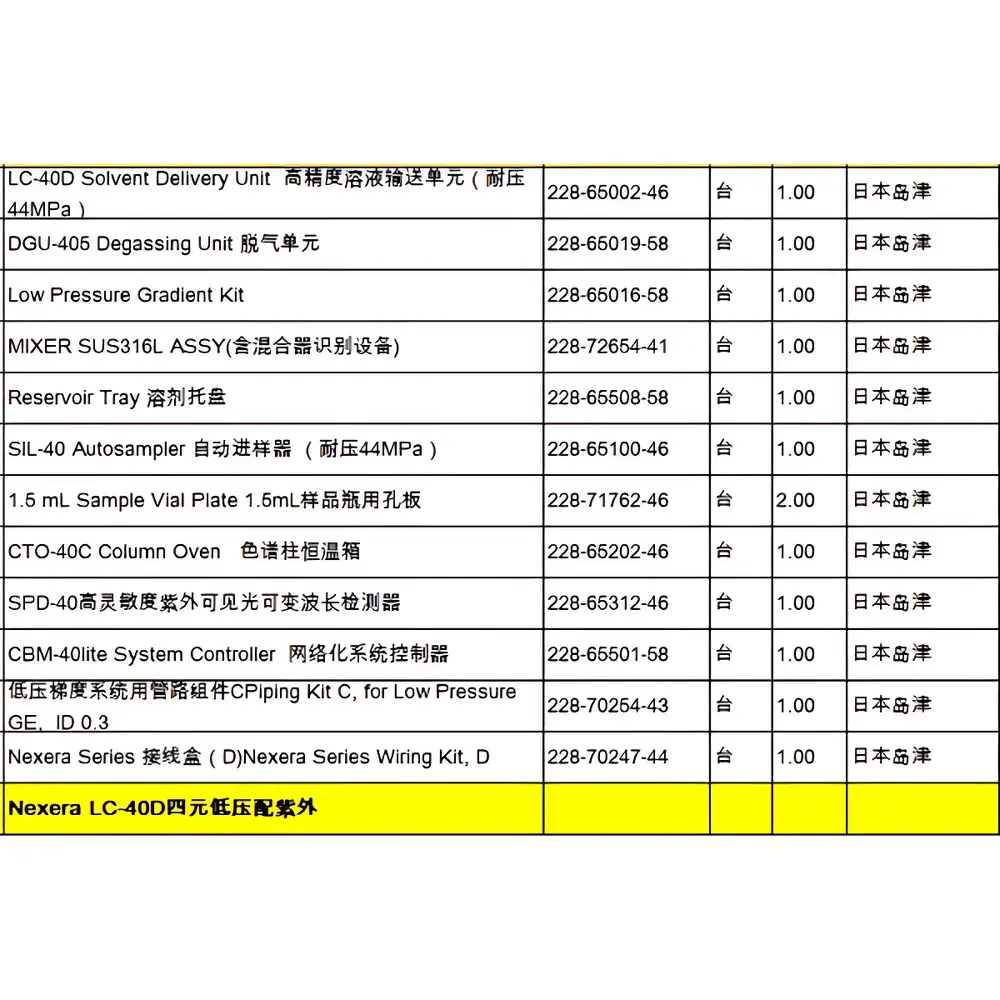
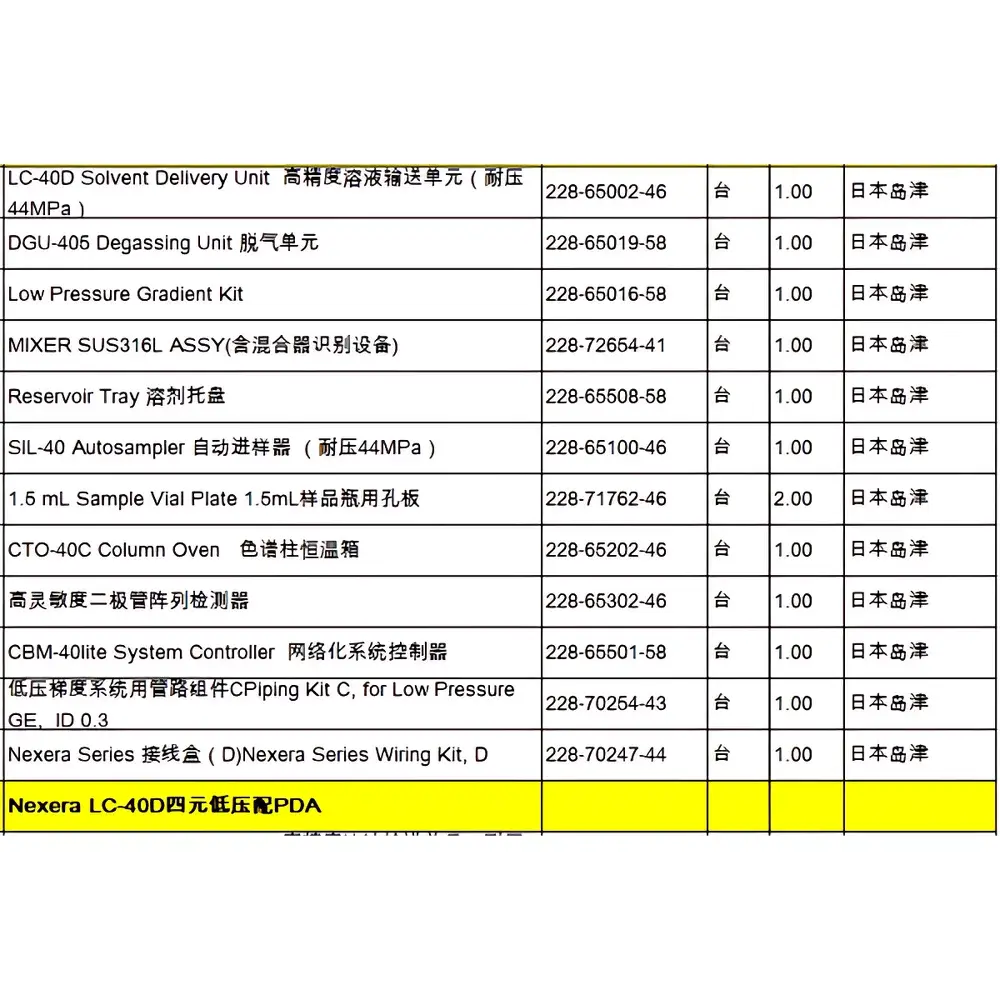
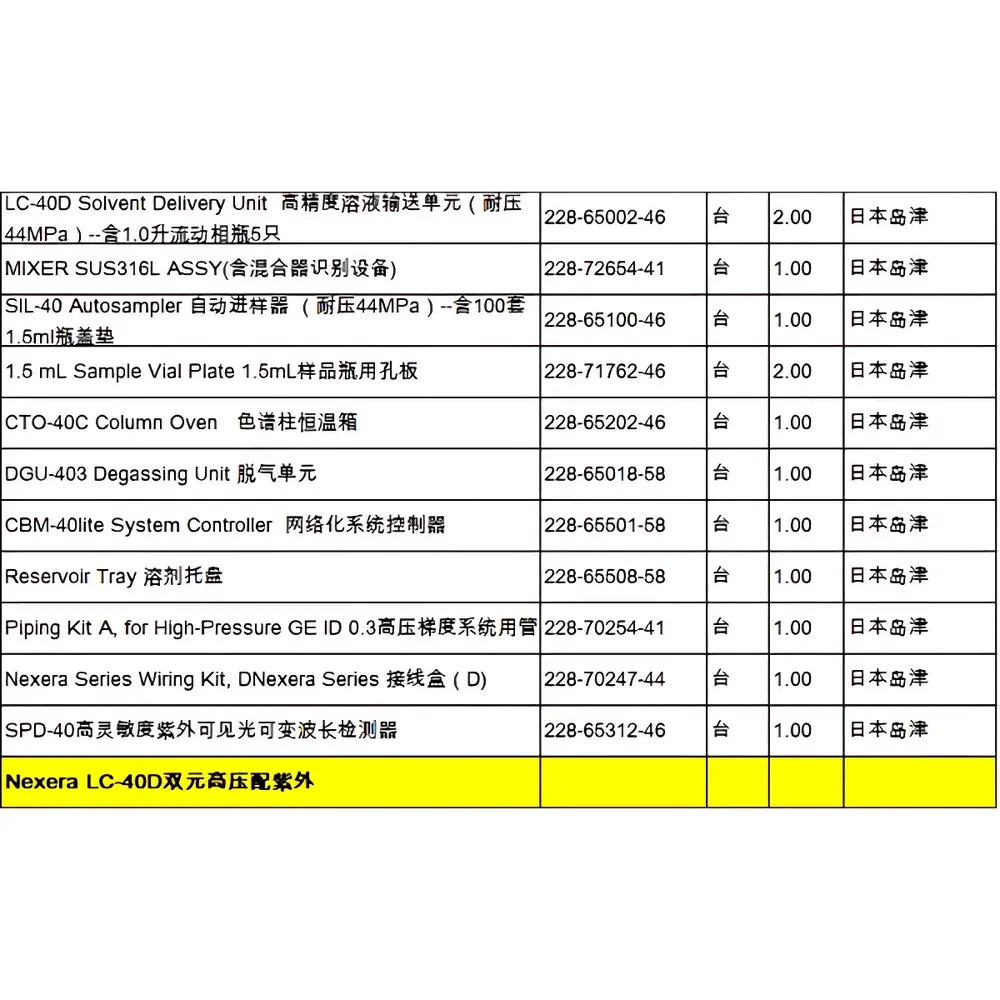
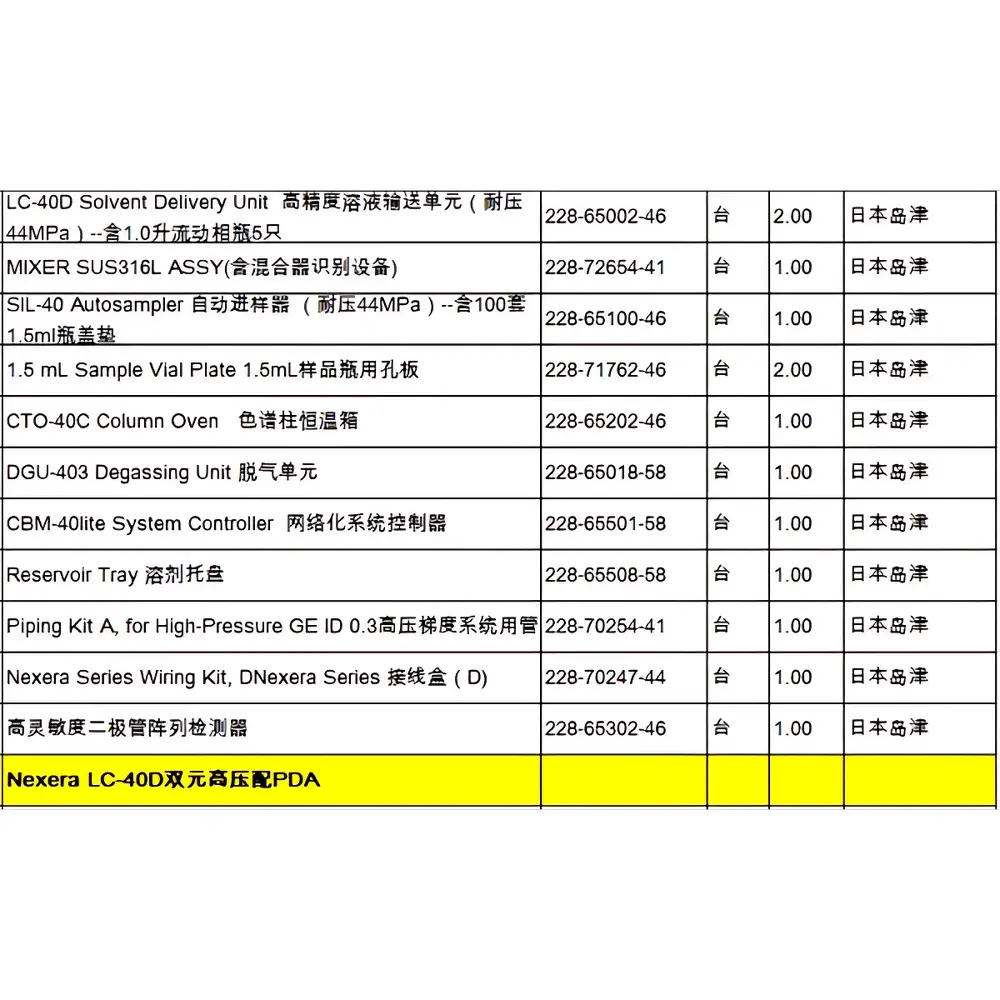
Shimadzu Nexera LC-40D Used High-Performance Liquid Chromatograph
| Brand | Shimadzu |
|---|---|
| Origin | Japan |
| Model | Nexera LC-40D |
| Instrument Type | Liquid Chromatograph |
| Year of Manufacture / Service Duration | <1 year |
| Warranty | 12 months |
| Compliance | Imported instrument, CE-marked configuration (per standard Shimadzu factory specifications) |
| Software Platform | LabSolutions LC-40 Series (v5.9 or later, validated for GLP/GMP environments) |
| Detection | UV-Vis detector (SPD-M40), dual-wavelength simultaneous monitoring, 190–800 nm range |
Overview
The Shimadzu Nexera LC-40D is a high-performance, modular liquid chromatography system originally engineered for regulated and research-intensive laboratories. Based on Shimadzu’s proven Nexera platform architecture, the LC-40D implements low-dispersion binary gradient pumping, active solvent pre-mixing, and real-time flow/pressure compensation to ensure exceptional retention time reproducibility (<0.05% RSD) and baseline stability over extended runs. Its core separation principle relies on reversed-phase, normal-phase, ion-exchange, or size-exclusion chromatography—configured via user-selectable column ovens, autosamplers, and detector modules. Designed for compliance-critical workflows, the system meets fundamental requirements for method transfer, system suitability testing, and routine QC/QA in pharmaceutical, biotech, food safety, and environmental analysis applications.
Key Features
- Binary ultra-high-pressure gradient pump (up to 66 MPa / 9570 psi) with pulse-free flow delivery and automatic solvent degassing
- High-precision autosampler (SIL-40D) supporting 44-position microplate handling, 0.1–100 µL injection volume range, and <0.25% RSD injection precision at 5 µL
- Column oven (CTO-40D) with triple-zone temperature control (4–85 °C, ±0.1 °C stability) and integrated solvent pre-heating
- UV-Vis photodiode array detector (SPD-M40) featuring dual-lamp switching (D₂ and W lamp), 1024-pixel linear array, and real-time spectral acquisition (1–5 Hz scan rate)
- Integrated system controller (CBM-40D) enabling synchronized timing, event-driven sequence execution, and hardware-level error logging
- Pre-owned unit certified to original factory specifications, fully refurbished with new pump seals, detector lamps, and flow cell; verified per Shimadzu PQ protocol
Sample Compatibility & Compliance
The LC-40D accommodates a broad range of sample matrices—including small-molecule APIs, peptides, organic acids, residual solvents, pesticides, and polar metabolites—when paired with appropriate stationary phases (C18, HILIC, phenyl-hexyl, etc.) and mobile phase chemistries. All configurations comply with ISO/IEC 17025:2017 general requirements for testing laboratories and support alignment with USP , ICH Q2(R2), and ASTM D7705 for chromatographic method validation. Data integrity is maintained through LabSolutions’ audit-trail-enabled acquisition mode, compliant with FDA 21 CFR Part 11 requirements when deployed with electronic signatures and role-based access controls.
Software & Data Management
Controlled exclusively by Shimadzu LabSolutions LC-40 software (v5.9 or newer), the system provides full method development, sequence management, peak integration, and report generation capabilities. The software supports multi-user environments with configurable permission levels, automatic backup of raw data (.lcd files), and export to CSV, PDF, and XML formats for LIMS integration. Built-in system suitability assessment tools calculate tailing factor, resolution, theoretical plates, and %RSD for replicate injections—all traceable within the electronic lab notebook (ELN) framework. Raw data files retain native metadata including instrument ID, timestamp, operator login, and hardware calibration status.
Applications
- Pharmaceutical QC: Assay, related substances, dissolution testing, and extractables/leachables profiling per USP monographs
- Biopharmaceutical analysis: Monoclonal antibody purity assessment (SEC-HPLC), peptide mapping (RP-HPLC), and glycan profiling
- Environmental monitoring: PAHs, PCBs, and endocrine disruptors in water and soil extracts (EPA Methods 8330B, 8082A)
- Food & beverage: Mycotoxin quantification (aflatoxins, ochratoxin A), preservative detection (sorbic/benzoic acid), and vitamin profiling
- Academic & contract research: Method development support, stability-indicating assays, and forced degradation studies
FAQ
Is this unit eligible for Shimadzu factory service contracts?
Yes—refurbished Nexera LC-40D systems sold through authorized pre-owned channels qualify for optional extended service agreements directly administered by Shimadzu Scientific Instruments, Inc., including remote diagnostics and priority parts dispatch.
Does the system include validated IQ/OQ documentation?
Standard delivery includes a comprehensive Pre-Delivery Qualification (PDQ) report covering mechanical, electrical, and functional verification. Full IQ/OQ protocols can be generated onsite by certified Shimadzu Field Application Specialists upon request.
Can the LC-40D be upgraded to support mass spectrometry coupling?
The system features standard ESI-compatible flow cell output and auxiliary valve ports, enabling seamless integration with Shimadzu LCMS-8040/8050 or third-party MS detectors via split-flow or direct coupling configurations.
What consumables and spare parts are included with purchase?
Each unit ships with one set of OEM pump seals, two UV lamp assemblies, one flow cell cleaning kit, and 12 months of firmware update entitlement via Shimadzu’s Customer Portal.
How is data security ensured during remote support sessions?
All remote connectivity uses TLS 1.2-encrypted VNC sessions initiated only after explicit user authorization; no persistent remote access is enabled by default, and session logs are retained per internal IT policy.