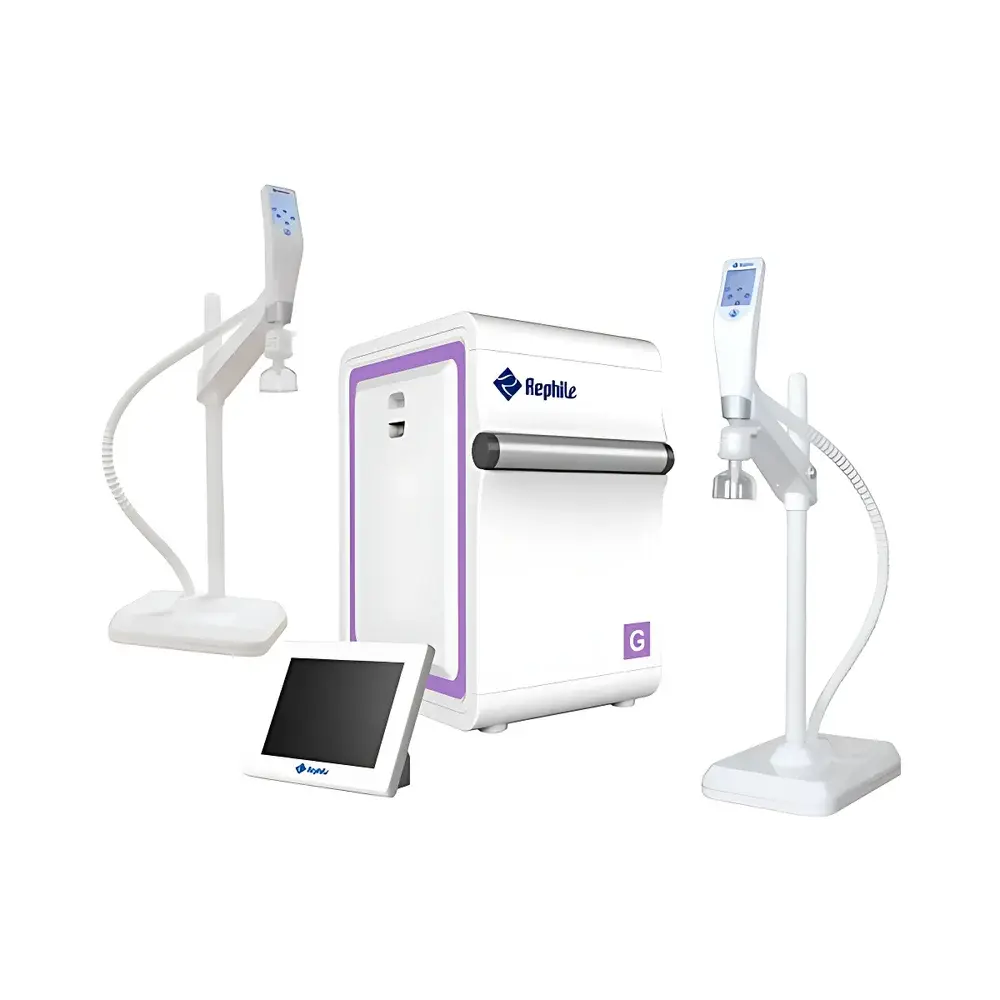
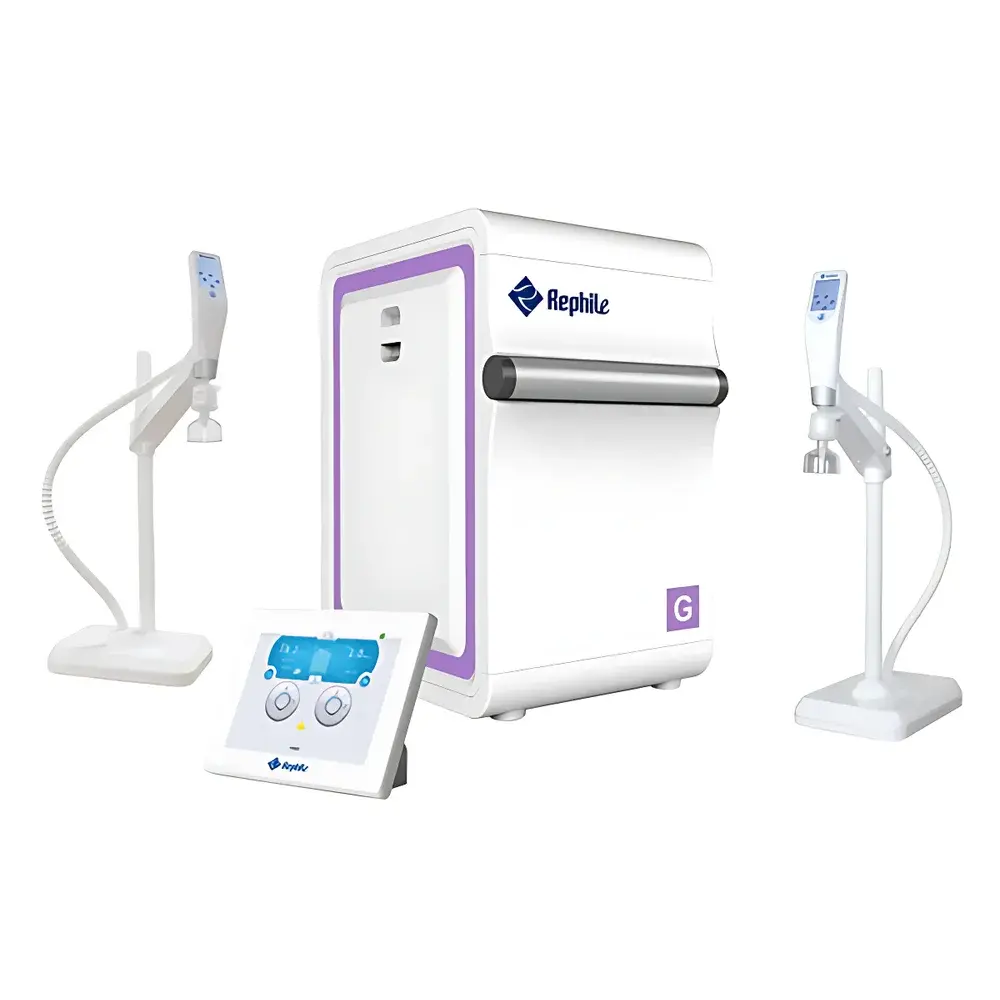
RephiLe Genie G5 Ultra-Pure Water System (Municipal Feedwater Input)
| Brand | RephiLe |
|---|---|
| Origin | Shanghai, China |
| Manufacturer Type | Authorized Distributor |
| Product Category | Domestic |
| Model | Genie G5 |
| Price Range | USD 14,000–42,000 (FOB) |
| Purified Water Grade | ASTM Type I (Ultra-Pure) |
| Resistivity | 18.2 MΩ·cm @ 25 °C (temperature-compensated) |
| Heavy Metals & Soluble Silica | < 0.01 ppb |
| Total Organic Carbon (TOC) | < 5 ppb (µg/L) |
| Endotoxin | < 0.001 EU/mL |
| Particles (>0.1 µm) | Zero particles >0.22 µm detected |
| Bacterial Count | < 0.01 CFU/mL |
| RNase | < 0.5 pg/mL |
| DNase | < 10 pg/mL |
| Flow Rate (Ultra-Pure) | 0–2 L/min (adjustable) |
| Flow Rate (Pure Water) | 5 L/hr (continuous operation) |
| UV Wavelengths | 185 nm & 254 nm dual-lamp system |
| Integrated EDI Module | Electrodeionization with auto-regenerating mixed-bed ion-exchange resin |
| Water Storage Tank | 30 L or 60 L conical-bottom tank with air filter, UV disinfection, anti-overflow safety, and ±1% level accuracy |
| TOC Monitor | On-line, ±1 ppb accuracy |
| Conductivity/Resistivity Sensors | Four independent cells (0.01 cm⁻¹ cell constant, ±0.1 °C temp. sensitivity) |
| Compliance | ISO 9001:2015 certified manufacturing |
Overview
The RephiLe Genie G5 Ultra-Pure Water System is an integrated, single-source solution engineered for laboratories requiring continuous, on-demand production of both Type III (pure) and ASTM Type I (ultra-pure) water from municipal feedwater. It employs a multi-stage purification architecture combining pre-filtration, reverse osmosis (RO), continuous electrodeionization (EDI), dual-wavelength ultraviolet oxidation (185 nm + 254 nm), and final polishing via high-capacity ultrafiltration and sub-micron terminal filtration. The system’s core measurement principle relies on real-time, temperature-compensated resistivity monitoring (using four calibrated 0.01 cm⁻¹ conductivity cells) and online TOC quantification (±1 ppb resolution), ensuring trace-level ionic and organic contaminant control. Designed for mission-critical applications—including HPLC mobile phase preparation, cell culture media formulation, molecular biology assays, and trace metal analysis—the Genie G5 delivers consistent 18.2 MΩ·cm resistivity, <5 ppb TOC, <0.001 EU/mL endotoxin, and undetectable RNase/DNase activity—meeting or exceeding ASTM D1193-20, ISO 3696:1987, CLSI EP21-A, and USP specifications.
Key Features
- Integrated EDI module with mixed-bed ion-exchange resin regenerated continuously by direct current—eliminating chemical regeneration and extending service life.
- Dual-wavelength UV system (185 nm for TOC photo-oxidation; 254 nm for microbial inactivation) housed in quartz sleeves with automatic intensity monitoring.
- Conical-bottom 30 L or 60 L stainless steel water storage tank featuring air filtration (0.2 µm hydrophobic membrane), internal UV-C irradiation, anti-overflow safety valve, and ±1% level sensing accuracy.
- Independent wireless remote dispensing handle with 8-inch capacitive touchscreen, height-adjustable arm, 360° rotation, programmable volume delivery, flow-rate control (0.1–2 L/min), and real-time display of resistivity, TOC, tank level, and consumable status.
- Full RFID authentication for all critical consumables (RO membranes, EDI cartridges, ultra-purification columns, UV lamps, 0.22 µm filters)—preventing counterfeit use and enabling automated lifetime tracking.
- Comprehensive leak detection system with automatic mains shutoff, audible/visual alarm, and safe standby mode upon moisture detection.
- 8-inch industrial-grade main touchscreen interface supporting multilingual operation (English, Chinese, Spanish, French), glove-compatible IP65-rated enclosure, and secure wireless LAN connectivity across host, dispensers, and peripheral modules (e.g., tank disinfection, leak sensor).
Sample Compatibility & Compliance
The Genie G5 is validated for compatibility with sensitive analytical workflows requiring ultrapure water free of ions, organics, nucleases, endotoxins, and particulates. Its output meets stringent international standards including ASTM Type I, ISO 3696 Class 1, and CLSI-recommended purity for PCR, qRT-PCR, NGS library prep, and ELISA. All wetted materials comply with USP Class VI biocompatibility requirements and are non-leaching under long-term exposure. The system supports full 3Q validation (IQ/OQ/PQ) documentation packages aligned with GMP (21 CFR Part 211), GLP (OECD Principles), and ISO/IEC 17025 laboratory accreditation frameworks. Built-in audit-trail functionality logs operator actions, maintenance events, calibration records, and water quality parameters with timestamped, non-erasable entries—fully compatible with FDA 21 CFR Part 11 electronic record requirements.
Software & Data Management
The embedded firmware provides comprehensive data governance capabilities. System-generated logs retain ≥2 years of historical water quality metrics (resistivity, TOC, temperature, flow rate, pressure differentials), consumable usage cycles, and alarm histories. Data export is supported via USB 2.0 port in CSV format—files are digitally signed and tamper-evident. The control software enables scheduled system sanitization protocols, remote diagnostics via encrypted Wi-Fi, and customizable alert thresholds for parameter deviations. All user interactions—including dispenser programming, column replacement confirmation, and service technician login—are captured with digital signature verification to satisfy ALCOA+ data integrity principles (Attributable, Legible, Contemporaneous, Original, Accurate, Complete, Consistent, Enduring, Available).
Applications
- HPLC, UHPLC, and LC-MS mobile phase and blank preparation where low background conductivity and TOC are essential for baseline stability and detection sensitivity.
- Cell culture media, serum-free formulations, and stem cell expansion protocols demanding endotoxin- and nuclease-free water to prevent differentiation artifacts or apoptosis.
- Molecular biology applications including CRISPR-Cas9 editing, RNA-seq, and single-cell genomics—where residual RNase/DNase or trace metals compromise assay fidelity.
- Atomic absorption spectroscopy (AAS), ICP-OES, and ICP-MS sample dilution and standard preparation requiring ppt-level metal contamination control.
- Pharmaceutical QC testing per USP , EP 2.2.43, and JP 17 standards for water for injection (WFI)-grade alternatives in non-sterile processes.
- Calibration laboratories performing traceable conductivity and TOC instrument verification using certified reference materials.
FAQ
What feedwater quality is required for optimal Genie G5 performance?
Municipal tap water meeting local drinking water standards (e.g., EPA 600/R-90/029 or WHO Guidelines) is sufficient. Pre-treatment is handled internally; however, high hardness (>200 ppm CaCO₃) or chlorine residuals >2 ppm may reduce RO membrane lifespan—optional external softening or activated carbon pre-filters are recommended in such cases.
Can the system be validated for GMP compliance?
Yes. RephiLe provides IQ/OQ/PQ protocol templates, executed reports, and raw data sets compliant with Annex 15, EU GMP, and FDA guidance. Full traceability of calibration certificates, sensor verifications, and performance testing is included.
How does the RFID system enhance operational reliability?
Each consumable carries a unique encrypted RFID tag storing batch number, expiration date, installation timestamp, and usage history. The system validates authenticity prior to activation and alerts users when replacement intervals are reached—reducing human error and ensuring regulatory defensibility.
Is remote monitoring supported beyond local Wi-Fi?
The Genie G5 supports optional cloud-based telemetry via secure MQTT over TLS. This enables centralized fleet management, predictive maintenance alerts, and cross-site performance benchmarking without compromising network security or data sovereignty.
What maintenance intervals are recommended for routine operation?
Pre-filters: every 3–6 months (based on feedwater quality); RO membrane: 2–3 years; EDI stack: 3–5 years; UV lamps: annually; ultra-purification columns: 6–12 months (application-dependent). All intervals are tracked automatically and displayed on-screen with color-coded urgency indicators.