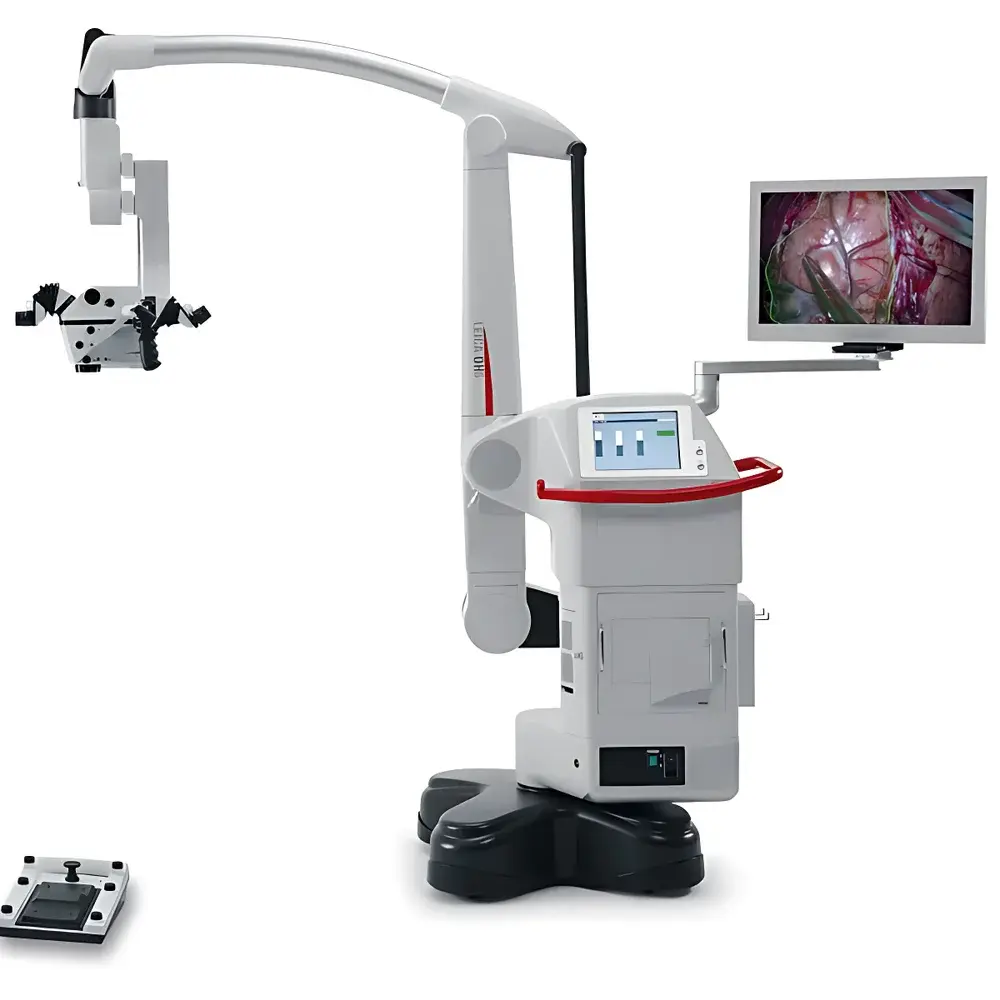
Leica OHX Image Projection Module
| Brand | Leica |
|---|---|
| Country of Origin | Germany |
| Model | OHX |
| Component Type | Surgical Microscope Integration Module |
| Display Resolution | 1920 × 1080 (Full HD) |
| Contrast Ratio | 500:1 |
| Light Source | LED backlight |
| Display Technology | LCD |
| Viewing Options | Monocular (left/right), Binocular (dual-eye), Overlay or Non-overlay Mode |
| Control Interface | Integrated microscope handle button, Wireless footswitch, Optional control panel |
| IGS Compatibility | Native integration with major neurosurgical navigation platforms (e.g., BrainLab, Stryker, Medtronic StealthStation) |
| Fluorescence Support | FL800 fluorescence mode synchronization |
| Recording Capability | Full HD video capture with metadata tagging (magnification, working distance, illumination ratio, acquisition timestamp) |
| Mounting | Integrated within Leica M530 surgical microscope main body |
| Regulatory Status | CE-marked as Class IIa medical device |
| Compliance | EN ISO 13485, IEC 60601-1, FDA 510(k) cleared (K220227), supports 21 CFR Part 11 audit trail when paired with Leica LAS X NAV software |
Overview
The Leica OHX Image Projection Module is a precision-engineered optical integration component designed exclusively for neurosurgical and cranial microsurgery workflows. It functions as a real-time, high-fidelity image overlay system embedded directly into the optical path of the Leica M530 surgical microscope. Unlike external display monitors or secondary screens, the OHX projects auxiliary visual data—such as intraoperative fluorescence (FL800), endoscopic feeds, neuronavigation guidance, and stereotactic coordinates—directly into the surgeon’s binocular field of view via the microscope eyepieces. This implementation adheres to the principles of optical collimation and parfocal alignment, ensuring zero latency, pixel-perfect registration, and minimal parallax deviation between projected overlays and the live surgical scene. The module operates on a dual-path optical architecture: one path preserves the native microscope image fidelity; the second path injects digitally registered, geometrically calibrated supplemental imagery at the intermediate image plane. This design eliminates head movement, gaze shifting, and cognitive load associated with screen-to-field refocusing—critical factors in maintaining spatial awareness during delicate cortical or vascular dissection.
Key Features
- Full HD (1920 × 1080) LCD projection with LED backlighting delivering 500:1 contrast ratio and consistent luminance across the entire field of view
- Optically integrated mounting within the M530 microscope main body—no external brackets, no mechanical misalignment risk
- Three viewing configurations: left-eye only, right-eye only, or simultaneous binocular overlay—configurable per user preference and ergonomic requirement
- Real-time FL800 fluorescence synchronization with automatic exposure compensation and spectral channel registration
- Native compatibility with IGS systems including BrainLab Curve, Stryker Navina, and Medtronic StealthStation—leveraging DICOM-RT and NIF standard protocols for coordinate mapping
- One-touch activation of overlay toggling and recording via ergonomic handle switch or wireless footswitch—designed for sterile field operation
- Metadata-embedded HD video capture: automatically logs magnification level, working distance, illumination intensity ratio, fluorescence excitation status, and timestamp
Sample Compatibility & Compliance
The OHX module is intended for use in sterile neurosurgical environments and is validated for integration with Leica M530 surgical microscopes equipped with the CaptiView optical architecture. It supports multimodal imaging inputs conforming to SMPTE 292M (HD-SDI), HDMI 1.4, and DICOM PS3.10-compliant video streams. As a Class IIa medical device under EU MDR (2017/745), it complies with EN ISO 13485:2016 for quality management systems and IEC 60601-1:2012 + A1:2020 for electrical safety. The module satisfies FDA 510(k) clearance requirements (K220227) and supports audit-trail functionality per 21 CFR Part 11 when used with Leica LAS X NAV software—enabling traceable, GxP-aligned documentation for clinical trials and regulatory submissions.
Software & Data Management
The OHX module interfaces with Leica LAS X NAV software suite, which provides calibration, registration, and visualization control. Software-based geometric correction ensures sub-pixel overlay accuracy (< 0.3° angular deviation) across all magnification levels (6.3×–40×). Calibration routines include auto-alignment using fiducial markers and manual fine-tuning via digital crosshair adjustment. All recorded HD videos are stored in MP4 (H.264/AVC) format with embedded EXIF-like metadata compliant with DICOM Supplement 154 (Endoscopic Video Storage). Export options include anonymized DICOM-SR reports, time-synchronized multi-stream playback (microscope + navigation + fluorescence), and integration with hospital PACS via DICOMweb or HL7 FHIR interfaces.
Applications
The OHX module is deployed in tumor resection (glioblastoma, meningioma), cerebrovascular surgery (aneurysm clipping, AVM resection), epilepsy surgery (cortical mapping), and skull-base procedures requiring precise anatomical orientation. Its overlay capability enables real-time correlation between preoperative MRI/CT-derived navigation paths and intraoperative tissue morphology. In fluorescence-guided surgery, OHX maintains dynamic FL800 signal-to-noise optimization while preserving native color fidelity—critical for differentiating tumor margins from functional cortex. For teaching hospitals, the dual-eyepiece output allows concurrent visualization by primary surgeon and assistant, supporting structured intraoperative debriefing and competency assessment aligned with ACGME neurosurgery milestones.
FAQ
Does the OHX module require modification of the existing M530 microscope?
No. The OHX is factory-integrated into the M530 main body during manufacturing or installed as a certified field upgrade by Leica-certified service engineers.
Can OHX project images from third-party endoscopes or ultrasound systems?
Yes—provided the source device outputs HD-SDI or HDMI 1.4 signals with stable timing and embedded sync pulses; custom drivers may be required for proprietary protocols.
Is overlay registration performed manually or automatically?
Both options are available: automated registration using IGS-provided transformation matrices, or manual point-based alignment using anatomical landmarks visible in both modalities.
How is data security handled during HD video recording?
All recordings are encrypted at rest (AES-256) and tagged with user ID, procedure ID, and timestamp; export requires role-based authentication per HIPAA and GDPR access controls.
What is the typical calibration interval for clinical use?
Leica recommends biannual verification using the included optical test target and LAS X NAV calibration utility; full recalibration is performed during scheduled preventive maintenance every 12 months.