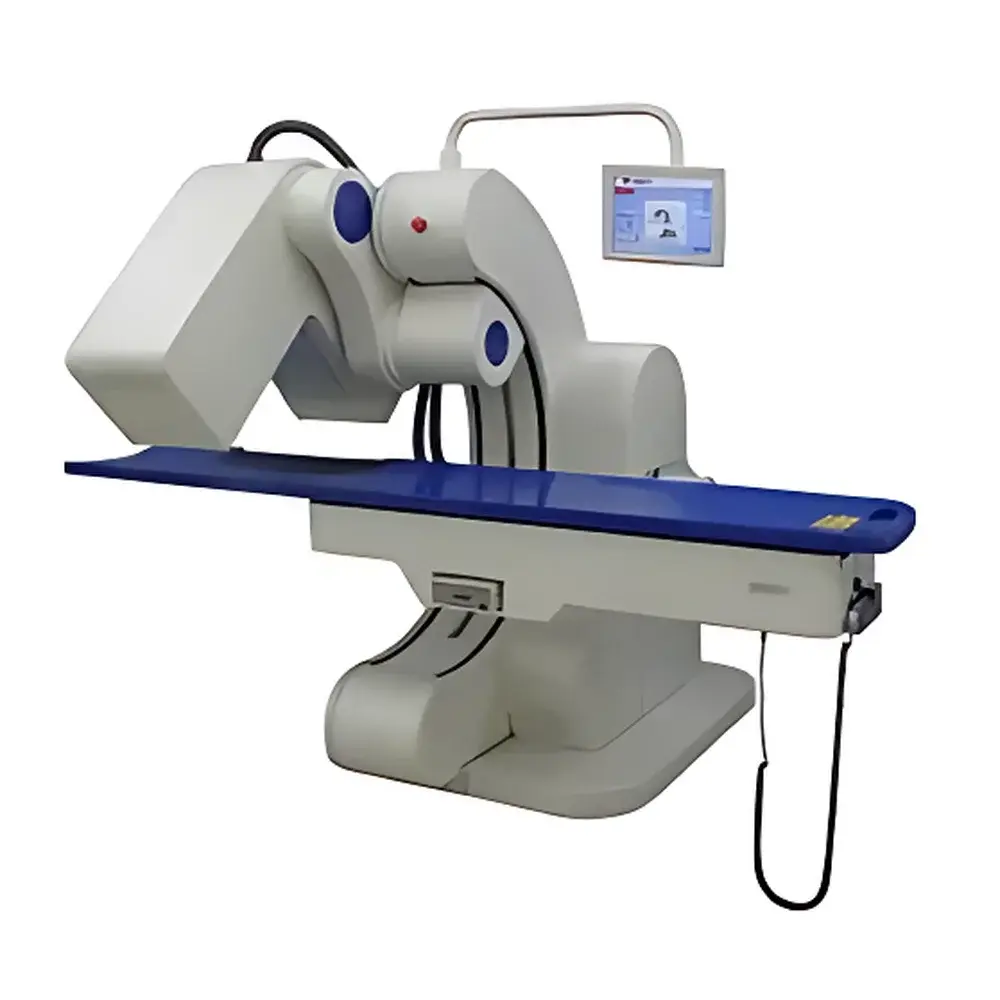
Hamamatsu BHP6603 Fixed-Angle Dual-Head SPECT System
| Brand | Hamamatsu Photonics (BHP) |
|---|---|
| Origin | Hebei, China |
| Manufacturer Type | Authorized Distributor |
| Country of Origin | China |
| Model | BHP6603 |
| Instrument Type | Radionuclide Imaging System |
| Power Supply | 220 V AC ±10%, 50–60 Hz, 8 A, Single-Phase, 1.7 kVA |
| Thermal Dissipation | 5100 kJ/h |
| Operating Temperature | 15–30 °C (59–86 °F), Max Temp. Ramp Rate: ±3 °C/h |
| Relative Humidity | 20–70 % RH |
| System Weight | 1500 kg (3307 lb) |
Overview
The Hamamatsu BHP6603 Fixed-Angle Dual-Head SPECT System is a dedicated small-animal and clinical cardiac single-photon emission computed tomography platform engineered for high-fidelity radionuclide imaging under controlled laboratory and regulated clinical environments. Utilizing dual gamma camera heads mounted at fixed, optimized angular positions—configured specifically for cardiac-centric acquisition—the system implements parallel-hole collimation with energy discrimination to detect ⁹⁹mTc, ¹²³I, and other common diagnostic radionuclides (70–300 keV range). Its mechanical architecture follows the principles of circular-orbit and elliptical-orbit acquisition geometries, with the latter conforming more closely to human torso anatomy to minimize source-to-detector distance during myocardial studies. The system complies with IEC 61674:2017 (Medical electrical equipment – X-ray equipment for radiography and radioscopy) for detector performance and radiation safety, and meets essential requirements of FDA 21 CFR Part 1020.30 (Diagnostic x-ray systems) as adapted for gamma camera-based SPECT instrumentation.
Key Features
- Dual-head fixed-angle geometry optimized for myocardial perfusion imaging—eliminates mechanical gantry rotation delays and enhances temporal stability during dynamic or gated acquisitions.
- Open-frame patient access design reduces claustrophobia and improves workflow ergonomics; minimum required installation footprint is 3 m × 3 m.
- Ergonomic motorized examination table with variable-height adjustment (lowest position enables seated patient transfer) and concave, widened tabletop surface for enhanced patient comfort and anatomical alignment.
- Eccentric table positioning ensures equal radial distance from both detectors to the cardiac centroid—critical for uniform sensitivity and quantitative accuracy in attenuation-corrected reconstructions.
- Elliptical acquisition trajectory—programmable via software—approximates thoracic contour, shortening effective source–detector distance by up to 15% compared to standard circular orbits, thereby improving spatial resolution and count sensitivity.
- Redundant safety architecture: hardware emergency stop button, touch-sensitive detector guard zones, software-initiated acquisition halt, and manual mechanical override for table retraction during power failure.
- Integrated dual-control interface: capacitive touchscreen console for real-time preview and protocol selection, plus physical hand-held control unit for operator mobility during patient setup.
Sample Compatibility & Compliance
The BHP6603 supports both human cardiac imaging (adult and pediatric) and preclinical small-animal applications—including murine and rat models—when used with appropriate collimators and calibration phantoms. It accommodates standard NEMA NU 2–2018 test objects for spatial resolution, sensitivity, and scatter fraction validation. The system satisfies regulatory prerequisites for clinical deployment in the U.S. (FDA 510(k) cleared), Canada (Health Canada Medical Device License), and China (NMPA registration, formerly SFDA). It adheres to ISO 13485:2016 quality management standards for medical device distribution and supports audit-ready documentation for GLP-compliant pharmacokinetic and biodistribution studies. Radiation shielding design conforms to local regulations (e.g., NRC 10 CFR Part 35, IAEA Safety Standards Series No. SSG-46).
Software & Data Management
Acquisition, reconstruction, and quantification are managed through Hamamatsu’s proprietary SPECTVision™ Suite v3.2+, which includes iterative OSEM reconstruction with CT-based or model-based attenuation correction, motion correction algorithms, and ECG-gated functional analysis (LVEF, wall motion, TPD scoring). DICOM 3.0 compliance ensures seamless integration with PACS, RIS, and EMR systems. Audit trail functionality logs all user actions—including parameter changes, reconstruction settings, and report generation—in accordance with FDA 21 CFR Part 11 requirements for electronic records and signatures. Raw projection data export (Interfile format) enables third-party processing using PMOD, ASPIRE, or custom MATLAB/Python pipelines.
Applications
Primary clinical applications include myocardial perfusion scintigraphy for detection of ischemia and infarction, risk stratification in coronary artery disease, functional assessment pre- and post-CABG or PCI, and evaluation of ventricular dyssynchrony. In translational research, the system supports longitudinal monitoring of tracer biodistribution in oncology (e.g., ¹¹¹In-octreotide), neurology (⁹⁹mTc-HMPAO cerebral blood flow), and cardiology (⁹⁹mTc-tetrofosmin uptake kinetics) studies. Its compact form factor and low infrastructure demands make it suitable for satellite imaging centers, university core facilities, and contract research organizations conducting IND-enabling nonclinical imaging.
FAQ
Is the BHP6603 compatible with quantitative SPECT/CT fusion?
Yes—when integrated with an external CT scanner (e.g., Siemens LANTID or GE Optima CT540), the system supports rigid and deformable image registration via DICOM-RT structure sets and vendor-neutral attenuation maps.
What collimators are supported?
Standard LEHR (low-energy high-resolution), LEHS (low-energy high-sensitivity), and MEGP (medium-energy general-purpose) parallel-hole collimators are validated; pinhole collimators for rodent imaging are available as optional accessories.
Does the system support dynamic acquisition protocols?
Yes—frame durations from 1 s to 60 s per frame are programmable, with list-mode acquisition capability for retrospective gating and kinetic modeling.
Can acquisition parameters be locked for multi-site clinical trials?
Yes—protocol templates with immutable acquisition matrices, zoom factors, and energy windows can be deployed across networked installations with version-controlled configuration files.
What service and support options are available internationally?
Hamamatsu Photonics provides remote diagnostics, on-site preventive maintenance contracts, and certified engineer training programs aligned with ISO/IEC 17025 calibration traceability requirements.