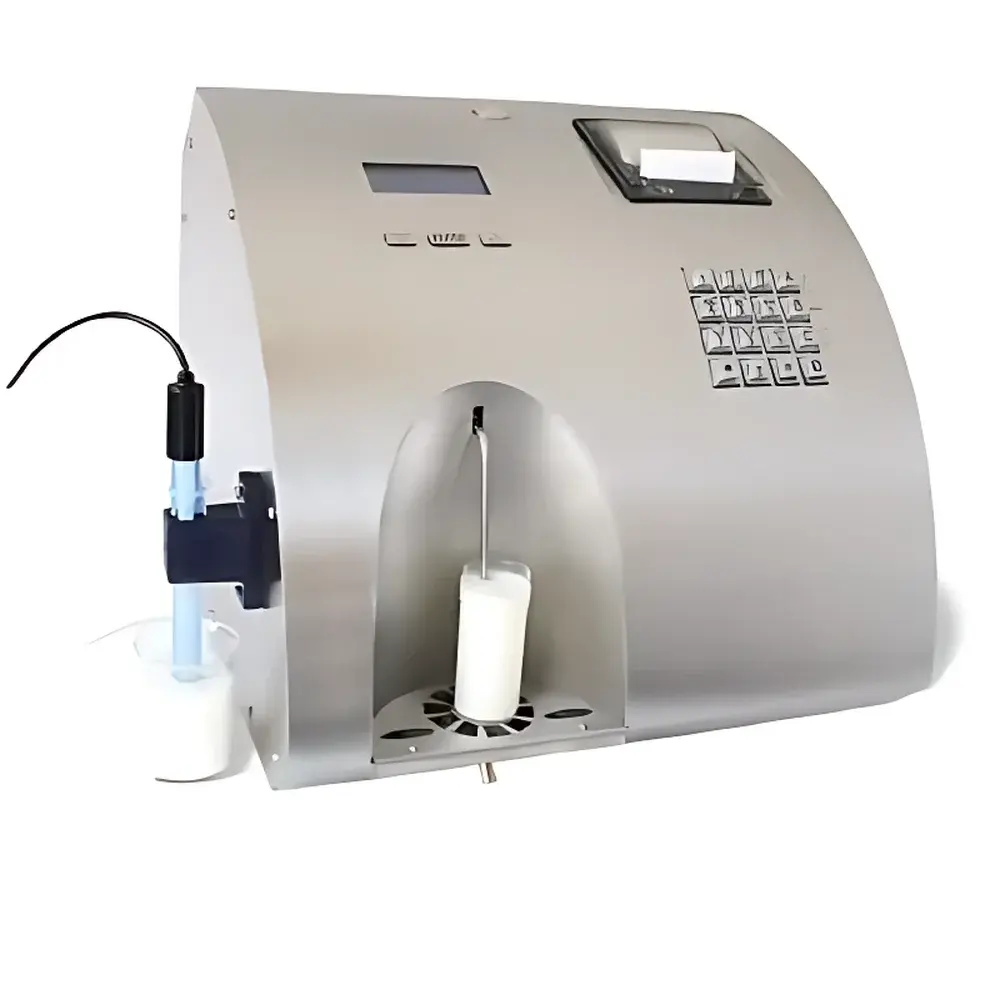
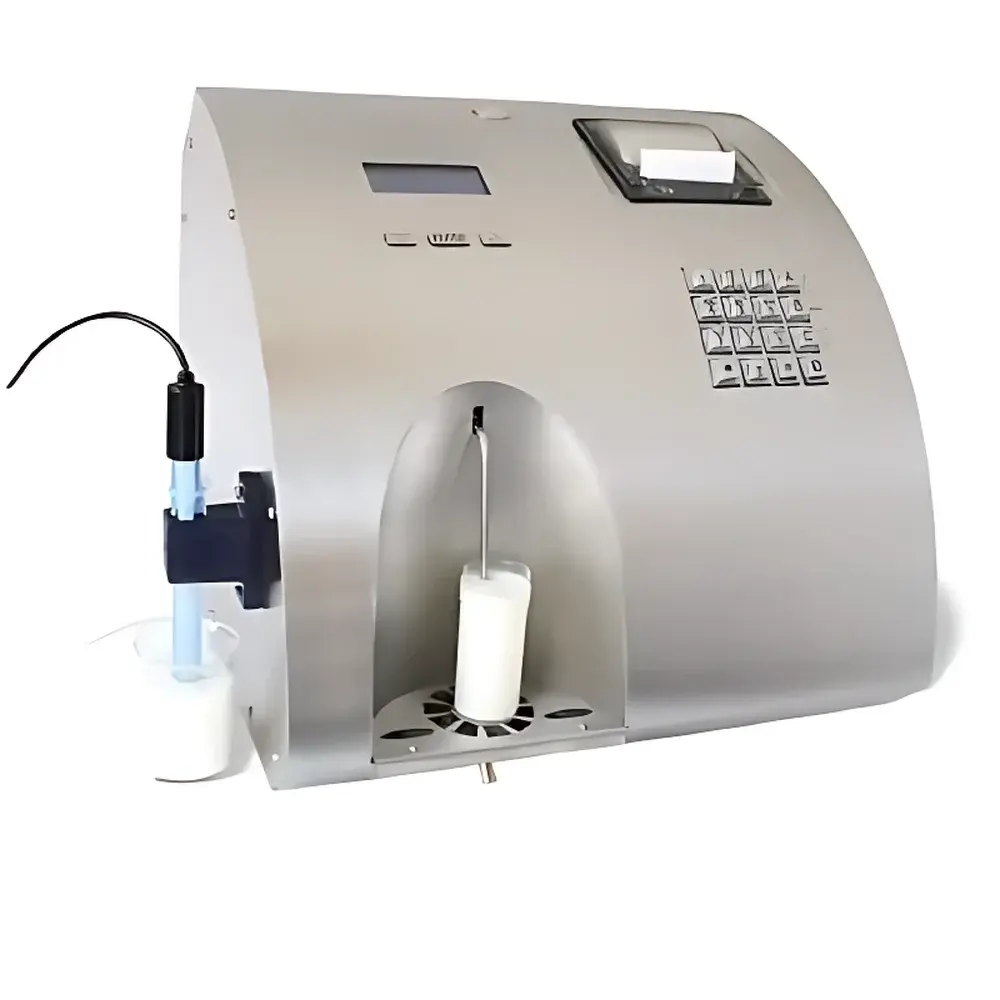
LACTOSCAN LactoScan MCC Dairy Analyzer
| Brand | LACTOSCAN |
|---|---|
| Origin | Bulgaria |
| Model | LactoScan MCC |
| Automation | Fully Automatic |
| Analysis Speed | 120 samples/hour |
| Measurement Range | Fat 0.01–45% |
| Repeatability | Fat ±0.06%, Protein ±0.15%, Lactose ±0.2%, Non-Fat Solids ±0.15%, Ash ±0.01%, Adulteration ±3%, Freezing Point ±0.005 °C, Sample Temperature ±1 °C, Density ±0.3 kg/m³, pH ±0.05, Conductivity ±0.05 mS/cm |
| Resolution | 0.01% |
Overview
The LACTOSCAN LactoScan MCC Dairy Analyzer is a fully automated, near-infrared (NIR) and ultrasonic-based milk composition analyzer engineered for high-throughput, routine quality control in dairy laboratories, collection centers, and processing facilities. Unlike wet-chemistry or chromatographic methods, the LactoScan MCC employs multi-wavelength NIR spectroscopy combined with ultrasonic velocity and attenuation measurements to simultaneously quantify key compositional parameters—including fat, protein, lactose, non-fat solids, ash, freezing point depression, density, pH, and electrical conductivity—without reagents or sample preparation. Its optical-acoustic hybrid architecture enables rapid, non-destructive analysis of raw, pasteurized, or standardized milk, delivering results compliant with ISO 9622:2013 (Milk — Determination of fat, protein, lactose, total solids and other constituents — Guidance on the use of mid-infrared spectrometry) and AOAC Official Method 972.16.
Key Features
- Fully automated operation with integrated peristaltic sampling, automatic rinsing, and self-diagnostic routines—requiring minimal operator intervention.
- Analysis throughput of ≥120 samples per hour, optimized for high-volume milk reception and payment testing environments.
- Robust calibration management system supporting user-defined, multi-laboratory, and factory-installed calibrations traceable to NIST-traceable reference materials.
- Integrated thermal stabilization module ensuring consistent sample temperature control (1–40 °C) during measurement, critical for reproducibility of density, freezing point, and conductivity.
- Reagent-free methodology eliminating hazardous solvents (e.g., ether, chloroform), reducing disposal costs and aligning with ISO 14001 environmental management requirements.
- RS-232 serial interface standard; optional Ethernet and USB connectivity for integration into LIMS or ERP systems under 21 CFR Part 11-compliant audit trail configurations.
Sample Compatibility & Compliance
The LactoScan MCC accepts raw bovine milk, buffalo milk, goat milk, and standardized blends without dilution or homogenization. It accommodates variations in somatic cell count (<1,000,000/mL), microbial load (<10⁶ CFU/mL), and minor thermal treatment (up to HTST pasteurization). All reported performance specifications—including repeatability (e.g., Fat ±0.06%, Freezing Point ±0.005 °C) and resolution (0.01% for compositional values)—are validated per ISO/IEC 17025:2017 accredited procedures. The instrument supports GLP/GMP documentation workflows, including electronic signature capability, version-controlled method storage, and full audit trail logging for all calibration, maintenance, and analysis events.
Software & Data Management
Bundled LACTOSCAN Analytical Suite v4.x provides real-time data visualization, statistical process control (SPC) charting, batch reporting, and customizable certificate generation. Raw spectral data (NIR + ultrasonic) are stored in HDF5 format for retrospective reprocessing and multivariate model refinement. The software supports export to CSV, XML, and PDF formats compliant with EU Regulation (EC) No 853/2004 Annex III Chapter XII for dairy product traceability. Optional FDA 21 CFR Part 11 modules enable role-based access control, electronic signatures, and immutable audit trails required for regulated QC environments.
Applications
- Milk payment testing at collection hubs based on fat, protein, and lactose content per contractual specifications.
- Process monitoring during standardization, homogenization, and thermal treatment to verify compositional consistency.
- Adulteration screening via freezing point depression and conductivity deviation detection (e.g., water addition, urea, melamine).
- Raw material release testing for cheese, yogurt, and infant formula manufacturing according to Codex Alimentarius STAN 206-1999.
- Research applications requiring rapid, high-reproducibility compositional profiling across large sample cohorts (e.g., herd genetics, feed trials, seasonal variation studies).
FAQ
What regulatory standards does the LactoScan MCC comply with?
It meets ISO 9622:2013, ISO 5725 (accuracy and precision), and AOAC 972.16 for milk analysis; optional configurations support 21 CFR Part 11 and EU Annex 11 compliance.
Is method validation support available for laboratory accreditation?
Yes—LACTOSCAN provides IQ/OQ/PQ protocols, uncertainty budgets per GUM (JCGM 100:2008), and technical assistance for ISO/IEC 17025 accreditation audits.
Can the instrument be calibrated using in-house reference materials?
Absolutely—the calibration manager supports user-generated models using certified reference milk samples with documented matrix-matched composition and uncertainty values.
What maintenance is required for long-term operational stability?
Daily rinse cycle, quarterly ultrasonic transducer cleaning, and annual optical alignment verification using supplied alignment standards; no consumables beyond deionized water and detergent.
Is remote diagnostics and firmware update capability supported?
Yes—via secure SSH-enabled Ethernet connection; firmware updates preserve all user calibrations and audit logs without interruption to scheduled analyses.