Tekscan BPMS Human Pressure Distribution Measurement System
| Brand | Tekscan |
|---|---|
| Origin | USA |
| Manufacturer Type | Authorized Distributor |
| Origin Category | Imported |
| Model | BPMS |
| Price Range | USD 14,000 – 70,000 |
Overview
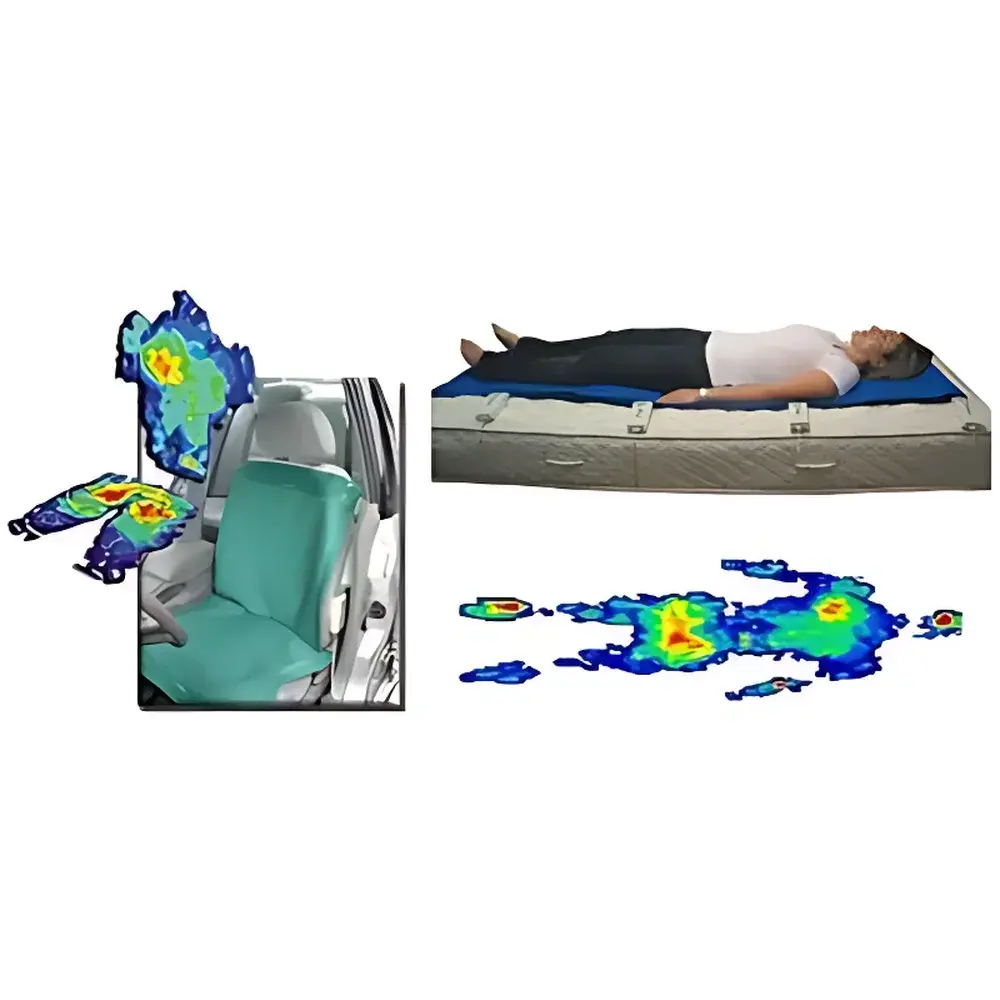
The Tekscan BPMS Human Pressure Distribution Measurement System is an engineered solution for quantitative, spatially resolved interface pressure analysis between the human body and support surfaces. Based on proprietary piezoresistive thin-film sensor technology, the system captures dynamic and static pressure distributions in real time using calibrated capacitive or resistive transduction principles—depending on sensor configuration—enabling high-fidelity mapping of localized load transfer across anatomical contact zones. Designed specifically for biomechanical, ergonomic, and clinical research applications, the BPMS delivers traceable, repeatable measurements compliant with ISO 2631-1 (mechanical vibration and shock — evaluation of human exposure), ASTM F1983 (standard practice for measuring seat interface pressure), and relevant sections of FDA guidance on human factors engineering for medical devices. Its modular architecture supports both laboratory-grade validation studies and field-deployable usability testing in seated, supine, or semi-reclined postures.
Key Features
- High-density flexible sensor arrays: Each pressure-sensitive mat features up to 2,016 discrete sensing elements per sheet, with spatial resolution of 1 sensor per cm² (100 sensors per 100 cm²)
- Scalable acquisition capacity: Supports up to 16,128 total sensing points via synchronized multi-mat operation—ideal for full-body or multi-zone assessment (e.g., seat + backrest + footrest)
- Ultra-thin profile: Sensor thickness of 0.35 mm (0.014 in) ensures minimal interference with natural surface compliance and interface mechanics
- Real-time data capture: USB interface supports sampling rates from 0–100 Hz; PCI-based acquisition extends range to 0–127 Hz for high-speed gait or impact events
- 8-bit pressure resolution: Enables discrimination of pressure increments down to 0.5 kPa (typical calibration range: 0–100 kPa), sufficient for clinical screening and product development thresholds
- Robust software toolkit: Includes real-time 2D heatmaps, interpolated 3D surface rendering (3×3 or 5×5 kernel), center-of-pressure (COP) trajectory tracking, and force-moment vector analysis
- Export-ready outputs: ASCII, CSV, and AVI-compatible video sequences for integration into MATLAB, Python (NumPy/Pandas), or statistical platforms such as JMP or SPSS
Sample Compatibility & Compliance
The BPMS is validated for use with anthropometrically representative human subjects (adults and adolescents), cadaveric models, and anthropomorphic test devices (ATDs). Sensor mats are compatible with standard seating systems (office chairs, automotive seats, wheelchairs), hospital-grade mattresses (foam, air, gel), orthopedic supports, prosthetic interfaces, and bicycle saddles. All hardware and firmware comply with IEC 61000-4 electromagnetic compatibility standards and RoHS Directive 2011/65/EU. Software modules support audit trails and electronic signatures aligned with FDA 21 CFR Part 11 requirements when deployed in GLP/GMP-regulated environments—including clinical trial sites and ISO 13485-certified medical device R&D labs.
Software & Data Management
Tekscan’s Pressure Mapping Software (v9.x or later) provides a deterministic, deterministic acquisition engine with timestamped frame synchronization, configurable trigger logic (hardware/software), and lossless raw data buffering. The platform includes built-in tools for region-of-interest (ROI) masking, temporal averaging, peak pressure localization, contact area quantification, and pressure gradient mapping. Data files retain full metadata (sensor ID, calibration date, subject ID, posture annotation), supporting FAIR (Findable, Accessible, Interoperable, Reusable) data principles. Export options include time-series plots of max/min/mean pressure per ROI, COP displacement vectors, and cumulative histogram analysis—exportable to PDF, PNG, or SVG formats. Remote collaboration is enabled via encrypted project file sharing and version-controlled session archives.
Applications
- Ergonomic seat and cushion design validation for automotive OEMs and Tier-1 suppliers (SAE J2730-compliant testing)
- Pressure ulcer risk assessment in acute care and long-term rehabilitation settings (per NPUAP/EPUAP guidelines)
- Material performance benchmarking: foam compression set, viscoelastic recovery, shear resistance under cyclic loading
- Biomechanical gait and posture analysis in physical therapy and sports science laboratories
- Human factors validation for wearable assistive devices and exoskeleton interfaces
- Academic research in psychophysics of comfort perception and somatosensory feedback modulation
- Durability and fatigue testing of seating systems under ISO 10385-1 (laboratory evaluation of office chair durability)
FAQ
What is the maximum number of sensor points supported in a single acquisition session?
Up to 16,128 sensing elements can be acquired simultaneously across multiple mats using the PCI-based data acquisition module.
Is the system suitable for pediatric or bariatric populations?
Yes—the modular mat configuration allows custom tiling to accommodate diverse anthropometric profiles, including pediatric seating and high-load bariatric support surfaces (calibrated up to 200 kPa upon request).
Can raw sensor data be accessed programmatically for custom algorithm development?
Yes—Tekscan provides documented C/C++ and Python SDKs with low-level memory-mapped I/O access to unprocessed frame buffers and calibration coefficients.
Does the software support automated report generation for regulatory submissions?
Yes—templates for ASTM F1983, ISO 2631-1, and ISO 10385-1 test reports are included, with configurable pass/fail criteria and export to PDF/A-2b compliant archives.
How often does the system require recalibration?
Annual factory recalibration is recommended; field verification using NIST-traceable dead-weight standards is supported via the software’s calibration validation module.