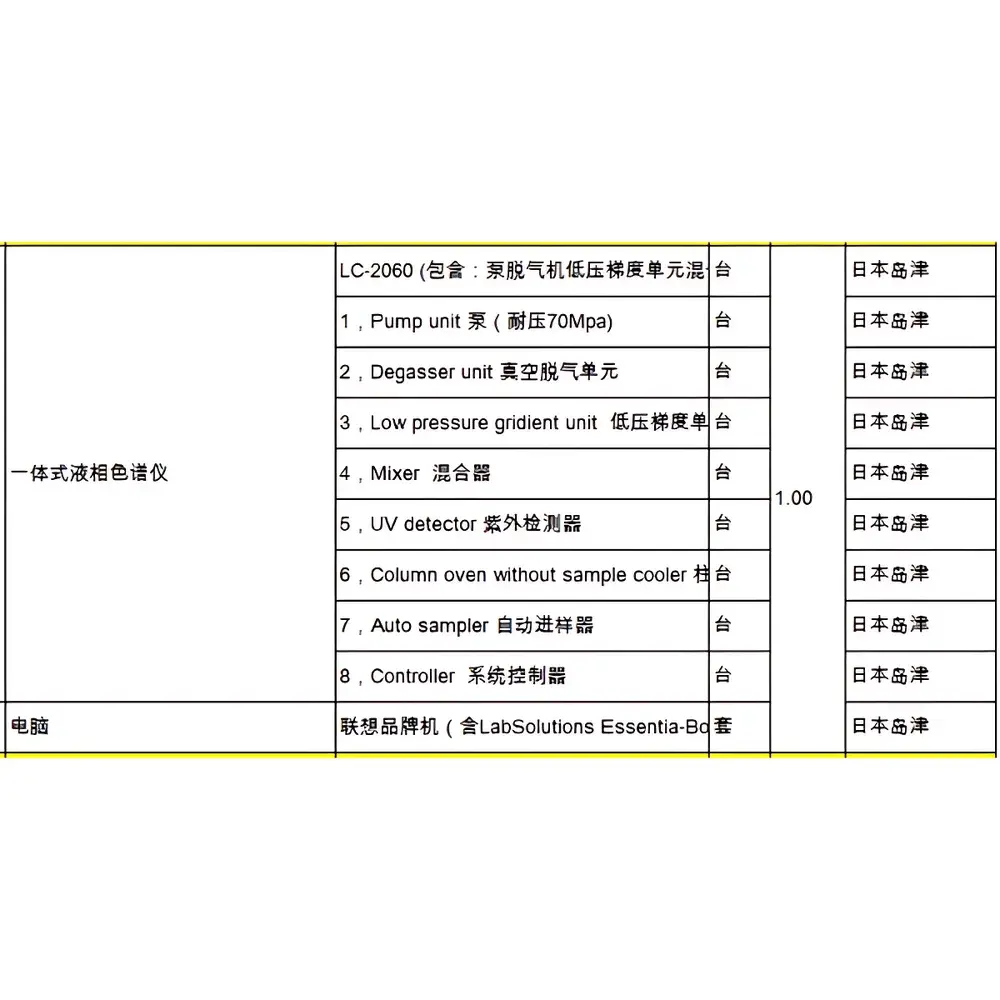
Shimadzu LC-2060 i-Series Integrated Liquid Chromatograph (Refurbished)
| Brand | Shimadzu |
|---|---|
| Origin | Japan |
| Model | LC-2060 |
| Configuration | Integrated HPLC System |
| Warranty | 1 Year |
| Category | Chromatography Instruments |
| Condition | Refurbished & Fully Qualified |
Overview
The Shimadzu LC-2060 i-Series Integrated Liquid Chromatograph is a factory-refurbished, fully validated high-performance liquid chromatography (HPLC) system engineered for precision, reproducibility, and operational continuity in regulated and research laboratory environments. As part of Shimadzu’s i-Series platform, the LC-2060 implements Analytical Intelligence (AI)—a deterministic, rules-based automation architecture—not as marketing terminology, but as an integrated suite of instrument-level control algorithms and software-defined workflows. Its core measurement principle relies on high-pressure gradient elution coupled with UV-Vis absorbance detection (standard PDA or single-wavelength configurations), enabling robust separation, identification, and quantification of small-molecule analytes across pharmaceutical, environmental, food safety, and academic applications. Unlike modular HPLC systems requiring external controllers or third-party data systems, the LC-2060 integrates pump, autosampler, column oven, and detector into a single chassis with unified firmware, minimizing dwell volume variability and inter-unit communication latency—critical factors in method transfer fidelity and retention time stability.
Key Features
- Integrated Analytical Intelligence (AI) Architecture: Embedded firmware modules—including FlowPilot (intelligent flow-rate ramping), Mobile Phase Pilot (real-time solvent level monitoring and predictive low-volume alerts), and LabSolutions™ Direct (browser-based remote instrument control)—enable unattended start-to-finish analysis without manual intervention.
- Automated System Suitability & Validation: Built-in Auto-Validation function executes pre-run checks per ICH Q2(R2) and USP guidelines—including pump flow accuracy, gradient composition verification, wavelength calibration, baseline noise/drift assessment, and photometric linearity—generating timestamped, audit-trail-enabled reports compliant with FDA 21 CFR Part 11 requirements.
- Dual Thermal Control System: TC-Optics technology actively stabilizes both the optical bench and flow cell temperature independently, reducing baseline drift by >70% under ambient fluctuations (±5 °C), thereby enhancing signal-to-noise ratio and improving LOQ for trace-level quantitation.
- Ultra-Low Carryover Design: Optimized fluidic pathways, inert wetted materials (e.g., titanium, sapphire, and PEEK-lined components), and programmable needle wash protocols achieve <0.005% cross-contamination—validated per ASTM D7943—ensuring integrity in high-throughput bioanalytical or impurity profiling workflows.
- i-PeakFinder & i-PDeA II Algorithms: These embedded chemometric tools apply multivariate curve resolution–alternating least squares (MCR-ALS) and adaptive baseline modeling to resolve co-eluting peaks, correct for baseline roll, and validate peak purity—eliminating subjective integration decisions and supporting GLP/GMP-compliant data review.
Sample Compatibility & Compliance
The LC-2060 accommodates standard 3–5 µm reversed-phase columns (up to 250 mm × 4.6 mm), HILIC, ion-exchange, and size-exclusion formats. It supports aqueous/organic mobile phases (including TFA, formic acid, ammonium acetate buffers), operates at pressures up to 66 MPa (9500 psi), and maintains gradient accuracy ±0.2% RSD over 0.1–100% B. The system complies with ISO/IEC 17025:2017 for testing laboratories, meets essential requirements of the EU Machinery Directive 2006/42/EC, and incorporates electronic signatures, audit trails, and role-based access controls aligned with FDA 21 CFR Part 11 and EU Annex 11 expectations. All refurbished units undergo full IQ/OQ protocol execution per Shimadzu’s internal standards, including leak testing, pressure decay validation, detector linearity verification (NIST-traceable standards), and chromatographic performance qualification using caffeine/acetaminophen test mixtures.
Software & Data Management
Controlled exclusively via Shimadzu LabSolutions CS v5.9x or later, the LC-2060 supports centralized deployment across LAN/WAN networks. LabSolutions CS provides secure, multi-user database management with hierarchical permissions (Administrator, Analyst, Reviewer), electronic batch records, and automated backup to network-attached storage (NAS) or cloud repositories (AWS S3, Azure Blob). Raw data (.lcd files) are stored in vendor-neutral format with embedded metadata (method parameters, instrument logs, calibration certificates). ACTO (Analytical Condition Transfer and Optimization) enables seamless migration of legacy methods from Shimadzu LC-2010, LC-2030, or competitive platforms (Waters, Agilent) by dynamically compensating for system volume differences and recalculating gradient delay times—reducing method revalidation effort by ≥60%. All software modules are licensed for perpetual use post-purchase.
Applications
The LC-2060 delivers validated performance in QC release testing (USP/EP monographs), stability-indicating assays, residual solvent analysis (ICH Q3C), pesticide residue screening (AOAC 2012.01), and metabolite profiling in non-clinical studies. Its low carryover and thermal stability make it suitable for peptide mapping, oligonucleotide purity assessment, and chiral separations requiring extended run times. Academic users leverage its remote operation capability for shared instrumentation cores, while contract research organizations deploy it in multi-client environments where method portability and data integrity are contractual obligations.
FAQ
Is this unit covered by Shimadzu’s original manufacturer warranty?
No. This is a professionally refurbished unit backed by a 12-month limited warranty covering parts and labor, administered by our ISO 9001-certified service center.
Does the system include current LabSolutions software licenses?
Yes. Each LC-2060 ships with a perpetual license for LabSolutions CS v5.9x and all AI-enabled modules (i-PeakFinder, FlowPilot, Mobile Phase Pilot).
Can I integrate this instrument into my existing LIMS or ELN?
Yes. LabSolutions CS supports ASTM E1384-compliant ASCII export, direct ODBC connectivity, and REST API endpoints for bidirectional data exchange with major LIMS vendors (Thermo SampleManager, LabVantage, STARLIMS).
What documentation is provided upon delivery?
You receive a complete refurbishment dossier: IQ/OQ reports, calibration certificates (NIST-traceable), maintenance log, firmware version record, and signed Certificate of Conformance.
Is method transfer support included?
Yes. Our application scientists provide one complimentary method transfer consultation—including ACTO parameter optimization and comparative chromatogram overlay—to ensure analytical equivalence between your legacy system and the LC-2060.