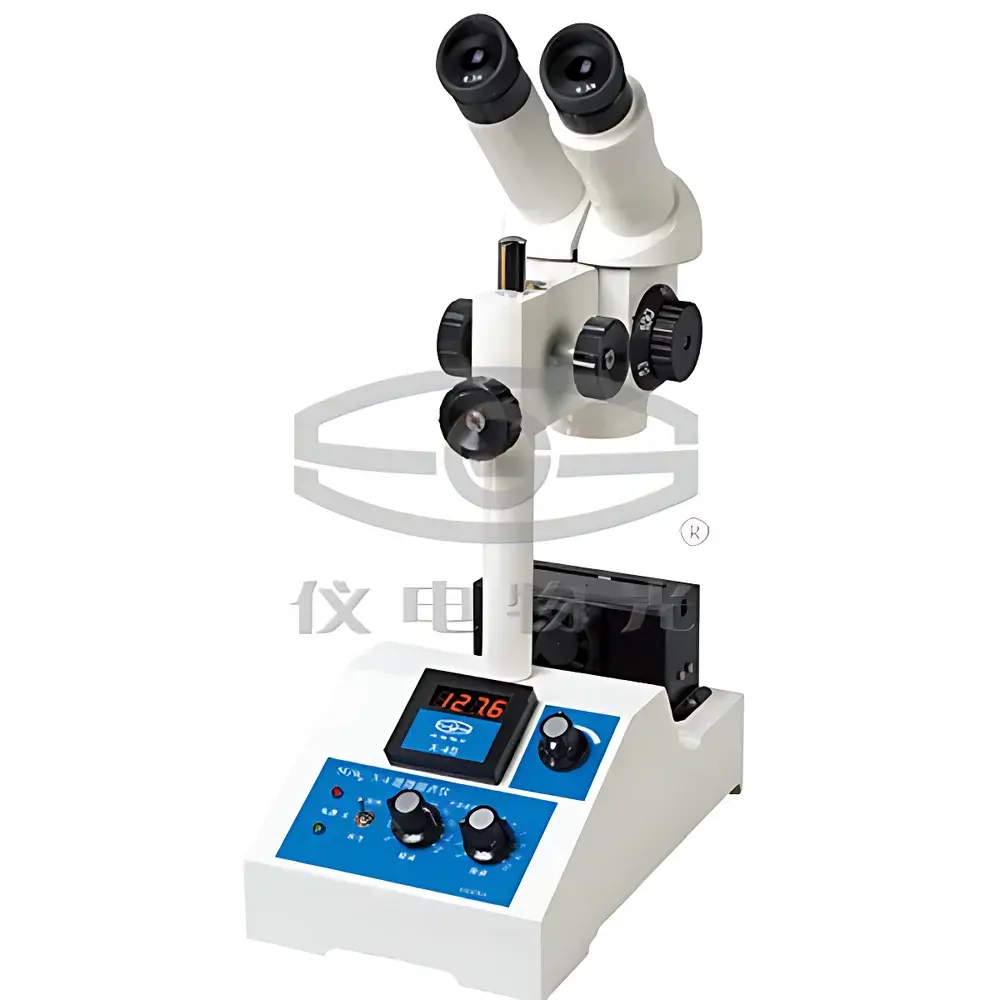
YDWG SGW® X-4B Microscopic Melting Point Apparatus
| Brand | YDWG |
|---|---|
| Origin | Shanghai, China |
| Model | SGW® X-4B |
| Temperature Range | Ambient to 330 °C |
| Temperature Repeatability | ±1 °C at ≤200 °C |
| Temperature Accuracy | ±0.1 °C |
| Temperature Resolution | 0.1 °C |
| Observation Method | Binocular Stereo Microscope |
| Magnification | 40×–100× (variable zoom) |
| Power Supply | 220 V ±22 V, 50 Hz ±1 Hz |
| Dimensions | 269 × 160 × 435 mm |
| Net Weight | 4 kg |
Overview
The YDWG SGW® X-4B Microscopic Melting Point Apparatus is a precision thermal analysis instrument engineered for accurate, visually confirmed determination of solid-phase transition temperatures in crystalline organic compounds. It operates on the principle of controlled heating coupled with real-time optical observation—enabling direct visualization of melting onset, meniscus formation, and complete liquefaction under magnification. Unlike automated photometric or differential thermal analysis (DTA) systems, the SGW® X-4B relies on human operator judgment validated by calibrated thermal control and high-fidelity stereoscopic imaging. This method remains the reference standard for pharmacopoeial compliance (e.g., USP , EP 2.2.14, JP 2.60) where visual endpoint confirmation is explicitly required. The apparatus integrates a thermally stabilized hot stage with dual-mode sample introduction—capillary tube method (ASTM E324-compliant) and hot-stage microscopy (glass slide/glass cover method)—making it suitable for both routine QC screening and research-grade polymorph characterization.
Key Features
- Thermally isolated heating block with PID-controlled temperature regulation, ensuring stable ramp rates and minimal thermal overshoot across the full 20 °C–330 °C operating range.
- Dual observation configuration: binocular stereo microscope with continuously variable zoom (40×–100×), adjustable interpupillary distance, and coaxial LED illumination optimized for low-glare crystal morphology assessment.
- Integrated anti-draft enclosure (standard on X-4B variant) minimizes convective heat loss and ambient air disturbance—critical for reproducible endpoint detection below ±1 °C at sub-200 °C.
- Digital temperature display with 0.1 °C resolution and NIST-traceable calibration capability; accuracy verified per ISO/IEC 17025 procedures using certified reference materials (e.g., pure naphthalene, adipic acid, anthraquinone).
- Compact benchtop footprint (269 × 160 × 435 mm) and lightweight aluminum alloy chassis (4 kg net weight) facilitate integration into regulated laboratory environments including GLP-compliant QC labs and university teaching facilities.
Sample Compatibility & Compliance
The SGW® X-4B accommodates standard 1-mm OD capillary tubes (90 mm length, compatible with WRS-series instrumentation) and standard microscope slides (76 × 26 mm) with #1.5 cover glasses. It supports both anhydrous and solvent-containing samples, though volatile or hygroscopic compounds require sealed capillaries or inert atmosphere purging (optional accessory). The instrument meets mechanical and electrical safety requirements per IEC 61010-1:2010. While not inherently 21 CFR Part 11 compliant (lacking electronic audit trail), its analog-digital hybrid architecture allows integration into validated workflows when paired with documented manual observation logs and external calibration records—fully supporting GMP Annex 15 and WHO TRS 986 validation expectations for Category B equipment.
Software & Data Management
The SGW® X-4B is a standalone hardware platform with no embedded firmware or proprietary software. Temperature readings are displayed via a high-contrast LCD panel; all observational data—including melt onset time, phase transition description, and operator ID—are recorded manually in bound lab notebooks or LIMS-integrated electronic worksheets. For laboratories requiring digital traceability, optional USB thermal camera modules (e.g., FLIR A35) may be mounted above the viewing port to capture time-stamped video sequences synchronized with external data loggers—a practice increasingly adopted for FDA pre-submission documentation and internal audit readiness.
Applications
- Pharmaceutical QC: Identity verification and purity assessment of active pharmaceutical ingredients (APIs) and excipients per USP monographs.
- Organic synthesis monitoring: Real-time tracking of reaction intermediates and final product crystallinity during process development.
- Polymorph screening: Differentiation of solid-state forms (e.g., Form I vs. Form II) based on distinct melting profiles and morphological behavior under thermal stress.
- Academic instruction: Hands-on training in thermal behavior fundamentals, crystal lattice energy concepts, and pharmacopoeial methodology for undergraduate and graduate chemistry curricula.
- Regulatory support: Generation of raw observational evidence for regulatory submissions requiring visual melting point confirmation (e.g., DMF filings, CMC sections).
FAQ
Is the SGW® X-4B compliant with USP & EP melting point methodology?
Yes—the instrument implements the capillary tube method per USP and Ph. Eur. 2.2.14, including specified heating rate control, observer positioning, and endpoint criteria.
Can it be used for decomposition point determination?
It is not designed for decomposition analysis; discoloration, charring, or gas evolution must be interpreted qualitatively by the operator and reported separately from melting onset.
What calibration standards are recommended?
NIST-traceable certified reference materials such as naphthalene (80.26 °C), adipic acid (152.1 °C), and anthraquinone (286.0 °C) are routinely employed for multi-point verification.
Does the unit support IQ/OQ/PQ validation protocols?
Yes—YDWG provides vendor documentation packages supporting installation qualification (IQ), operational qualification (OQ), and performance qualification (PQ) per ASTM E2500 and EU GMP Annex 15 guidelines.
Is third-party 3Q certification available?
Authorized service partners offer on-site 3Q execution with full documentation, including uncertainty budgets, calibration certificates, and deviation reports aligned with ISO/IEC 17025 requirements.