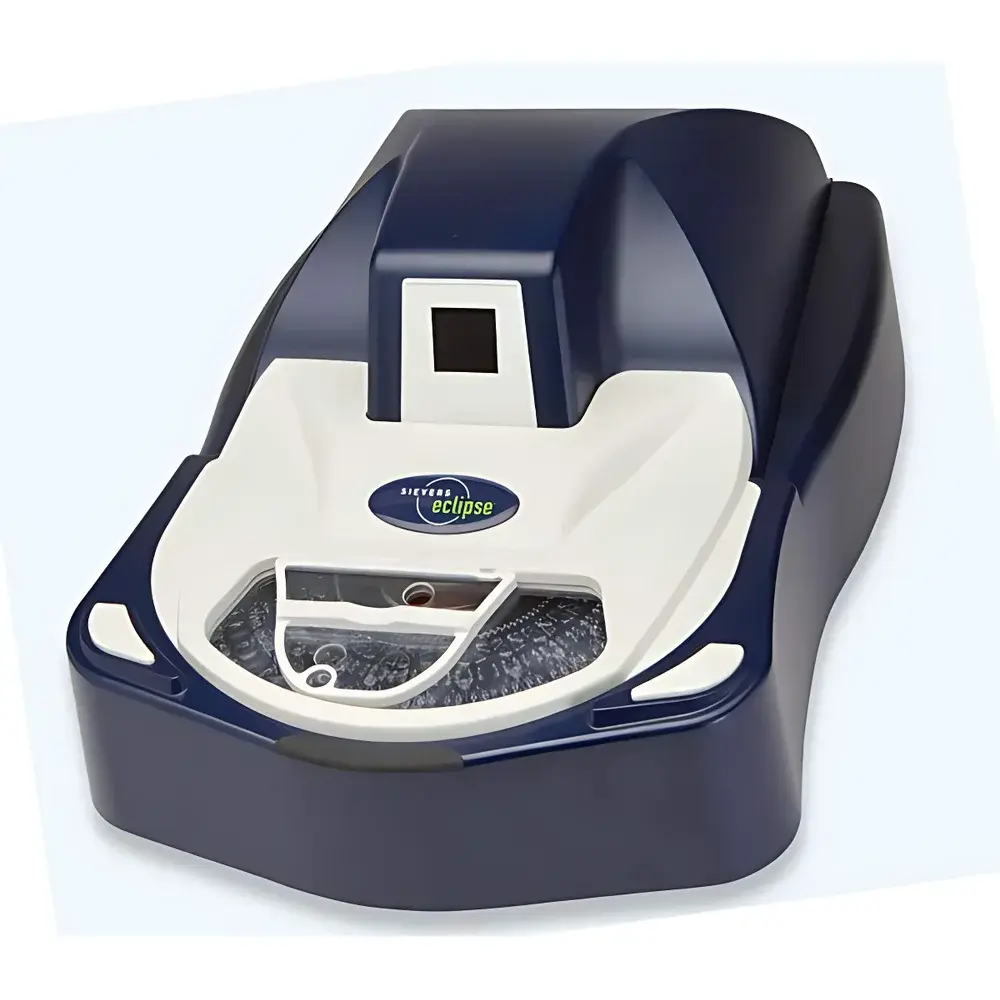
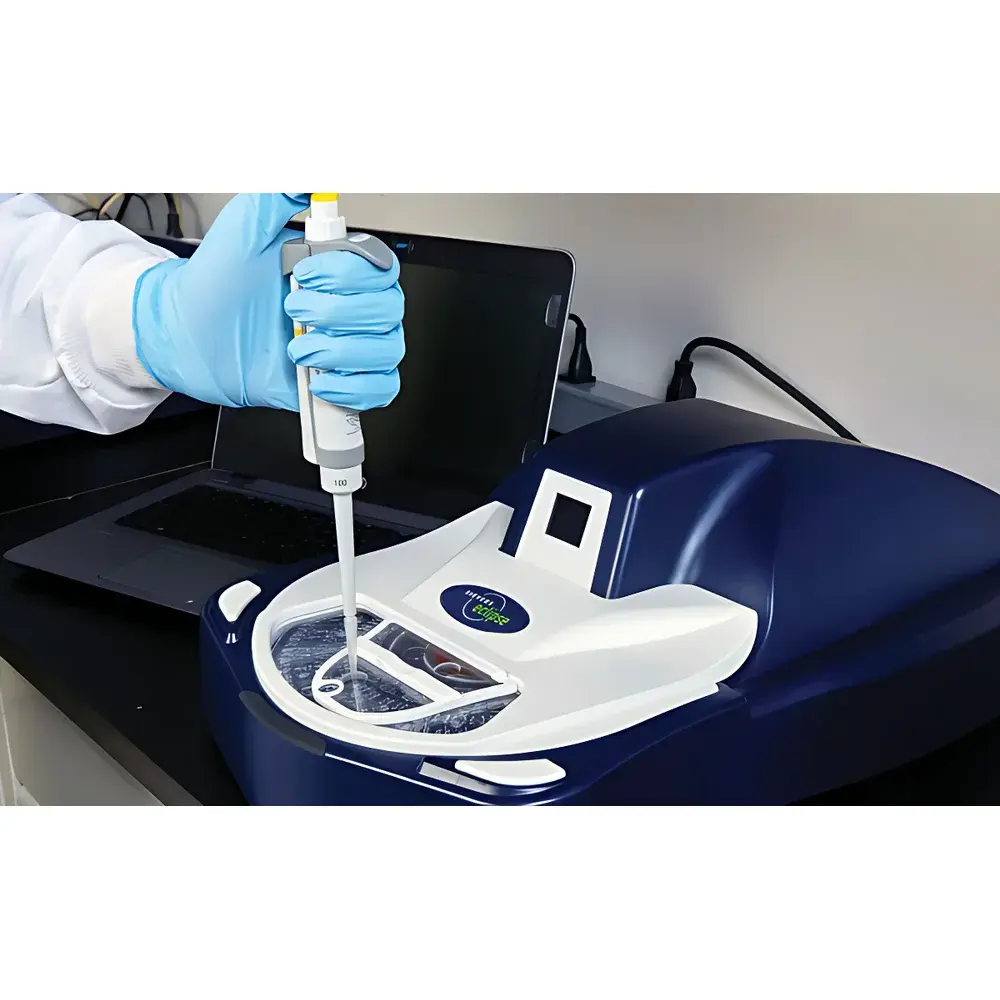
Sievers Eclipse Bacterial Endotoxin Testing System
| Brand | Sievers (Veolia) |
|---|---|
| Origin | USA |
| Manufacturer Status | Authorized Distributor |
| Import Status | Imported |
| Model | Sievers Eclipse |
| Pricing | Available Upon Request |
Overview
The Sievers Eclipse Bacterial Endotoxin Testing System is a fully automated, pharmacopeia-compliant instrument engineered for quantitative detection of bacterial endotoxins in water, parenteral drugs, and medical device rinsates. It employs the kinetic chromogenic assay method, a well-established LAL (Limulus Amebocyte Lysate) technique that measures the rate of color development (absorbance at 405 nm) proportional to endotoxin concentration. Unlike endpoint assays, the kinetic approach delivers superior precision and dynamic range by monitoring enzymatic reaction kinetics in real time under tightly controlled thermal conditions (37 ± 0.5 °C). Designed specifically for regulated pharmaceutical and biotechnology environments, the Eclipse platform meets the stringent analytical requirements of global compendial standards—including USP <85>, EP 2.6.14, ChP IV <1143>, and JP 4.01—while reducing reliance on horseshoe crab-derived reagents through optimized reagent consumption and high assay efficiency.
Key Features
- Regulatory-Ready Automation: Integrated sample handling supports up to 21 samples per run (including duplicates and positive product controls), minimizing manual intervention and operator-induced variability.
- Kinetic Chromogenic Detection: Real-time absorbance measurement at 405 nm using a stable LED light source and precision optical filters; optical linearity R ≥ 0.980 ensures reliable quantitation across the full dynamic range.
- Tight Thermal Control: On-board incubation block maintains reaction temperature at 37 ± 0.5 °C—critical for reproducible enzymatic kinetics and compliance with pharmacopeial temperature specifications.
- Robust Optical Architecture: Dual-wavelength capability includes a 1450 nm emitter for fluid path integrity monitoring, enabling automatic detection of air bubbles, insufficient volume, or pipetting errors prior to assay initiation.
- Validated Performance: Achieves ≤15% coefficient of variation (CV) for precision and accuracy within 50–200% of true value—fully aligned with USP <85> validation criteria for quantitative LAL methods.
- Extended Calibration Stability: System calibration remains valid for up to 12 months when operated under specified environmental conditions (ambient temperature 17–30 °C, humidity-controlled laboratory).
Sample Compatibility & Compliance
The Eclipse system is validated for aqueous matrices commonly encountered in pharmaceutical quality control: purified water, water for injection (WFI), saline solutions, buffer formulations, and extractables from dialysis equipment and Class III medical devices. All sample introduction is performed via calibrated electronic pipettes—no direct syringe or vial contact required. The platform supports full traceability for GLP and GMP environments, including audit-ready electronic records, user access controls, and secure data storage. Its kinetic chromogenic method satisfies FDA expectations for assay robustness and is compatible with 21 CFR Part 11-compliant software configurations when deployed with validated LIMS or ELN integrations.
Software & Data Management
Instrument operation and data analysis are managed through the Sievers Eclipse Software Suite—a Windows-based application supporting protocol configuration, run scheduling, real-time kinetic curve visualization, and automated report generation. Each assay generates a complete digital record containing raw absorbance values (recorded every 5 seconds), temperature logs, calibration history, user ID, timestamped actions, and pass/fail status against pre-defined acceptance criteria. Data export formats include CSV and PDF, facilitating integration into enterprise quality systems. The software architecture supports multi-level user permissions, electronic signatures, and configurable audit trails—enabling alignment with ICH E6(R3), Annex 11, and ISO/IEC 17025 documentation requirements.
Applications
- Endotoxin testing of Water for Injection (WFI) and Purified Water in pharmaceutical manufacturing suites
- Batch release testing of sterile injectables, ophthalmic solutions, and lyophilized biologics
- Validation and routine monitoring of depyrogenation tunnels and autoclave cycles
- Extractable testing of hemodialysis membranes, catheters, and implantable device components
- Environmental monitoring of cleanroom rinse waters and HVAC system condensates
- Supporting comparability studies during process changes or facility transfers
FAQ
What pharmacopeial methods does the Eclipse system support?
It fully complies with USP <85>, EP 2.6.14, ChP IV <1143>, and JP 4.01 for kinetic chromogenic endotoxin testing.
Can the Eclipse system be used for both water and product testing?
Yes—it is validated for low-endotoxin water matrices and higher-tolerance pharmaceutical products, provided appropriate dilution and inhibition testing are performed per USP <85>.
What is the minimum detectable endotoxin concentration?
The validated lower limit of quantitation (LLOQ) is 0.005 EU/mL under standard assay conditions.
How long does a typical assay take?
Analysis duration is method-dependent but typically completes within 90–120 minutes, including incubation, kinetic readout, and automated calculation.
Is the system compatible with 21 CFR Part 11 requirements?
When configured with appropriate IT infrastructure and procedural controls, the Eclipse software supports electronic records and signatures compliant with 21 CFR Part 11 and Annex 11.