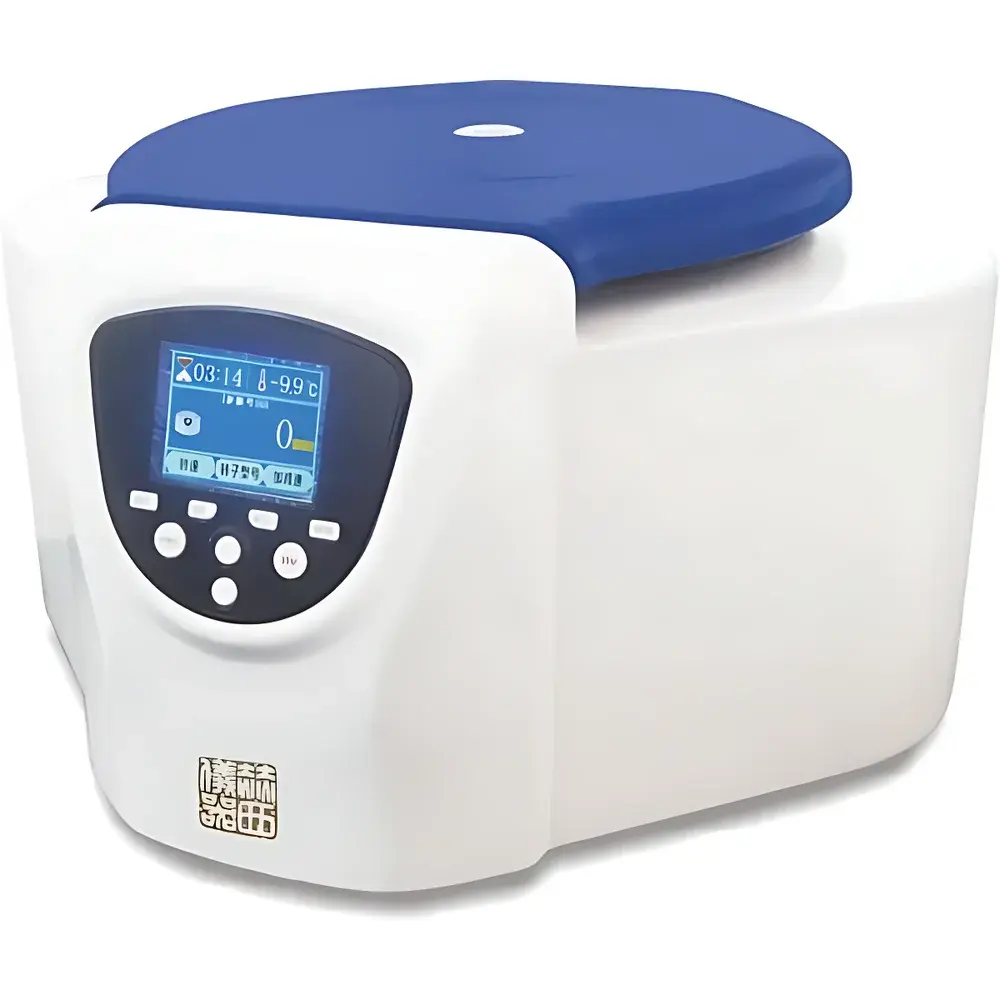
Herexi TDZ4 Cytocentrifuge
| Brand | Herexi |
|---|---|
| Origin | Hunan, China |
| Model | TDZ4 Cytocentrifuge |
| Instrument Type | Benchtop Centrifuge |
| Speed Range | 0–4000 rpm |
| Max. RCF | 1824 × g |
| Max. Capacity | 12 cytology slides (card-type) |
| Rotor Type | Fixed-angle slide rotor |
| Speed Accuracy | ±50 rpm |
| Timer Range | 0–99 h 59 min |
| Noise Level | ≤60 dB(A) |
| Power Supply | AC 220 V, 50 Hz |
| Power Consumption | 120 W |
| Dimensions (L×W×H) | 500 × 450 × 280 mm |
| Net Weight | 20 kg |
| Control System | Microprocessor-based with 4.3″ TFT color touchscreen |
| Motor | Brushless frequency-controlled DC motor |
| Chamber Material | High-grade stainless steel (AISI 304) |
| Safety Features | Automatic imbalance detection, dual mechanical/electronic lid lock, soft-braking deceleration |
| Compliance | Designed to meet general laboratory safety and electromagnetic compatibility (EMC) requirements per IEC 61010-1 |
Overview
The Herexi TDZ4 Cytocentrifuge is a purpose-built benchtop centrifugal device engineered specifically for the preparation of monolayer cytological smears from low-cellularity body fluids. Unlike general-purpose centrifuges, the TDZ4 employs controlled, low-speed sedimentation kinetics—operating within a precise 0–4000 rpm range—to gently deposit cells onto glass slides while preserving cellular morphology and spatial integrity. Its core principle relies on regulated radial acceleration (up to 1824 × g) applied uniformly across a fixed-angle 12-position slide rotor, enabling reproducible cell monolayer formation without clumping, distortion, or nuclear artifact. This makes it indispensable in clinical hematology, transfusion medicine, and diagnostic cytopathology laboratories where accurate morphological assessment under light microscopy is critical—for example, in interpreting TCT (ThinPrep Cytology Test) preparations, detecting malignant cells in pleural effusions, or evaluating cerebrospinal fluid (CSF) for meningeal carcinomatosis.
Key Features
- Benchtop design with compact footprint (500 × 450 × 280 mm), optimized for space-constrained clinical labs and mobile testing units.
- Brushless frequency-controlled DC motor ensures maintenance-free operation, eliminates carbon brush dust contamination, and delivers consistent torque across speed ranges.
- 4.3-inch full-color TFT touchscreen interface with intuitive dual-control (touch + physical buttons) for rapid parameter entry and real-time status monitoring.
- Stainless steel (AISI 304) centrifuge chamber resists corrosion from saline, alcohol, and fixative agents commonly used in cytology workflows.
- Integrated multi-stage vibration damping system and automatic dynamic balance compensation ensure stable operation even with asymmetric slide loading.
- 20 programmable user protocols stored in non-volatile memory; last-used protocol auto-recalls at power-on for workflow continuity.
- Soft deceleration profile minimizes slide displacement during stop phase; lid interlock prevents accidental opening during rotation.
Sample Compatibility & Compliance
The TDZ4 accommodates standard cytology cards (12-slide capacity) compatible with conventional glass slides (75 × 25 mm) and automated staining platforms. It supports all clinically relevant low-viscosity biological fluids—including ascites, pleural effusion, pericardial fluid, synovial fluid, CSF, bronchoalveolar lavage (BAL) fluid, urine sediment, and fine-needle aspiration (FNA) washings. The instrument complies with IEC 61010-1 for electrical safety in laboratory equipment and meets EN 61326-1 for electromagnetic compatibility. While not certified for GLP/GMP-regulated environments out-of-the-box, its programmable protocols, audit-ready parameter logging (via optional RS232/USB export), and traceable run records support alignment with ISO 15189 pre-analytical requirements for cytology specimen processing.
Software & Data Management
The embedded microcontroller firmware enables time-stamped protocol execution logging (date, time, rpm, duration, rotor ID). Although no proprietary PC software is bundled, raw log data can be exported via USB or serial interface for integration into LIMS or quality management systems. The touchscreen interface supports multilingual UI (English default), password-protected administrator mode for calibration and service access, and configurable audible alerts for cycle completion and fault conditions (e.g., imbalance, lid open, overtemperature). All parameters adhere to traceable metrological principles—speed accuracy ±50 rpm is verified against NIST-traceable tachometric references during factory calibration.
Applications
- Routine preparation of Papanicolaou (Pap)-stained cervical cytology specimens in gynecological screening programs.
- Concentration and monolayer deposition of rare tumor cells from serous cavity effusions for immunocytochemical analysis.
- Standardized processing of CSF samples in neurology labs to enhance detection sensitivity for lymphoma or metastatic carcinoma.
- Supporting blood bank serology workflows—including Coombs testing—by preparing standardized red blood cell monolayers for antigen-antibody reaction assessment.
- Research applications in translational oncology, where high-fidelity cell morphology preservation is required prior to next-generation sequencing library prep from cytospin-derived DNA/RNA.
FAQ
What types of cytology slides are compatible with the TDZ4 rotor?
The TDZ4 accepts standard 12-position card-style rotors designed for 75 × 25 mm microscope slides. Adapters for specialized cytology cards (e.g., ThinPrep® vials) are not included but may be sourced separately.
Is the TDZ4 suitable for use in ISO 15189-accredited medical laboratories?
Yes—the device supports documented, repeatable protocols and provides basic run logs. For full compliance, users must validate performance per CLSI EP15-A3 and maintain records of routine preventive maintenance and speed calibration.
Does the TDZ4 require external cooling or ventilation?
No. Its 120 W power consumption and passive thermal management allow safe continuous operation in ambient lab conditions (10–30 °C, <80% RH) without dedicated HVAC support.
Can the centrifuge be integrated into an automated laboratory workflow?
While it lacks native LIS connectivity, the RS232 port enables third-party automation via ASCII command protocol for start/stop control and status polling—subject to custom middleware development.
What maintenance is recommended for long-term reliability?
Monthly cleaning of the rotor chamber with 70% ethanol; annual verification of speed accuracy and brake response time by authorized service personnel using calibrated instrumentation.