CS Bio CS536 Series Peptide Synthesizer
| Brand | CS Bio |
|---|---|
| Origin | USA |
| Model | CS536 Series |
| Synthesis Scale | Pilot-scale |
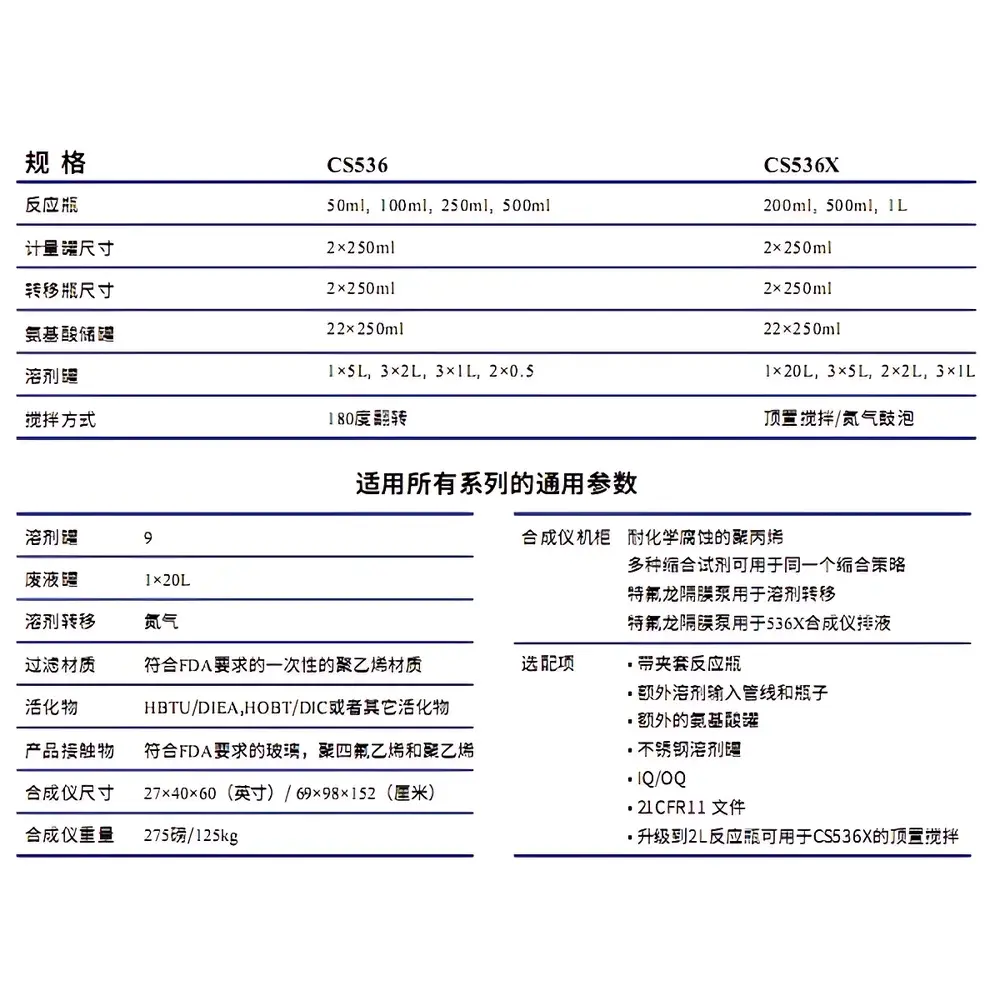
| Reactor Volume Range | 50 mL to 1 L |
| Stirring Options | 180° Tilt-based Mixing or Overhead Stirring |
| Intended Use | Process Development & Scale-up Studies |
| Distribution Status | Authorized Distributor (Non-OEM) |
Overview
The CS Bio CS536 Series Peptide Synthesizer is an automated, benchtop-to-pilot-scale solid-phase peptide synthesis (SPPS) platform engineered for reproducible, scalable manufacturing of custom peptides under controlled GMP-adjacent conditions. Unlike laboratory-scale synthesizers optimized for milligram outputs, the CS536 implements a single-channel, batch-mode architecture designed specifically to bridge the gap between discovery-phase synthesis and early-stage clinical material production. Its core operation adheres to Fmoc- or Boc-based SPPS chemistry protocols, with precise reagent delivery, inert atmosphere control (N₂ or Ar), and real-time monitoring of coupling/deprotection cycles. The system’s mechanical architecture supports both 180° tilt-based agitation — which enhances resin swelling and mass transfer in viscous reaction media — and optional overhead magnetic or mechanical stirring, enabling method transfer across diverse resin types (e.g., Wang, Rink amide, Sieber amide) and challenging sequences (e.g., hydrophobic, sterically hindered, or long-chain peptides >50 residues).
Key Features
- Pilot-scale synthesis capacity: accommodates resin loads from 1 g to 100 g, supporting batch outputs ranging from hundreds of milligrams to multi-gram quantities per run.
- Modular reactor system: interchangeable reaction vessels (50 mL, 100 mL, 250 mL, 500 mL, and 1 L) constructed from chemically resistant glass and PTFE-lined components to ensure compatibility with aggressive reagents (e.g., TFA, DCM, DMF, piperidine).
- Dual agitation modes: user-selectable 180° tilting motion for uniform resin suspension without shear-induced fragmentation, or overhead stirring with programmable speed and torque control for high-viscosity couplings.
- Integrated fluidic management: eight independently controlled solvent/reagent channels with gravimetric or volumetric dispensing options, pressure-regulated gas purging, and waste collection with solvent recovery compatibility.
- Process-critical environmental controls: sealed reaction chamber with continuous inert gas flow, temperature monitoring (ambient to 60°C), and dew-point-controlled drying steps to minimize side reactions during cleavage.
- Robust mechanical design: stainless-steel frame, IP54-rated enclosure, and CE-compliant electrical architecture suitable for installation in ISO Class 7/8 cleanrooms or QC laboratories operating under GLP guidelines.
Sample Compatibility & Compliance
The CS536 supports standard polystyrene- and PEG-based resins (e.g., NovaSyn® TG, ChemMatrix®, Tentagel®), as well as specialty supports for cyclization, on-resin labeling, or orthogonal protection schemes. It is compatible with common coupling reagents (HBTU, HATU, DIC/Oxyma), activators, scavengers, and cleavage cocktails (TFA/H2O/TIS, TFA/EDT/phenol). The system meets fundamental requirements for documentation traceability aligned with FDA 21 CFR Part 11 (when paired with validated electronic lab notebook integration), and its operational parameters are configurable to support compliance with ICH Q5C (stability of biotechnological/biological products), USP <1043> (peptide characterization), and ASTM E2500-13 (verification of pharmaceutical equipment). Full audit trails, user access levels, and electronic signature capability can be implemented via optional software modules.
Software & Data Management
The CS536 operates via CS Bio’s proprietary SynthAssist™ control software, a Windows-based application offering intuitive method creation, real-time cycle logging, and deviation alerting. Each synthesis run generates a timestamped, immutable log file containing reagent lot numbers, flow rates, dwell times, pressure profiles, and temperature history — all exportable in CSV or PDF format for regulatory submission. The software supports method templating for rapid protocol replication, version-controlled method libraries, and integration with LIMS platforms through OPC UA or RESTful API interfaces. Data integrity safeguards include automatic backup to network drives, dual storage redundancy, and encryption-at-rest for archived synthesis records.
Applications
- Process development for therapeutic peptides, including optimization of coupling efficiency, deprotection kinetics, and resin loading strategies.
- Manufacturing of reference standards and impurity controls for pharmacopeial testing (USP, EP, JP).
- Scale-up studies supporting IND-enabling toxicology batches and Phase I clinical trial material synthesis.
- Production of peptide antigens, epitope mapping libraries, and constrained macrocycles for structural biology and assay development.
- Method validation and transfer between R&D labs and contract development and manufacturing organizations (CDMOs).
FAQ
What is the maximum peptide length achievable on the CS536?
The system has successfully synthesized peptides up to 75 residues under optimized conditions; performance depends on sequence composition, resin choice, and coupling strategy.
Can the CS536 be integrated into a cGMP manufacturing environment?
Yes — when deployed with validated software, documented IQ/OQ/PQ protocols, and qualified reagent handling procedures, it supports GMP-aligned operations for non-sterile active pharmaceutical ingredient (API) synthesis.
Does the CS536 support real-time monitoring of coupling completion?
It does not include in-line analytics (e.g., FTIR or conductivity sensors); endpoint verification requires offline Kaiser or TNBS tests, though automated sampling ports can be added as an option.
Is remote operation or monitoring supported?
Remote desktop access is permitted within secure corporate networks; however, live control requires local operator presence per safety and data integrity policies.
What maintenance intervals are recommended for routine operation?
Daily visual inspection of fluid paths and seals; quarterly calibration of mass flow controllers and pressure transducers; annual full system validation per manufacturer-recommended SOPs.