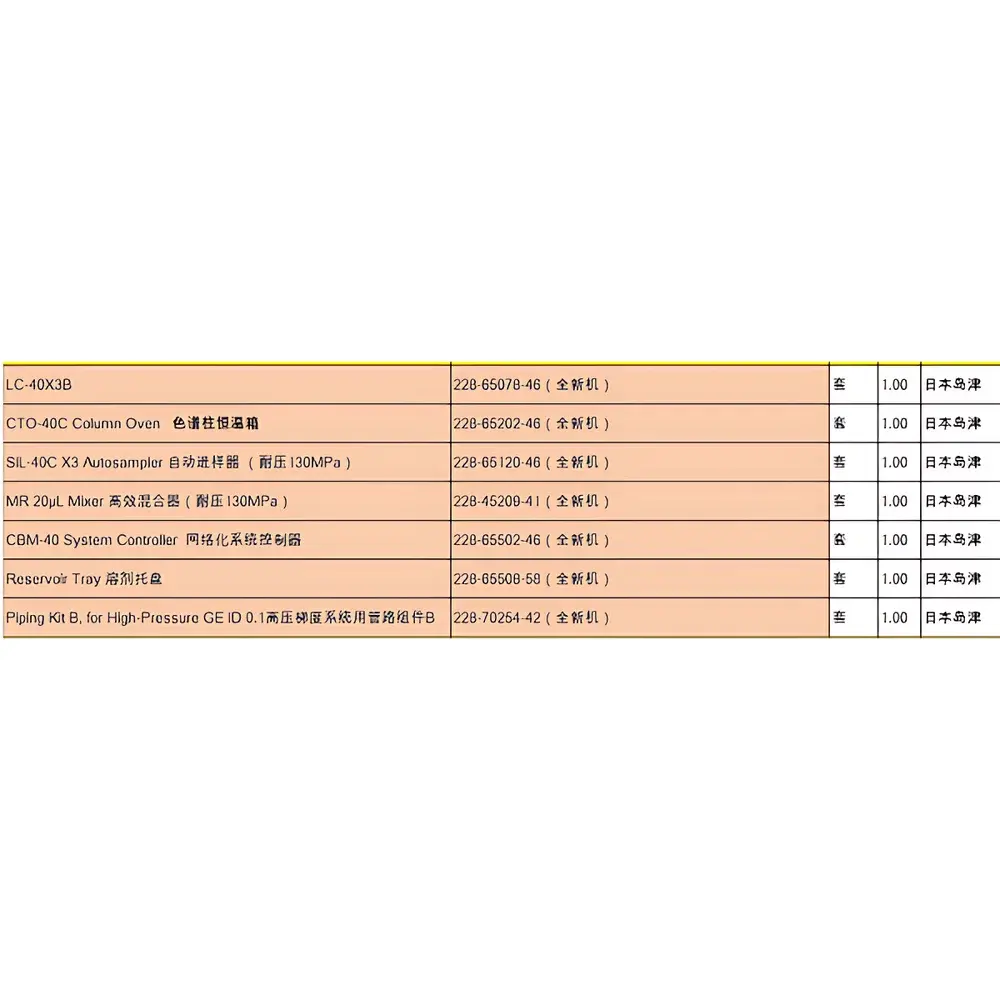
Shimadzu Nexera LC-40X3B Liquid Chromatography System (Used, Refurbished, Japan-Origin)
| Brand | Shimadzu |
|---|---|
| Origin | Japan |
| Model | LC-40X3B |
| Category | Liquid Chromatograph (HPLC/UHPLC) |
| Configuration | LC Front-End for LC-MS Coupling |
| Warranty | 12 months |
| Compliance | Designed for ISO/IEC 17025, GLP, and FDA 21 CFR Part 11–ready workflows |
| Software Platform | LabSolutions LCMS v5.1 or later |
| Power Consumption (Standby) | ≤68% reduction vs. prior generation |
| Auto-sampler Injection Speed | ≤5 sec per injection |
| Carryover | <0.001% (typical, validated with caffeine/benzodiazepine test mixtures) |
| Sample Capacity | Up to 44 × 96-well MTP plates via optional Plate Changer |
| Dual-Independent Flow Path Support | Yes (via SIL-40CX dual-inlet configuration) |
| Temperature Control | Active airflow management in sample compartment (±0.5 °C stability, condensation-suppressed) |
Overview
The Shimadzu Nexera LC-40X3B is a high-performance, modular liquid chromatography front-end system engineered for robust, high-throughput coupling with mass spectrometry platforms—including triple quadrupole (LC-MS/MS), Q-TOF, and ion trap instruments. As a refurbished and fully requalified unit sourced directly from Japan, this LC-40X3B configuration retains full hardware compatibility with Shimadzu’s latest LabSolutions LCMS software suite and meets original factory specifications for gradient accuracy (±0.2% RSD), flow precision (<0.1% RSD at 0.2–2.0 mL/min), and dwell volume consistency (<100 µL). Its architecture follows the Nexera design philosophy: ultra-low dispersion fluidics, thermally stabilized column ovens (ambient to 85 °C, ±0.1 °C), and pressure-rated operation up to 130 MPa—enabling both conventional HPLC and UHPLC method transfer without hardware modification. The system is optimized for regulated environments where trace-level quantitation, method reproducibility, and audit readiness are mandatory—particularly in bioanalytical laboratories performing PK/PD studies, clinical toxicology, and small-molecule impurity profiling.
Key Features
- Ultra-fast autosampling: SIL-40 series injector achieves ≤5-second cycle time with <0.001% carryover (validated per USP and ASTM D7260-19 protocols), critical for high-sensitivity LC-MS/MS applications.
- Intelligent sample compartment climate control: “Chamber Airflow Manager” dynamically regulates internal airflow to maintain stable temperature (±0.5 °C) and eliminate condensation—even during ambient humidity fluctuations or cold-sample injections.
- Dual independent flow path capability: Optional SIL-40CX module enables concurrent analysis of two distinct sample sets under separate gradient methods—ideal for parallel amino acid/organic acid profiling or vitamin panel screening without system reconfiguration.
- Energy-optimized operation: Standby power draw reduced by >68% versus LC-20 series; meets ENERGY STAR® criteria for laboratory instrumentation and supports LEED-certified facility compliance.
- Real-time interface synchronization: 10.1-inch capacitive touchscreen display mirrors LabSolutions LCMS workstation status—including pump pressure, column oven temperature, detector signal, and sequence progress—with touch-initiated pause/resume and emergency stop.
Sample Compatibility & Compliance
The LC-40X3B accommodates standard 1.5–2.0 mL vials, 96- and 384-well microtiter plates (MTP), and custom plate formats via Shimadzu’s certified Plate Changer modules. All wetted parts—including pump heads, check valves, and injector rotor seals—are chemically resistant to aqueous, organic, and acidic mobile phases (pH 1.0–12.0). System validation documentation includes IQ/OQ templates aligned with ISO/IEC 17025:2017 and GxP requirements. Full 21 CFR Part 11 compliance is achievable through LabSolutions LCMS audit trail configuration, electronic signature enforcement, and role-based user access controls—supporting inspection readiness for FDA, EMA, and PMDA audits.
Software & Data Management
Controlled exclusively via LabSolutions LCMS v5.1 or later, the LC-40X3B integrates seamlessly into enterprise LIMS environments through ASTM E1384-compliant data export (CSV, CDF, ANDI-MS). Method development tools include automatic dwell volume compensation, real-time peak tracking, and retention time prediction algorithms trained on >10,000 reference compounds. Raw data files are stored in vendor-neutral .lcd format with embedded metadata (instrument ID, operator, calibration timestamp, environmental logs). Audit trails record all parameter changes, sequence edits, and user logins with immutable timestamps—retained for ≥36 months per default policy.
Applications
- Bioanalytical quantitation: Plasma, urine, and tissue homogenate analysis for drug metabolites and endogenous biomarkers (e.g., cortisol, testosterone, vitamin D3) at sub-pg/mL levels.
- Pharmaceutical QC/QA: Forced degradation studies, residual solvent testing (ICH Q3C), and genotoxic impurity screening per ICH M7 guidelines.
- Clinical diagnostics: High-volume therapeutic drug monitoring (TDM) for immunosuppressants (tacrolimus, cyclosporine) and antiepileptics (valproic acid, lamotrigine).
- Food & environmental safety: Mycotoxin detection (aflatoxin B1, ochratoxin A), pesticide residue screening (multi-residue QuEChERS workflows), and PFAS analysis in drinking water.
FAQ
Is this unit eligible for Shimadzu factory warranty or service contract renewal?
Yes—refurbished LC-40X3B systems qualify for Shimadzu’s Extended Service Agreement (ESA) upon successful completion of post-refurbishment performance verification (PV) report.
Can the system be upgraded to support 2D-LC or heart-cutting configurations?
Yes—the LC-40X3B chassis supports optional 2D-LC modules (CBM-40 controller + dual pumps) and valve-driven heart-cutting via the VP-40 switching valve unit.
Does the quoted price include installation, qualification, and method transfer support?
The base price covers system hardware, pre-delivery operational testing, and factory calibration certificate. Installation, IQ/OQ, and application-specific method transfer services are available as billable add-ons.
What documentation is provided with the instrument?
You receive full refurbishment report, updated electrical safety test (EST) certificate, pressure test records, chromatographic performance verification (CPV) data, and LabSolutions license key for perpetual use.