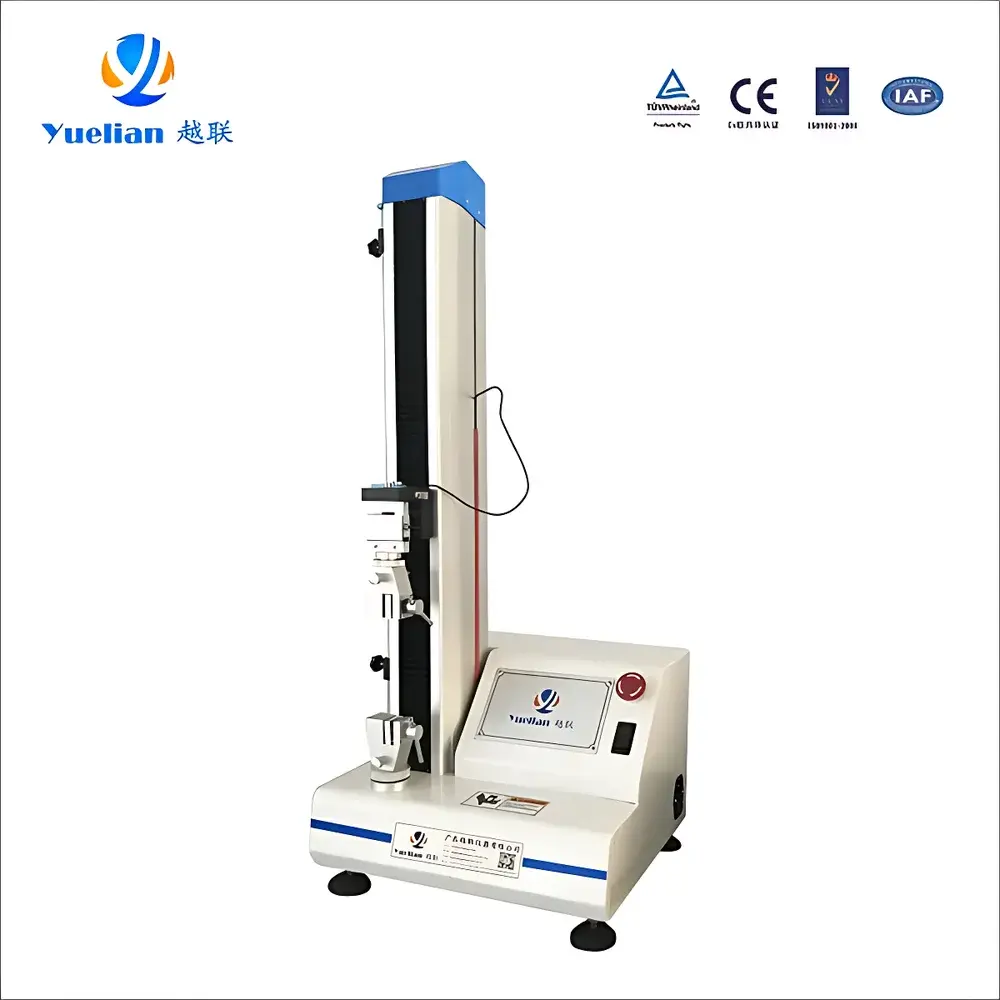
Yuelian YL-S91 Sterility Pouch Seal Integrity Tester
| Key | Brand: Yuelian |
|---|---|
| Origin | Guangdong, China |
| Model | YL-S91 |
| Max Test Force | 1 kN |
| Force Range | 1 kN (FS) |
| Force Accuracy | ±0.5% FS |
| Travel Distance | 800 mm |
| Displacement Resolution | 1/500,000 mm |
| Test Speed | 0.1–500 mm/min (software-controlled) |
| Power Supply | 220 V, 50/60 Hz, 10 A |
| Motor Type | Stepper Motor with Precision Ball Screw Drive |
| Weight | ~35 kg |
| Dimensions (W×D×H) | 40 × 37 × 120 cm |
Overview
The Yuelian YL-S91 Sterility Pouch Seal Integrity Tester is a dedicated electromechanical tensile testing system engineered for quantitative evaluation of seal strength in medical-grade sterilization packaging—primarily heat-sealed peelable pouches used for single-use surgical instruments, gauze, drapes, and implantable devices. It operates on the principle of controlled uniaxial tensile separation at the seal interface, measuring peak force, seal elongation, and failure mode in accordance with ASTM F88/F88M-23 (“Standard Test Method for Seal Strength of Flexible Barrier Materials”). The instrument delivers traceable, repeatable mechanical data required for validation of packaging integrity under ISO 11607-2:2019 and supports compliance with FDA 21 CFR Part 820 and EU MDR Annex I essential requirements for sterile barrier systems.
Key Features
- Precision stepper motor drive coupled with high-rigidity ball screw transmission ensures smooth, low-vibration motion control across the full 800 mm travel range.
- Force transducer calibrated to ±0.5% full scale (FS), with 1/500,000 internal resolution enabling detection of sub-mN force deviations during seal initiation and propagation.
- Software-defined test profiles support standardized ramp-and-hold protocols—including ASTM F88 peel angle (90° or 180°), constant-rate separation (0.1–500 mm/min), and programmable load-hold sequences for creep or relaxation analysis.
- Integrated safety architecture includes overload cutoff, emergency stop, automatic specimen break detection, and dual hardware/software limit switches—fully compliant with EN ISO 13850 and IEC 61508 functional safety principles.
- Modular fixture system accommodates standard ASTM F88 test specimens (e.g., 15 mm wide seals), with optional pneumatic or manual quick-clamp adapters for high-throughput lab environments.
- Compact footprint (40 × 37 × 120 cm) and low power consumption (180 W) enable deployment in QC labs, cleanroom-adjacent testing bays, or ISO 17025-accredited facilities without HVAC retrofitting.
Sample Compatibility & Compliance
The YL-S91 is validated for use with Tyvek®/polyethylene, polyester/aluminum foil laminates, paper-plastic composites, and other flexible barrier materials conforming to ISO 11607-1:2019 specifications. It accepts standard specimen widths (15 mm minimum) and supports both manual and automated sample loading workflows. All test methods align with regulatory expectations for packaging validation per FDA Guidance for Industry: “Container Closure Systems for Packaging Human Drugs and Biologics” (1999) and ISO 11607-2 Annex A verification protocols. Data output meets ALCOA+ criteria when paired with audit-trail-enabled software, supporting GLP/GMP documentation and 21 CFR Part 11 electronic record compliance.
Software & Data Management
Bundled Windows-based control software provides real-time force-displacement curve visualization, automatic calculation of seal strength (N/15 mm), average peel force, and failure classification (cohesive vs. adhesive). Raw data exports to CSV, Excel, and PDF formats; metadata tagging (operator ID, lot number, environmental conditions) is fully configurable. Optional database integration enables direct upload to LIMS platforms via ODBC or REST API. Audit trail functionality logs all user actions—including method edits, calibration events, and result overrides—with immutable timestamps and digital signatures.
Applications
- Validation of heat seal parameters (temperature, dwell time, pressure) during packaging process qualification (IQ/OQ/PQ).
- Comparative evaluation of seal integrity across different film suppliers or sterilization cycles (steam, EtO, gamma).
- Stability testing of aged pouches per ISO 11607-2 Clause 7.4 (accelerated aging + seal strength retention).
- Root cause analysis of field failures—correlating seal morphology (via post-test optical microscopy) with mechanical performance metrics.
- Supporting submissions to notified bodies for CE marking under MDR 2017/745 Annex I, Section 10.5.2 (sterile barrier system verification).
FAQ
Does the YL-S91 comply with ASTM F88 testing requirements?
Yes—it implements the prescribed 90° or 180° peel geometry, force measurement accuracy (±0.5% FS), and specimen width tolerance (15 ± 0.1 mm) defined in ASTM F88/F88M-23.
Can it be integrated into an existing LIMS environment?
Yes—via optional ODBC driver or HTTP-based API, supporting structured data ingestion including test ID, timestamp, operator, force curve, and pass/fail status.
Is calibration traceable to national standards?
Force calibration is performed using NIST-traceable deadweight standards; certificate includes uncertainty budget per ISO/IEC 17025:2017 requirements.
What maintenance is required for long-term accuracy?
Annual verification of force transducer linearity and displacement encoder resolution is recommended; no routine lubrication of the ball screw assembly is needed due to sealed pre-greased design.
Does the system support multi-language UI and reporting?
The base software supports English and Simplified Chinese interfaces; custom language packs (e.g., German, Spanish, Japanese) are available upon request for regulated markets.