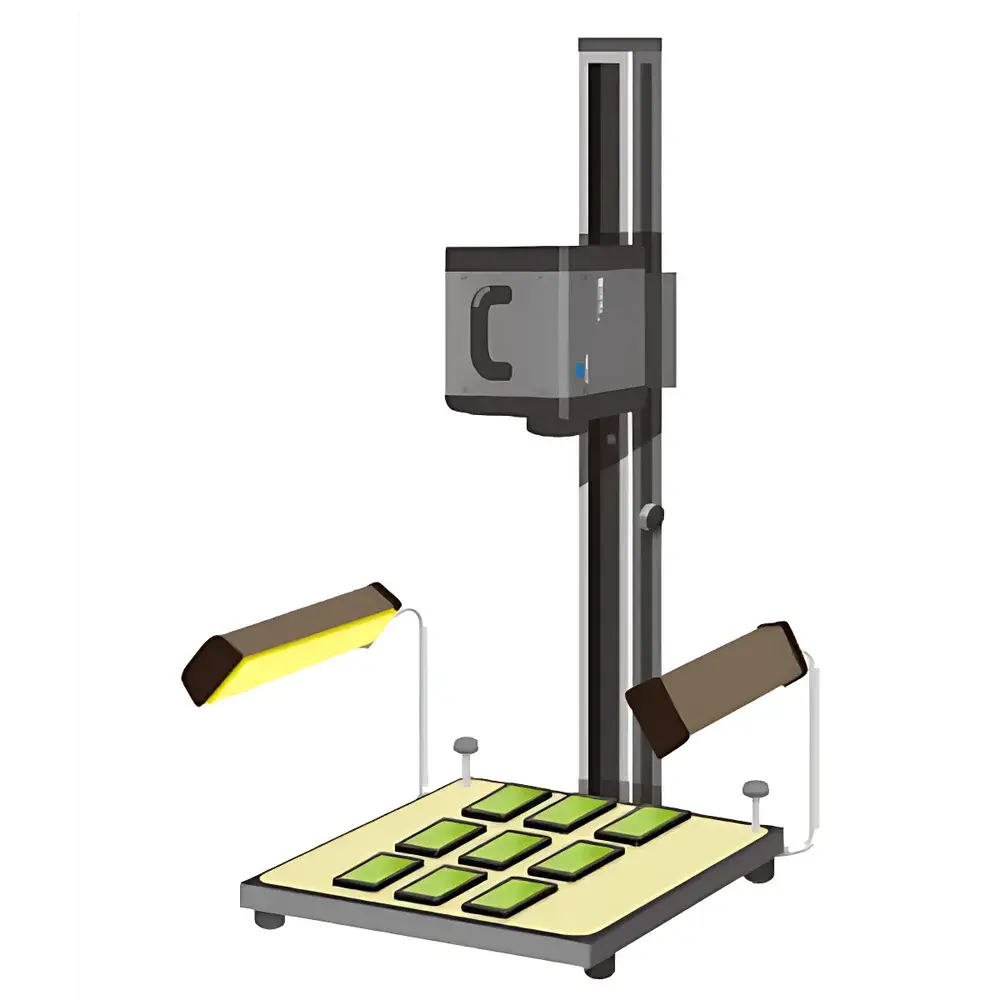
Optometric 16S-300-009 UV Solar Simulator for PMMA Plate Pre-Irradiation in In Vitro SPF Testing
| Brand | Optometric |
|---|---|
| Origin | USA |
| Manufacturer Type | Authorized Distributor |
| Import Status | Imported |
| Model | 16S-300-009 |
| Light Source Type | Xenon Short-Arc Lamp |
| Irradiation Mode | External Vertical Illumination |
| Spectral Range | UVA (320–400 nm), UVB (280–320 nm), UVA+UVB, Full Spectrum AM0/AM1.5 |
| Uniformity | ≤5% (measured across 6-inch square field) |
| Dose Accuracy | ±2% (with PMA2100 radiometer & PMA2101 erythemal detector) |
| Dose Delivery | 4 MED in 23 min (per COLIPA/FDA protocol) |
| Sample Temperature Rise | <0.2 °C during full irradiation cycle |
| Compatible Substrates | HD2, HD6 PMMA plates (2", 4", 6" diameter or square) |
| Optional Configurations | LS1000-4S-009 (4-ft² field, 4 plates), custom 6×6 inch field (9 plates) |
Overview
The Optometric 16S-300-009 UV Solar Simulator is a precision-engineered pre-irradiation light source designed exclusively for in vitro Sun Protection Factor (SPF) testing on polymethyl methacrylate (PMMA) substrates. It replicates the solar ultraviolet spectrum—specifically UVA (320–400 nm), UVB (280–320 nm), and their combined output—in strict accordance with internationally harmonized regulatory protocols. The system employs a stabilized xenon short-arc lamp coupled with calibrated optical filters to deliver spectrally accurate irradiance that conforms to the erythemal action spectrum defined in ISO 24443:2021, FDA Guidance for Industry (2011), COLIPA (now Cosmetics Europe) Method 2011, JCIA JIS T 0001:2017, and Australian Standard AS/NZS 2604:2012. Unlike broadband mercury-based sources, this simulator provides continuous spectral output with minimal temporal drift (<0.5% intensity variation over 1,000 hours), enabling high reproducibility in dose-critical photobiological assays. Its vertical external illumination geometry ensures collimated, uniform exposure across planar PMMA test plates—critical for eliminating edge effects and ensuring compliance with FDA’s requirement for ≤20% spatial non-uniformity (achieved here at ≤5%).
Key Features
- Xenon short-arc lamp with integrated thermal management, delivering stable UV output over extended operational lifetimes (rated >1,500 hours at nominal power)
- Optically filtered beam path producing spectrally validated UVA, UVB, and full-spectrum (AM0/AM1.5) irradiance profiles traceable to NIST-calibrated reference detectors
- Adjustable current-controlled lamp driver enabling precise irradiance modulation (±0.1% resolution) without spectral shift
- Compact benchtop footprint (W × D × H: 32 × 38 × 42 cm) with modular mounting interface for integration into automated SPF testing workflows
- Field uniformity ≤5% across 6-inch square area—exceeding FDA’s 20% tolerance and supporting multi-plate validation per ISO 24443 Annex C
- Thermal load management limiting PMMA substrate temperature rise to <0.2 °C during full 4-MED irradiation (23 min), preventing polymer degradation or sunscreen film migration
- Compatible with PMA2100 digital radiometer and PMA2101 erythemal detector for real-time, weighted dose monitoring and closed-loop automatic endpoint control
Sample Compatibility & Compliance
The 16S-300-009 is validated for use with HD2 and HD6 grade PMMA plates (2″, 4″, and 6″ diameter or square formats), as specified in ISO 24443:2021 Annex B and FDA’s 2011 SPF Final Rule. Its irradiation geometry supports both single-plate and multi-plate configurations—including optional LS1000-4S-009 (4-ft² field, simultaneous 4-plate exposure) and custom 6×6 inch variants (9-plate throughput). All delivered doses are referenced to the CIE erythemal action spectrum (ISO 17166:2019) and reported in Minimal Erythemal Doses (MEDs). System calibration is performed using a NIST-traceable thermopile-based reference standard, and full audit trails—including lamp hours, filter usage, and radiometer calibration dates—are maintained in accordance with GLP principles and FDA 21 CFR Part 11 requirements when paired with compliant software.
Software & Data Management
When operated with the PMA2100 radiometer, the system supports automated dose logging, timestamped irradiance profiling, and CSV-exportable dose history files. The PMA2100 firmware implements IEC 61547-compliant electromagnetic immunity and includes built-in self-diagnostic routines for sensor health verification. All measurement data are stored with embedded metadata: lamp serial number, filter ID, ambient temperature/humidity, operator ID, and calibration expiration date. For regulated environments, optional IQ/OQ documentation packages are available—including installation qualification checklists, operational performance tests against ISO/IEC 17025 criteria, and electronic signature support compatible with laboratory LIMS integration.
Applications
- In vitro SPF determination per ISO 24443:2021, FDA 21 CFR §352.50, and Cosmetics Europe Guideline (2011)
- UVA-PF (Protection Factor) assessment using persistent pigment darkening (PPD) or critical wavelength methods
- Photostability evaluation of organic and inorganic UV filters under controlled spectral stress
- Accelerated aging studies of sunscreen formulations on synthetic skin mimics
- Calibration and validation of UV spectroradiometers and dosimeters used in dermatological research
- Reference irradiation source for inter-laboratory round-robin SPF testing programs coordinated by SCC, IFSCC, or ISO/TC 217
FAQ
What regulatory standards does the 16S-300-009 explicitly comply with?
It meets the spectral, uniformity, and dose accuracy requirements of ISO 24443:2021, FDA Guidance for Industry (2011), Cosmetics Europe (COLIPA) Method 2011, JCIA JIS T 0001:2017, and AS/NZS 2604:2012.
Can this unit be used with non-PMMA substrates?
While optimized for HD2/HD6 PMMA, it may be adapted for other UV-transparent substrates (e.g., quartz, fused silica) provided optical transmission characteristics and thermal expansion coefficients are validated per ISO 24443 Annex D.
Is the PMA2100 radiometer required for regulatory compliance?
Yes—FDA and ISO mandate traceable, erythemally weighted dose measurement; the PMA2100 + PMA2101 combination provides the necessary spectral responsivity and audit-ready data logging.
How often must the lamp and filters be recalibrated?
Lamp output stability is verified quarterly using the PMA2101 detector; optical filters are inspected annually and replaced after 1,200 operating hours or upon spectral deviation >3% from baseline (per ISO/IEC 17025).