Autosampler Injection Volume Range | 0.1–100 µL |
| Carryover | ≤0.0025% |
| Temperature Control | Dual-zone (Optics & Flow Cell) |
| Data System | LabSolutions v5.9+ with ICM (Interactive Communication Mode) |
| Compliance | FDA 21 CFR Part 11 Ready (Audit Trail, Electronic Signatures), GLP/GMP Support Enabled |
| Warranty | 12 Months Comprehensive Coverage |
Overview
The Shimadzu LC-2040 Nexera-i is a factory-refurbished, fully validated integrated ultra-high-performance liquid chromatography (UHPLC) system engineered for precision, reproducibility, and operational continuity in regulated and research-intensive laboratories. Designed as the high-pressure evolution of the Prominence-i platform, the LC-2040 operates at up to 66 MPa (9,500 psi), enabling robust use of sub-2.5 µm fully porous and core-shell particle columns without requiring 100 MPa-class instrumentation. Its measurement principle relies on high-pressure gradient elution coupled with real-time UV-Vis absorbance detection (190–700 nm), delivering high-resolution separations with exceptional baseline stability—achieved via dual-zone thermal regulation of both optical components and flow cell. This architecture reduces baseline drift to <1/20th that of legacy Alliance-class systems, even under ambient temperature fluctuations or low-wavelength detection (e.g., 210 nm). As a pre-configured, benchtop-integrated system, the LC-2040 eliminates inter-unit cabling complexity and minimizes footprint while maintaining full compliance with ISO/IEC 17025 method validation requirements and ICH Q2(R2) guidelines for analytical procedure verification.
Key Features
- Integrated UHPLC architecture with embedded quaternary pump, autosampler, column oven, and UV-Vis detector—no external controllers or PC dependency required for basic operation
- 7-inch full-color capacitive touchscreen interface supporting real-time chromatogram display, method editing, and batch table creation directly on instrument panel
- Interactive Communication Mode (ICM): Enables seamless bidirectional synchronization between instrument control panel and LabSolutions software—methods and batches created on-device auto-upload to workstation for execution
- Dual-zone temperature control system stabilizes optical path and flow cell independently, ensuring <0.001 mAU/h baseline drift under continuous operation at 210 nm
- Ultra-low carryover (<0.0025%) validated across polarity gradients—from formic acid/water to acetonitrile/methanol—enabling trace-level analysis of high-potency compounds without rinsing artifacts
- Intelligent power management: 95% reduction in standby power consumption versus prior-generation Shimadzu HPLC systems; automatic shutdown post-analysis with environmental reporting
- Pre-installed LabSolutions CS v5.9+ with full 21 CFR Part 11 functionality—including electronic signatures, audit trail, role-based access control, and instrument qualification documentation templates
Sample Compatibility & Compliance
The LC-2040 supports broad sample matrix compatibility—from aqueous biological extracts and plasma supernatants to organic-soluble small molecules and synthetic polymers. Its 0.1–100 µL injection range accommodates both micro-volume pharmacokinetic samples (e.g., DMPK studies) and macro-volume QC injections without dilution. The system meets ASTM D8073 (UHPLC method transfer), ISO 17025:2017 clause 7.2.2 (method validation), and USP chromatography requirements. All refurbished units undergo full IQ/OQ/PQ protocol execution per Shimadzu’s Global Refurbishment Standard (GRS-2023), including pressure decay testing, gradient accuracy verification (±0.2% RSD), retention time repeatability (<0.15% RSD over 10 injections), and system suitability test (SST) pass criteria per ICH Q2(R2). Each unit ships with a Certificate of Refurbishment, calibration records, and a complete set of consumables for first-run validation.
Software & Data Management
LabSolutions CS serves as the native data acquisition and processing environment, configured with pre-loaded ICH-compliant report templates (e.g., SST Summary, Peak Purity, Resolution Matrix). The system supports automated workflow orchestration: from instrument startup and consumable status check (via built-in sensor network), through mobile-phase priming, baseline assessment, SST execution, and final report generation—including PDF export with embedded digital signatures. Audit trail logs capture all user actions, parameter changes, and data modifications with immutable timestamps and operator IDs. Raw data files (.lcd format) are stored in hierarchical folder structures compliant with ALCOA+ principles (Attributable, Legible, Contemporaneous, Original, Accurate, Complete, Consistent, Enduring, Available). Remote monitoring via Shimadzu CloudLink allows secure, browser-based access to real-time chromatograms and system health metrics using iOS/Android devices—without exposing internal lab networks.
Applications
The LC-2040 is routinely deployed in pharmaceutical quality control labs for release testing of APIs and finished dosage forms per USP monographs; in CMC development for forced degradation studies, impurity profiling, and stability-indicating method development; and in academic and contract research settings for metabolomics, natural product isolation, and polymer molecular weight distribution analysis. Its pressure capability and low dispersion volume make it ideal for coupling with triple-quadrupole and high-resolution mass spectrometers (e.g., LC-MS/MS for bioanalysis). In environmental labs, it supports EPA Method 8330B (PAHs) and 8082A (PCBs) with enhanced sensitivity and throughput. The system’s reliability enables unattended overnight runs across 384-well plate formats—reducing total cycle time by 47 minutes compared to LC-2010-based workflows.
FAQ
Is this a genuine Shimadzu factory-refurbished unit, and what does the refurbishment process include?
Yes. Each LC-2040 undergoes Shimadzu’s certified Global Refurbishment Standard (GRS-2023), including full mechanical inspection, pump seal replacement, detector lamp recalibration, autosampler needle wear assessment, firmware update to latest stable release, and comprehensive performance qualification against 21+ critical parameters.
Does the 1-year warranty cover parts, labor, and on-site service?
Yes—the warranty includes all components, labor, and unlimited remote technical support. On-site service is provided within 72 business hours in North America, EU, and APAC regions covered by Shimadzu’s authorized service network.
Can this system be validated for GxP environments?
Absolutely. The unit ships with full IQ/OQ documentation packages, 21 CFR Part 11 configuration files, and pre-validated LabSolutions CS installation media—all aligned with FDA, EMA, and PMDA expectations for computerized system validation.
What consumables and accessories are included with the system?
Standard delivery includes one set of pump seals, two UV lamps, one autosampler syringe, column oven insulation kit, and a complete starter pack of HPLC-grade solvents and certified reference standards for initial system qualification.
Is LabSolutions software included, and is training provided?
Yes—full perpetual license for LabSolutions CS v5.9+ is included. Complimentary virtual instructor-led training (8 hours) covers method setup, data review, audit trail management, and routine maintenance procedures.