testo 190 CFR Software – 21 CFR Part 11-Compliant Data Acquisition, Validation & Reporting System for Temperature and Humidity Data Loggers
| Brand | testo |
|---|---|
| Origin | Germany |
| Model | 0554 1901 |
| Channel Capacity | Multi-channel (up to 8 loggers simultaneously programmed/read |
| Regulatory Compliance | Fully compliant with 21 CFR Part 11 (electronic signatures, audit trail, user access control, data integrity safeguards) |
| Deployment | Local PC installation (Windows 7/8/10 or newer, 32/64-bit) |
Overview
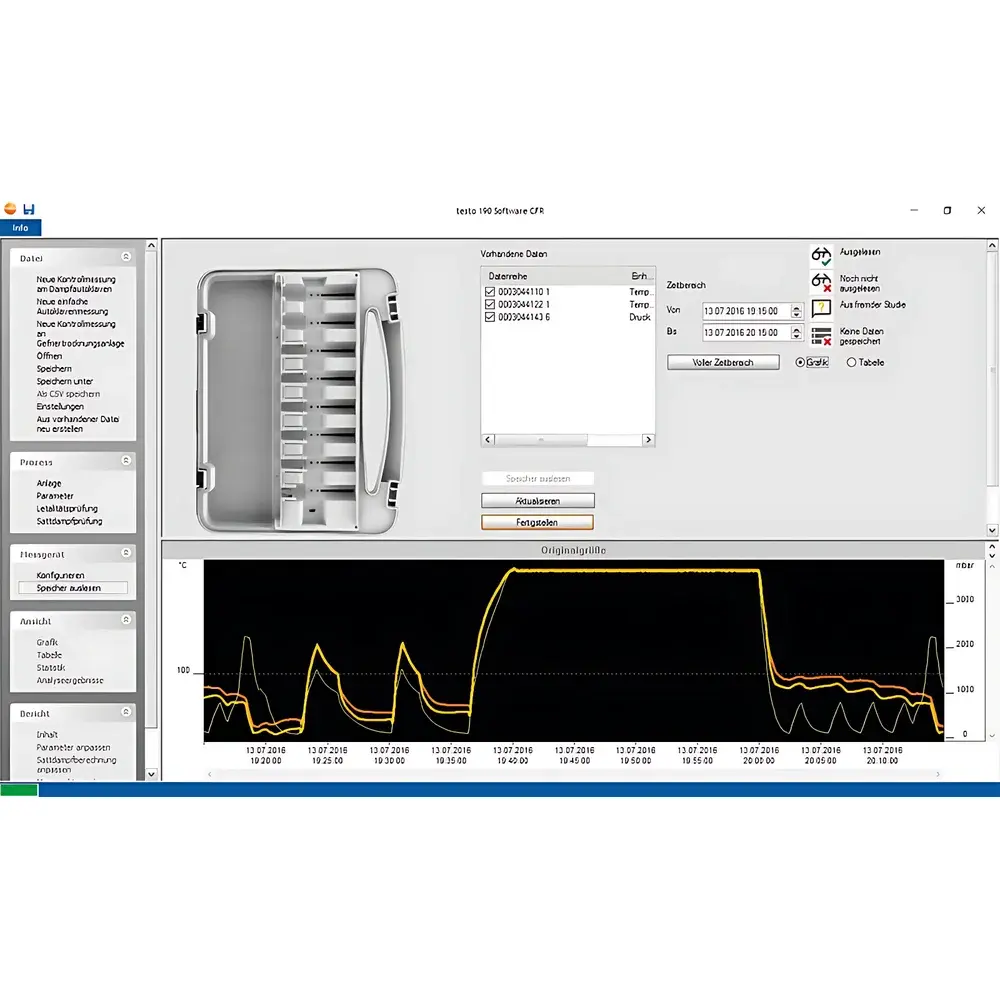
The testo 190 CFR Software is a purpose-built, regulatory-grade application designed exclusively for managing, analyzing, and validating temperature and humidity measurement data collected by testo 190 series CFR-compliant data loggers. Engineered to meet the stringent requirements of pharmaceutical, biotechnology, and medical device manufacturing environments, this software implements a closed-loop validation workflow grounded in the principles of ALCOA+ (Attributable, Legible, Contemporaneous, Original, Accurate, Complete, Consistent, Enduring, Available). It operates on a deterministic, locally installed architecture—eliminating cloud dependencies—to ensure full data sovereignty, reproducibility, and compliance with FDA 21 CFR Part 11, EU Annex 11, and ISO 13485-aligned quality systems. The software supports critical thermal processes including autoclave sterilization, lyophilization (freeze-drying), environmental chamber qualification, cleanroom monitoring, and cold chain validation—where traceable, tamper-evident, and auditable records are non-negotiable.
Key Features
- FDA 21 CFR Part 11 Certification: Built-in electronic signature capability with role-based user authentication, hierarchical password policies, and immutable audit trail logging for all system events—including user login/logout, configuration changes, data export, report generation, and device programming.
- Multi-Logger Synchronization: Simultaneous programming and readout of up to eight testo 190 CFR data loggers via USB interface; scalable support for up to 254 loggers within a single validation protocol execution.
- Integrated F0 Calculation: Automated, real-time calculation of microbial lethality (F0) values per ICH Q5C and USP / guidelines, using configurable Z-value and reference temperature inputs.
- Visual Context Mapping: Geospatial annotation functionality allows users to import and embed photographs of sterilizers, freeze dryers, rooms, or equipment layouts—then overlay time-synchronized temperature/humidity profiles directly onto those images for unambiguous spatial correlation.
- Zero-Export Workflow: All analysis, visualization, and reporting occur natively within the software environment—no reliance on external spreadsheet tools or manual data reformatting, thereby eliminating transcription errors and version control risks.
- Configurable Process Parameters: Automatic interpretation and graphical highlighting of critical process attributes—including temperature band compliance, minimum hold time attainment, maximum equilibration duration, and deviation thresholds—aligned with WHO TRS 961, PDA Technical Report No. 36, and ISO 14644-3.
Sample Compatibility & Compliance
The testo 190 CFR Software is validated exclusively for use with testo 190 series CFR-certified data loggers (e.g., testo 190-T1, 190-T2, 190-H1). These devices feature NIST-traceable calibration certificates, IP65-rated enclosures, and internal memory with write-protection mechanisms. The entire hardware-software ecosystem conforms to GxP requirements under FDA 21 CFR Part 11, EU GMP Annex 11, and ISO/IEC 17025:2017. All generated reports include embedded digital signatures, timestamped audit logs, and cryptographic hash verification to satisfy GLP/GMP inspection readiness criteria. The software itself undergoes periodic re-validation per SOP-QA-027 and supports IQ/OQ/PQ documentation templates aligned with ASTM E2500-13 and ISPE Baseline Guide Volume 5.
Software & Data Management
Installed locally on Windows platforms (7/8/10/11, 32- or 64-bit), the software enforces strict data integrity controls: raw binary logger files remain unaltered and are archived alongside processed datasets; every report export (PDF/A-1a, CSV, or XML) includes embedded metadata verifying origin, authorship, and revision history. Audit trails are stored separately in encrypted SQLite databases with automatic rollover and retention scheduling (configurable from 3 months to permanent). User roles (Administrator, Validator, Operator, Reviewer) enforce segregation of duties; password policies mandate minimum length, complexity, and expiration intervals. All actions are logged with ISO 8601 timestamps, machine identifiers, and contextual event descriptions—fully exportable for regulatory submissions or internal QA review.
Applications
This software serves as the central validation engine for thermal process qualification across regulated industries: steam sterilization cycle development and routine release testing; lyophilization cycle mapping and endpoint determination; warehouse and stability chamber temperature uniformity studies; HVAC system performance qualification (IQ/OQ); cold chain logistics monitoring (e.g., vaccine transport); and environmental monitoring in Grade A/B cleanrooms. Its deterministic architecture ensures consistent interpretation of thermodynamic parameters—supporting root cause analysis during out-of-specification investigations and facilitating CAPA documentation in accordance with ICH Q9 and 21 CFR Part 211.
FAQ
Is the testo 190 CFR Software compatible with non-testo data loggers?
No. It is exclusively validated and supported for use with testo 190 series CFR-compliant loggers.
Does the software require internet connectivity for operation or signature validation?
No. All functions—including electronic signature generation, audit trail recording, and report export—are fully offline-capable after initial license activation.
Can historical data from legacy testo ComSoft versions be migrated into the 190 CFR environment?
Yes—via standardized .tdf file import—but only if original acquisition was performed using CFR-mode-enabled firmware and signed log files.
How is user training and qualification addressed?
The software includes built-in procedural wizards and context-sensitive help; formal qualification (e.g., UAT, PQ) must be performed per site-specific validation protocols referencing Annex 11 §5.3 and FDA Guidance on Computerized Systems in Clinical Trials.
What happens if a data logger’s internal clock drifts during a long-duration validation run?
The software automatically compensates using synchronized time-stamp alignment upon readout, provided the logger’s firmware supports NTP or manual time correction prior to deployment.