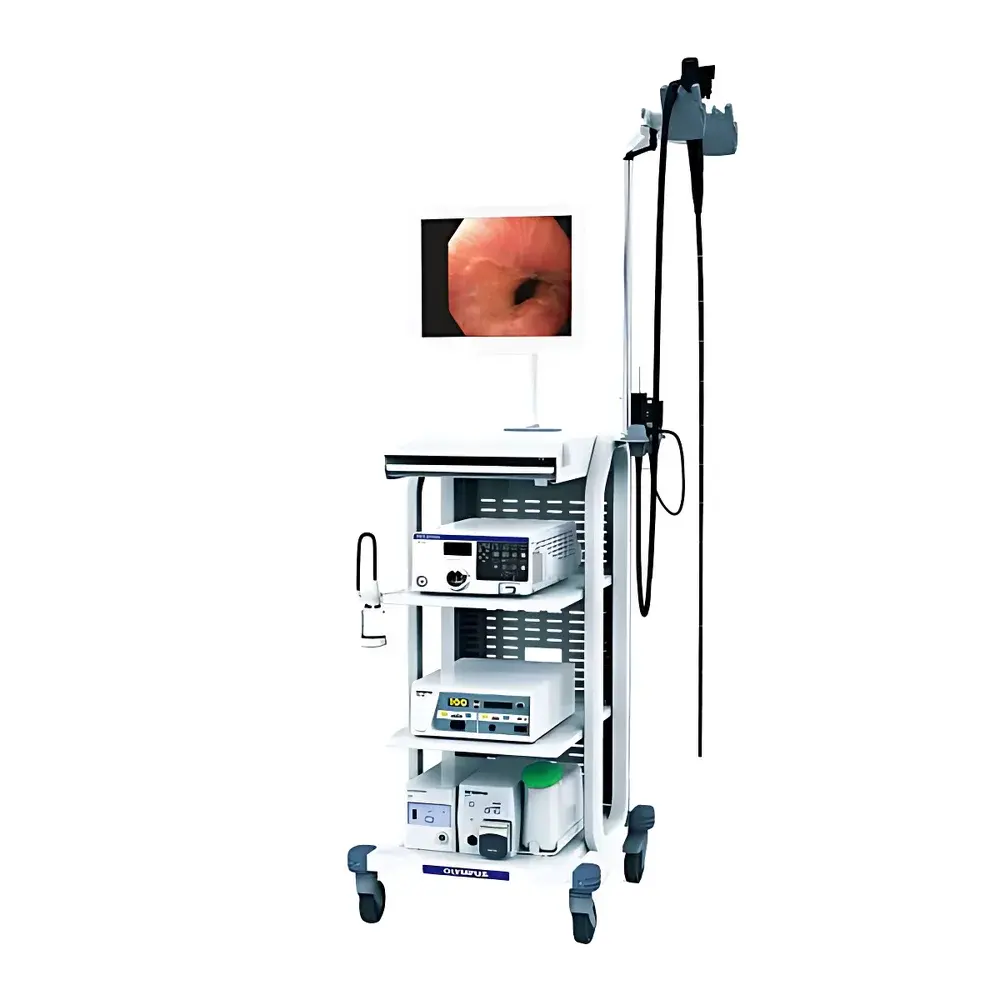
Olympus CV-170 Video Processor System with GIF-H170 Gastroscope and CF-H170L/i Colonoscope
| Brand | Olympus |
|---|---|
| Country of Origin | Japan |
| Model | CV-170 + GIF-H170 + CF-H170L/i |
| Regulatory Clearance | China NMPA Import Registration No. 20152221796 (CV-170), 20143222972 (GIF-H170), 20143222973 (CF-H170L), 20143222945 (CF-H170i) |
| Power Input | 100–240 V AC, 50/60 Hz ±1 Hz |
| Dimensions (W×H×D) | 295 × 145 × 425 mm |
| Weight | 11.0 kg |
| Light Source | Integrated Long-Life LED |
| Video Output | HD-SDI, SD-SDI, DVI-D, RGB/YPbPr (1080i), Composite/Y/C (480i/576i) |
| Image Storage | MAJ-1925 Portable Memory (2 GB, TIFF/JPEG compression) |
| Compatibility | EVIS 100/130/140, OLYMPUS 150, EXERA 160, EXERA II 180, GI/BF/VISERA Series Endoscopes |
Overview
The Olympus CV-170 Video Processor System is a CE-marked, NMPA-registered gastrointestinal endoscopy platform engineered for clinical precision, workflow efficiency, and long-term operational reliability. Designed as the central imaging hub for Olympus’ 170-series endoscopes—including the GIF-H170 gastroscope and CF-H170L/i colonoscope—the CV-170 integrates HDTV (1080i) image acquisition with Narrow Band Imaging (NBI) optical enhancement technology. Its core architecture employs a solid-state LED illumination source delivering stable, consistent brightness without thermal drift or lamp degradation—eliminating routine bulb replacement and reducing total cost of ownership. The system operates on a real-time digital signal processing pipeline optimized for low-latency image rendering, dynamic contrast adjustment, and noise suppression. As a Class I medical electrical device per IEC 60601-1, the CV-170 supports dual-standard video output (NTSC/PAL), enabling seamless integration into both legacy analog and modern HD-SDI-equipped endoscopy suites. It is intended for use in gastroenterology, colorectal screening, and therapeutic endoscopy procedures under physician supervision in hospital-based or ambulatory surgical centers.
Key Features
- HDTV Imaging Engine: Delivers 1080i resolution with edge-preserving interpolation, enabling visualization of mucosal microarchitecture—including capillary networks, pit patterns, and subtle color gradients—at clinically relevant working distances.
- Narrow Band Imaging (NBI): Utilizes spectral filtering to enhance hemoglobin absorption at 415 nm and 540 nm wavelengths, increasing contrast between superficial vasculature and surrounding epithelium—supporting early neoplastic lesion detection without dye chromoendoscopy.
- Prefreeze Functionality: Automatically buffers up to 10 consecutive frames prior to freeze command execution; selects and saves the highest-resolution frame from the buffer—reducing operator dependency on precise timing during static image capture.
- Dual-Mode Structural Enhancement: Offers A-type (optimized for high-contrast visualization of colonic mucosal folds) and B-type (enhanced vascular delineation for esophagogastric junction and duodenal assessment) algorithms, each with seven adjustable intensity levels.
- LED Illumination Architecture: Provides >10,000-hour rated lifetime, zero warm-up delay, and <35 dB(A) acoustic noise—meeting ISO 14155 requirements for quiet procedural environments.
- Integrated Data Management: Native support for MAJ-1925 portable memory (2 GB, USB 2.0 compatible) enables lossless TIFF or compressed JPEG (1:5 or 1:10) image export, patient metadata embedding (ID, name, DOB, procedure timestamp), and system configuration backup.
Sample Compatibility & Compliance
The CV-170 is validated for interoperability with Olympus’ full range of 170-series flexible electronic endoscopes—including GIF-H170 (gastroscope), CF-H170L/i (colonoscopes), and GIF-XP170N—and maintains backward compatibility with EVIS 100/130/140, OLYMPUS 150, EXERA 160, EXERA II 180, and GI/BF/VISERA platforms. All connected endoscopes comply with ISO 8600-1:2018 (flexible endoscope safety) and carry valid NMPA import registrations. The system meets electromagnetic compatibility (EMC) requirements per IEC 60601-1-2:2014 and adheres to essential performance criteria outlined in EN 62304:2015 (medical device software lifecycle). For regulatory traceability, audit logs record user login events, image capture timestamps, NBI activation status, and storage media insertion/removal—supporting GLP/GCP documentation workflows and FDA 21 CFR Part 11-compliant electronic records when deployed with validated PACS or EMR interfaces.
Software & Data Management
The CV-170 firmware includes embedded DICOM SR (Structured Reporting) capability for structured annotation of findings (e.g., “NBI-positive Barrett’s esophagus,” “Paris classification Type 0-IIa polyp”). Patient data fields—name, ID, gender, age, birth date, and procedure time—are editable via front-panel keypad or external keyboard and persist across sessions. Image metadata is embedded in EXIF headers and preserved during TIFF/JPEG export. The MAJ-1925 memory interface supports bidirectional transfer: images and settings upload from CV-170 to PC, while preconfigured user profiles and calibration files download to the processor. Firmware updates are performed via USB drive using Olympus-provided signed binaries—ensuring integrity verification and rollback protection. No cloud connectivity or remote access functionality is included, preserving data sovereignty in accordance with HIPAA and GDPR-aligned institutional IT policies.
Applications
This system is indicated for diagnostic and therapeutic upper and lower GI endoscopy, including but not limited to: surveillance of Barrett’s esophagus using NBI-guided targeted biopsy; detection of diminutive colorectal adenomas (<5 mm) during screening colonoscopy; evaluation of gastric intestinal metaplasia and early gastric cancer; assessment of post-polypectomy resection margins; and endoscopic mucosal resection (EMR) guidance. Its HDTV+NBI combination supports adherence to ESGE quality indicators—particularly withdrawal time ≥6 minutes and adenoma detection rate (ADR) optimization. The CF-H170L/i’s variable stiffness control and GIF-H170’s 2–100 mm depth-of-field enable reliable intubation in anatomically challenging cases (e.g., post-surgical adhesions, diverticular disease), while the waterproof connector design streamlines reprocessing per AAMI ST91:2023 standards.
FAQ
Is the CV-170 compatible with non-Olympus endoscopes?
No. The CV-170 is designed exclusively for Olympus 170-series and legacy EVIS/EXERA-compatible endoscopes. Third-party scopes lack electrical, mechanical, and optical interface alignment required for NBI synchronization and structural enhancement processing.
What is the expected service life of the integrated LED light source?
The LED module is rated for ≥10,000 operating hours under continuous use at nominal output—equivalent to approximately 5–7 years of typical clinical volume (15–20 procedures/day). No scheduled replacement is required within this period.
Does the system support video recording to external devices?
Yes. Via HD-SDI or DVI-D output, the CV-170 can feed live video to certified medical-grade DVRs, surgical recording systems, or PACS archives—provided the receiving device accepts SMPTE 292M or WUXGA 1920×1200 signals.
Can patient data be exported in HL7 or FHIR format?
No. The CV-170 exports only image files with embedded EXIF metadata. HL7/FHIR integration requires middleware solutions interfacing with hospital EMR systems via DICOM SR or custom API bridges.
Is the MAJ-1925 memory device reusable across multiple CV-170 units?
Yes. The MAJ-1925 is formatted to Olympus’ proprietary FAT32 partition schema and retains compatibility across all CV-170 installations without reformatting—enabling centralized image archiving and cross-departmental case review.