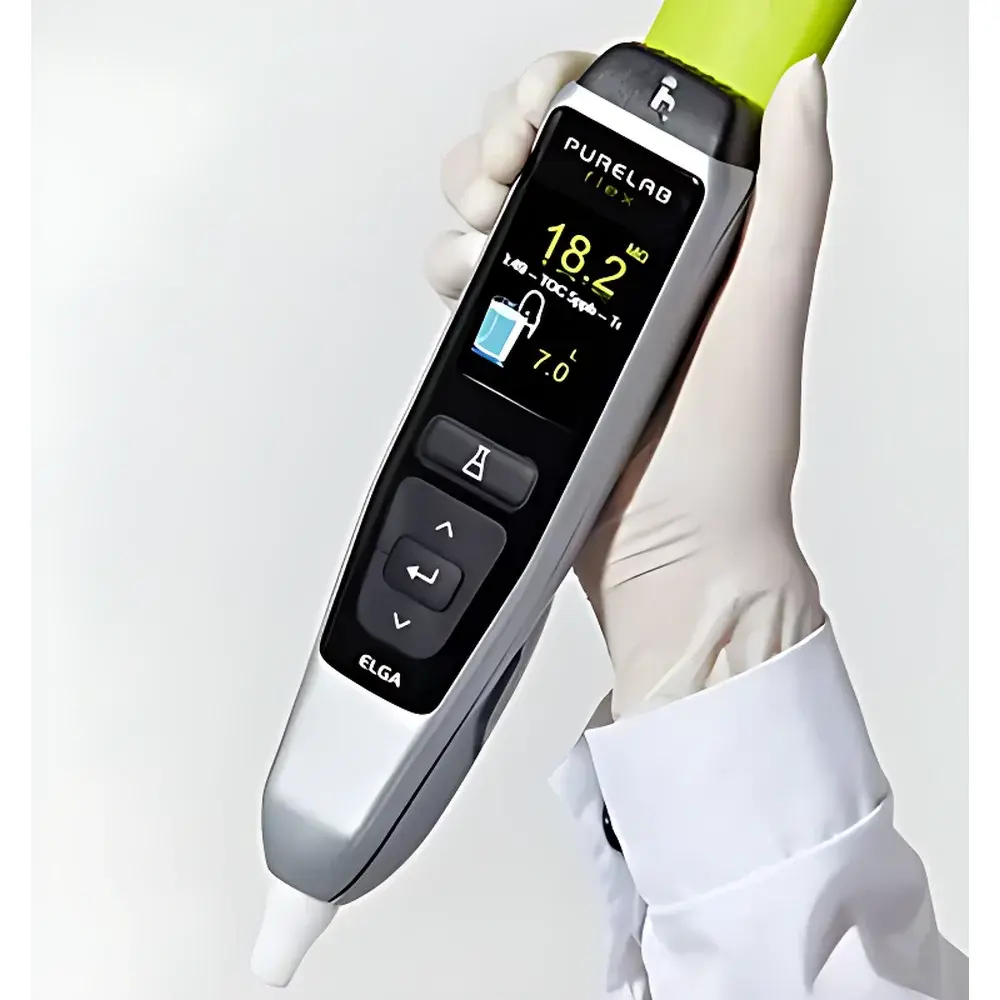
ELGA PURELAB Flex Integrated Pure & Ultrapure Water System
| Brand | ELGA (Veolia) |
|---|---|
| Origin | United Kingdom |
| Manufacturer | Veolia Water Technologies |
| Product Type | Imported Laboratory Water Purification System |
| Model | PURELAB Flex |
| Pure Water Grade | Type I (ASTM D1193-20, ISO 3696:1987 Class 1) |
| Resistivity | 18.2 MΩ·cm @ 25 °C |
| Heavy Metals & Soluble Silica | <0.1 ppb |
| Total Organic Carbon (TOC) | <5 ppb |
| Microbial Count | <0.001 CFU/mL |
| Endotoxins | <0.001 EU/mL |
| Particulates (>0.1 � | m): <1 particle/mL |
| RO Production Rate | up to 10 L/h |
| Dispense Flow Rate | 0.01–2 L/min |
| Integrated Touchscreen Display | Real-time resistivity, conductivity, TOC, temperature, and system status |
Overview
The ELGA PURELAB Flex Integrated Pure & Ultrapure Water System is an engineered solution for laboratories requiring reliable, on-demand access to both Type III (RO) water and ASTM Type I ultrapure water from a single platform. Utilizing a multi-stage purification architecture—including pretreatment, reverse osmosis (RO), electrodeionization (EDI), dual-wavelength UV oxidation (185/254 nm), and final polishing via nuclear-grade mixed-bed ion exchange and ultrafiltration—the system delivers consistent water quality compliant with ISO 3696:1987 Class 1, ASTM D1193-20 Type I, and CLSI EP22-A guidelines. Its closed-loop recirculation design ensures that water at the point-of-use (POU) matches the real-time parameters displayed on the integrated touchscreen—eliminating discrepancies between monitored and delivered water quality. Designed for continuous operation in research, QC, and regulated environments, the PURELAB Flex meets the foundational requirements for trace-level analytical applications such as ICP-MS, HPLC-MS, and cell culture media preparation.
Key Features
- Integrated dual-output architecture: Simultaneous production of RO-grade water (Type III) from the reservoir outlet and ASTM Type I ultrapure water (18.2 MΩ·cm, <5 ppb TOC, <0.001 EU/mL endotoxin) from the ergonomic dispensing handle.
- Human-centered dispensing interface: Pipette-style handle with height-adjustable arm, tactile flow control, and programmable dispensing modes—from single-drop precision to automated volumetric delivery (50 mL to 60 L).
- Real-time, POU-integrated monitoring: In-line sensors for resistivity, temperature, TOC (on Flex 2/3/3+ models), and UV absorbance continuously verify water quality at the dispense point—not upstream or in the reservoir.
- Modular scalability: Supports field-upgradable configurations including optional 254/185 nm UV lamps, ultrafiltration cartridges, and bio-resistant polishing columns to meet evolving application-specific purity requirements.
- Sustainable engineering: Constructed with >70% recycled polymer components; features low-waste RO recovery optimization and energy-efficient EDI operation aligned with ISO 14001 environmental management principles.
- Digital compliance readiness: Embedded data logging with timestamped audit trails, USB export, and network-capable reporting—supporting adherence to GLP, GMP, and FDA 21 CFR Part 11 requirements for electronic records and signatures.
Sample Compatibility & Compliance
The PURELAB Flex is validated for use across life science, analytical chemistry, and clinical diagnostics workflows where water purity directly impacts assay reproducibility and regulatory acceptance. It produces water suitable for molecular biology (DNase/RNase-free operation with optional biofilters), mammalian cell culture (endotoxin <0.001 EU/mL), and high-sensitivity elemental analysis (ICP-MS, GF-AAS). All system components—including tubing, tanks, and sensor housings—are constructed from USP Class VI-certified materials and undergo rigorous extractables testing per ISO 10993-12. The system complies with ISO 9001:2015 quality management standards and is CE-marked under the EU Machinery Directive 2006/42/EC and Electromagnetic Compatibility Directive 2014/30/EU.
Software & Data Management
The PURELAB Flex incorporates an embedded Linux-based control system with a 7-inch capacitive touchscreen interface. All critical water quality parameters are logged at user-defined intervals (1 second to 24 hours) with automatic timestamping, operator ID tagging, and tamper-resistant storage (≥12 months of full-resolution data). Reports can be exported via USB 2.0 in CSV or PDF format, or transmitted over Ethernet to laboratory information management systems (LIMS) using standard HTTP/HTTPS protocols. Optional ELGA Connect software enables remote system diagnostics, predictive maintenance alerts, and centralized fleet monitoring across multi-site installations—facilitating ISO/IEC 17025 accreditation audits and internal quality reviews.
Applications
- High-performance liquid chromatography (HPLC), ultra-high-performance liquid chromatography (UHPLC), and liquid chromatography–mass spectrometry (LC-MS)
- Inductively coupled plasma mass spectrometry (ICP-MS), graphite furnace atomic absorption spectroscopy (GF-AAS), and ion chromatography (IC)
- Mammalian and insect cell culture, stem cell expansion, and CRISPR-Cas9 transfection protocols
- Next-generation sequencing (NGS) library preparation, qPCR, RT-PCR, and microarray hybridization
- Preparation of electrophysiology buffers, ELISA reagents, and radiolabeling solutions
- Calibration standards for trace metal analysis and reference material certification
FAQ
What water grades does the PURELAB Flex produce?
It delivers Type III (RO) water from the reservoir port and ASTM Type I ultrapure water from the dispensing handle—both continuously monitored and validated at the point of use.
Is the system compliant with FDA 21 CFR Part 11?
Yes—when configured with ELGA Connect software and enabled audit trail settings, it supports electronic signature capture, role-based access control, and immutable data archiving required for regulated pharmaceutical and clinical labs.
Can TOC be measured in real time?
TOC monitoring is standard on PURELAB Flex 2, Flex 3, and Flex 3+ models, using a dual-wavelength UV oxidation + conductivity detection method compliant with USP and EP 2.2.44.
How often do consumables require replacement?
Cartridge lifetimes are dynamically calculated based on actual usage metrics (e.g., total volume processed, resistivity decay rate); typical service intervals range from 6 to 12 months depending on feedwater quality and daily demand.
Does the system support integration with central water networks?
Yes—the PURELAB Flex can operate as a standalone unit or as a polishing node fed by a central RO loop, supporting distributed Type I water delivery across multiple lab benches via dedicated POU points.