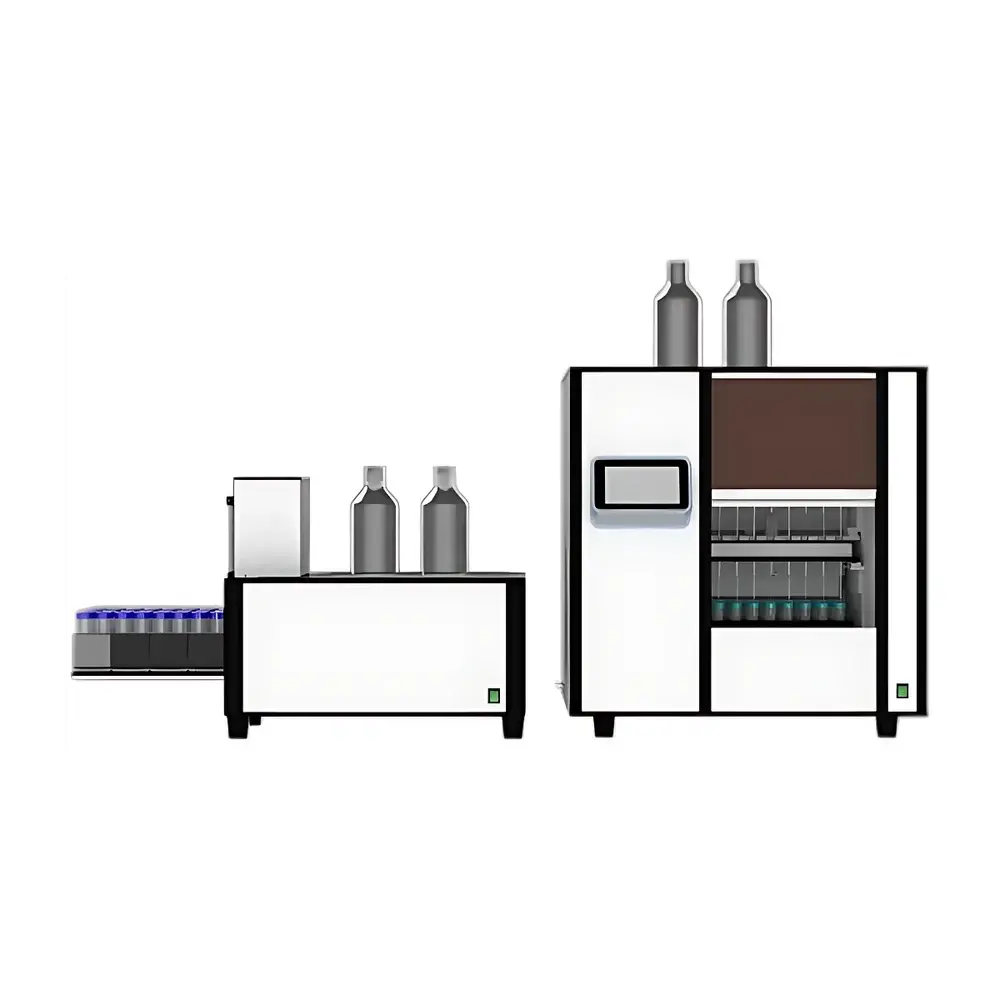
UXS SPE800 Fully Automated Solid Phase Extraction System
| Brand | UXS |
|---|---|
| Origin | Beijing, China |
| Manufacturer Type | Original Equipment Manufacturer (OEM) |
| Product Origin | Domestic (China) |
| Model | SPE800 |
| Automation Level | Fully Automated |
| Number of Channels | 8 (expandable to 16) |
| Cartridge/Membrane Compatibility | Universal (column and disk formats) |
| Flow Rate Control Range | 0–100 mL/min |
| Solvent Selection Capacity | Up to 9 different eluents |
| Sample Capacity | ≥32 positions |
| Sample Loading Volume | 0.1–80 mL |
| Wetted Materials | Polytetrafluoroethylene (PTFE) only |
| Light-Sensitive Mode | Integrated dark-chamber transfer mechanism for photolabile analytes |
Overview
The UXS SPE800 is a fully automated solid phase extraction (SPE) system engineered for high-throughput, reproducible sample preparation in regulated and research-intensive laboratories. It operates on the principle of selective adsorption and elution using reversed-phase, ion-exchange, or mixed-mode sorbents packed in cartridges or membrane disks. Designed for method robustness and minimal operator intervention, the SPE800 integrates precision fluid handling, programmable multi-step protocols, and hardware-level solvent management—enabling consistent recovery of target analytes from complex matrices including biological fluids, environmental water, food extracts, and pharmaceutical formulations.
Key Features
- Fully automated, walk-away operation with pre-programmable, multi-step SPE protocols—including conditioning, loading, washing, drying, and elution phases.
- 8 independent processing channels (expandable to 16 via modular upgrade), each with dedicated peristaltic pump, valve manifold, and position-sensing actuator—ensuring true parallel processing without cross-channel interference.
- Universal cartridge and membrane disk compatibility: accommodates standard 1–6 mL columns and 47 mm/90 mm disk formats without hardware modification.
- PTFE-only fluidic path: all solvent- and sample-contacting components—including tubing, valves, manifolds, and probe tips—are constructed from chemically inert polytetrafluoroethylene, minimizing analyte adsorption and carryover.
- Integrated dark-chamber module: automatically transfers cartridges into a light-shielded enclosure during conditioning and elution steps—critical for stabilizing photolabile compounds such as vitamins A, D, E, retinoids, and certain mycotoxins.
- Programmable flow rate control (0–100 mL/min) with real-time pressure monitoring to prevent column channeling or breakthrough under variable matrix viscosity.
- 9-solvent reservoir system with independent solvent selection per step, supporting gradient elution and multi-solvent wash protocols required by EPA, ISO, and AOAC methods.
Sample Compatibility & Compliance
The SPE800 supports sample volumes ranging from 0.1 mL (e.g., plasma, serum) to 80 mL (e.g., drinking water, milk supernatant), with optional large-volume water modules enabling up to 2 L pre-concentration. Its modular architecture allows integration of application-specific accessories—including vitamin A/D/E purification kits, lactoferrin enrichment cartridges, and high-flow aqueous filtration manifolds—each validated against relevant pharmacopoeial and regulatory guidelines. The system meets core design requirements for GLP-compliant laboratories and supports audit-ready documentation when paired with compliant LIMS or ELN systems. While not inherently 21 CFR Part 11–certified, its software architecture permits integration with third-party electronic signature and audit-trail solutions.
Software & Data Management
The UXS ControlSuite v3.x software provides intuitive graphical protocol builder, real-time run monitoring, and full event logging—including solvent usage, pressure profiles, valve actuation timestamps, and error codes. All method files are stored with version control and user-access metadata. Export options include CSV, PDF, and XML formats compatible with laboratory information management systems (LIMS). Method validation reports can be auto-generated per ICH Q2(R2) principles, documenting repeatability (n ≥ 6), intermediate precision, and system suitability parameters. Remote diagnostics and firmware updates are supported over secure LAN/Wi-Fi connections.
Applications
- Pharmaceutical QC: residue analysis of antibiotics, steroids, and antivirals in raw materials and finished dosage forms per USP & EP monographs.
- Clinical toxicology: extraction of benzodiazepines, opioids, and stimulants from whole blood and urine prior to LC-MS/MS analysis.
- Food safety: multi-residue screening of pesticides, mycotoxins, and veterinary drug residues in dairy, meat, and infant formula matrices.
- Environmental monitoring: trace-level isolation of PAHs, PCBs, and PFAS from surface water, wastewater, and soil leachates per EPA Methods 508.1, 525.3, and 1633.
- Nutritional analysis: quantitative recovery of fat-soluble vitamins (A, D, E, K) and carotenoids from fortified foods and dietary supplements under controlled photostability conditions.
FAQ
Is the SPE800 compatible with third-party SPE cartridges and membranes?
Yes—the system accepts all standard 1–6 mL SPE cartridges and 47 mm/90 mm membrane disks from major suppliers (e.g., Waters, Agilent, Thermo Fisher, Phenomenex) without adapters.
Can method parameters be locked to prevent unauthorized changes in shared-lab environments?
Yes—administrator-defined user roles (Operator, Supervisor, Administrator) enforce parameter editing restrictions and require electronic sign-off for critical modifications.
What maintenance is required to ensure long-term flow accuracy and low carryover?
Routine maintenance includes monthly PTFE tubing replacement, quarterly valve cleaning with methanol/water, and annual calibration of peristaltic pump RPM-to-flow-rate mapping using gravimetric verification.
Does the system support unattended overnight runs?
Yes—integrated run queue management, automatic solvent level detection, and emergency stop logic enable 24-hour batch processing with zero manual intervention.
How is photolabile compound stability verified during dark-chamber operation?
Each dark-chamber cycle includes internal UV-vis sensor feedback confirming ambient light exposure <0.1 µW/cm² at 365 nm throughout the entire extraction sequence.