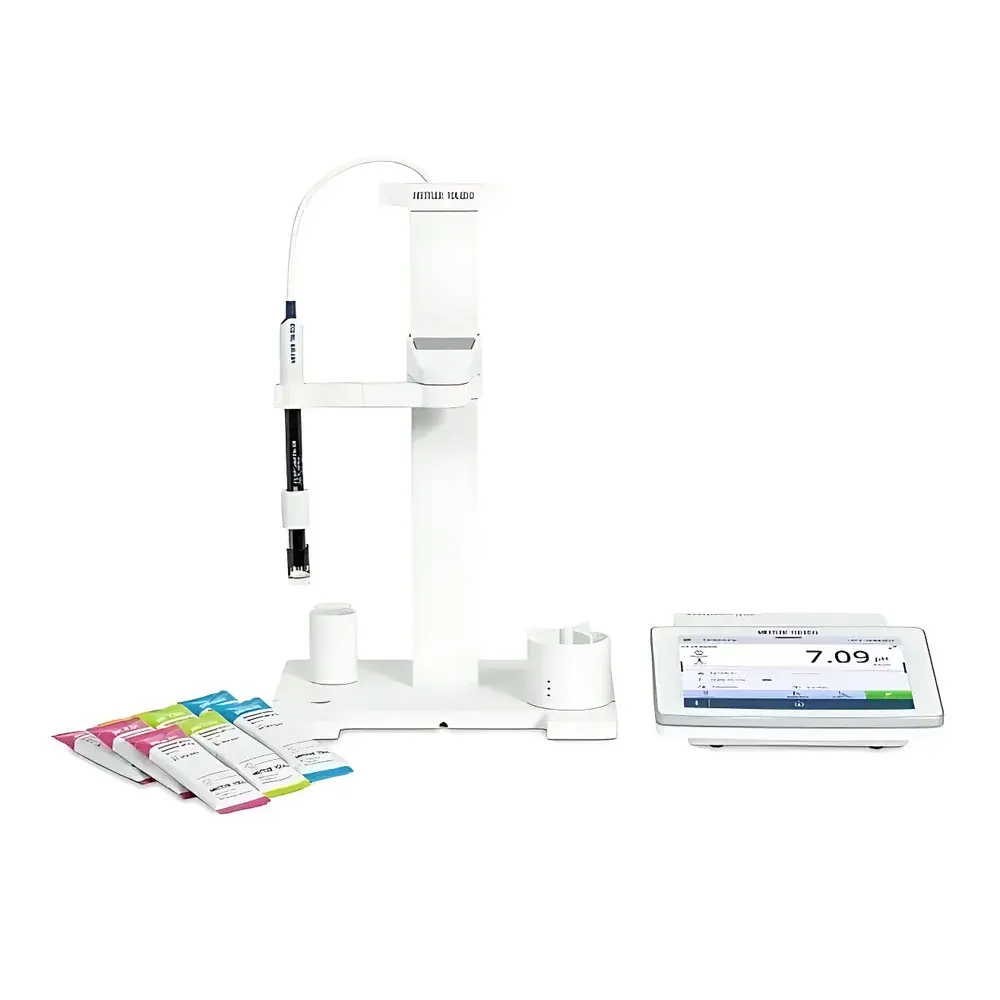
METTLER TOLEDO SevenDirect SD20 Benchtop pH Meter
| Brand | METTLER TOLEDO |
|---|---|
| Origin | Switzerland |
| Instrument Type | Benchtop |
| Model | SD20 |
| Measurement Parameters | pH, mV (ORP), ref mV, Temperature |
| pH Range | –2.000 to 20.000 |
| mV Range | –2000.0 to 2000.0 |
| Temperature Range | –30.0 to 130.0 °C |
| Resolution | 0.001/0.01/0.1 pH (adjustable), 0.1/1 mV, 0.1 °C |
| Accuracy | ±0.002 pH |
| Temperature Compensation | Automatic & Manual |
| Calibration Points | Up to 5-point |
| Buffer Sets | 11 preloaded + 10 customizable |
| Endpoint Modes | Auto, Manual, Time Interval |
| Language Support | 10 languages (English, Chinese, German, French, Italian, Russian, Korean, Spanish, Portuguese, Dutch) |
| Data Storage | 2000 GLP-compliant entries with timestamp, sample ID, electrode ID, user ID |
| Connectivity | USB for PC export and printer output |
| IP Rating | IP54 |
| Display | 7-inch full-color capacitive touchscreen |
| User Management | Two-level access control (Operator/Admin) |
| Compliance | Fully GLP-compliant with audit trail-ready data logging |
Overview
The METTLER TOLEDO SevenDirect SD20 is a high-precision benchtop pH meter engineered for rigorous laboratory environments where regulatory compliance, measurement traceability, and operational robustness are non-negotiable. Built upon METTLER TOLEDO’s legacy in electrochemical metrology, the SD20 implements potentiometric measurement principles per ISO 3696, ASTM E70, and USP , delivering stable, reproducible pH and redox potential (ORP) readings across diverse aqueous and semi-aqueous matrices. Its architecture integrates a high-impedance input amplifier (>1015 Ω), low-drift reference junction design, and intelligent electrode diagnostics—ensuring minimal drift and rapid stabilization even with high-resistance samples or low-ionic-strength solutions. Designed explicitly for QC/QA labs, R&D facilities, and contract testing organizations, the SD20 supports full traceability from measurement initiation through data export, satisfying core requirements of ISO/IEC 17025, FDA 21 CFR Part 11 (when used with LabX software), and EU GMP Annex 11.
Key Features
- 7-inch full-color capacitive touchscreen with glove-compatible operation—functional with latex, nitrile, and cotton gloves—enabling hygienic, contamination-controlled workflows in cleanrooms or biosafety cabinets.
- Intuitive guided interface with context-sensitive on-screen prompts for calibration, measurement, and verification—reducing operator training time and minimizing procedural deviations.
- EasyMix™ electrode holder with vertical adjustment, multi-axis positioning, and integrated magnetic stirrer—ensuring consistent electrode immersion depth and hydrodynamic stability during kinetic measurements.
- Dual-layer user management (Operator/Admin) with password protection and configurable permissions—preventing unauthorized modification of calibration records, method settings, or system configurations.
- IP54-rated enclosure with replaceable protective cover and corrosion-resistant sealed connector housing—providing reliable operation in humid, splash-prone, or chemically aggressive lab environments.
- Auto-buffer recognition with 11 factory-loaded buffer sets and support for 10 custom-defined buffers—including IUPAC, DIN, NIST, and pharmaceutical-grade standards—enabling seamless compliance with pharmacopeial protocols.
- Three endpoint detection modes (Auto, Manual, Time Interval) with adjustable stability criteria—optimized for viscous samples, unstable equilibria, or kinetic studies requiring fixed-interval sampling.
Sample Compatibility & Compliance
The SD20 accommodates a broad spectrum of sample types—from ultrapure water (resistivity >18.2 MΩ·cm) and biological buffers to aggressive acids, alkaline cleaners, and high-salinity industrial effluents. Its InLab® Expert Pro-ISM electrode (included) features a reinforced glass membrane, low-noise ISM (Intelligent Sensor Management) chip, and ceramic junction optimized for clogging resistance—delivering stable response in protein-rich, colloidal, or particulate-laden media. All stored data include mandatory GLP fields: date/time stamp, sample ID, electrode serial number, user login ID, calibration history, and environmental temperature. Audit-ready exports (CSV, PDF, XML) preserve metadata integrity and support retrospective review during internal audits or regulatory inspections (e.g., FDA, EMA, MHRA).
Software & Data Management
Data handling conforms to ALCOA+ principles (Attributable, Legible, Contemporaneous, Original, Accurate, Complete, Consistent, Enduring, Available). The SD20 stores up to 2000 fully annotated measurements locally, with automatic timestamping and electronic signature capture upon calibration or result confirmation. USB export enables direct transfer to secure network drives or LIMS without manual transcription. When paired with METTLER TOLEDO LabX™ PC software (optional), the system supports full 21 CFR Part 11 compliance—including electronic signatures, role-based access control, change history logs, and automated backup—making it suitable for regulated pharmaceutical, food safety, and clinical diagnostics applications.
Applications
- Pharmaceutical quality control: Dissolution testing, buffer preparation verification, stability-indicating assays per ICH Q5C.
- Food & beverage analysis: pH monitoring in dairy fermentation, meat processing, juice pasteurization, and shelf-life validation.
- Environmental testing: Wastewater pH profiling, soil extract analysis, and surface water compliance reporting per EPA Method 150.1.
- Academic & industrial research: Electrode characterization, reaction kinetics, titration endpoint detection, and ion-selective electrode (ISE) calibration.
- Chemical manufacturing: Batch release testing of caustic soda, sulfuric acid, and amine-based formulations under ISO 9001-certified QA systems.
FAQ
Does the SD20 support ISO/IEC 17025-compliant calibration records?
Yes—the instrument automatically logs calibration date, buffer IDs, slope, offset, and electrode impedance, all traceable to user credentials and timestamped to the millisecond.
Can I import custom calibration methods from previous METTLER TOLEDO instruments?
Yes—method files (.mth) created on SevenCompact or SevenExcellence platforms can be imported via USB, preserving all parameter configurations and limit definitions.
Is the touchscreen responsive when wearing sterile surgical gloves?
Yes—the capacitive screen is calibrated for operation with standard medical-grade nitrile and latex gloves, verified per EN 455-2 mechanical performance requirements.
How does the SD20 ensure data integrity during power interruption?
All measurements and calibrations are written to non-volatile memory immediately upon confirmation—no data loss occurs during unexpected shutdowns or battery replacement.
What documentation is provided for IQ/OQ validation?
The instrument ships with a comprehensive Validation Guide (PDF), pre-filled test protocols for installation qualification (IQ) and operational qualification (OQ), and a Certificate of Conformance traceable to METTLER TOLEDO’s Swiss metrology lab.