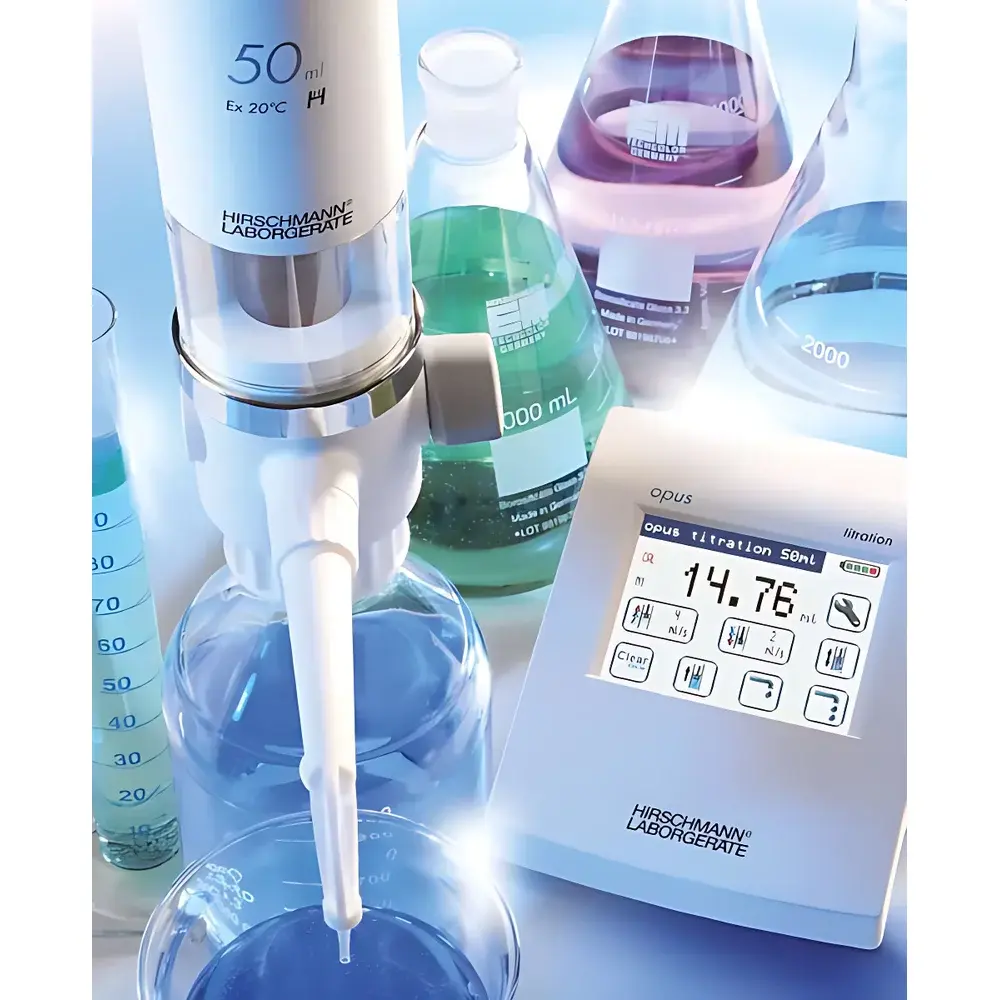
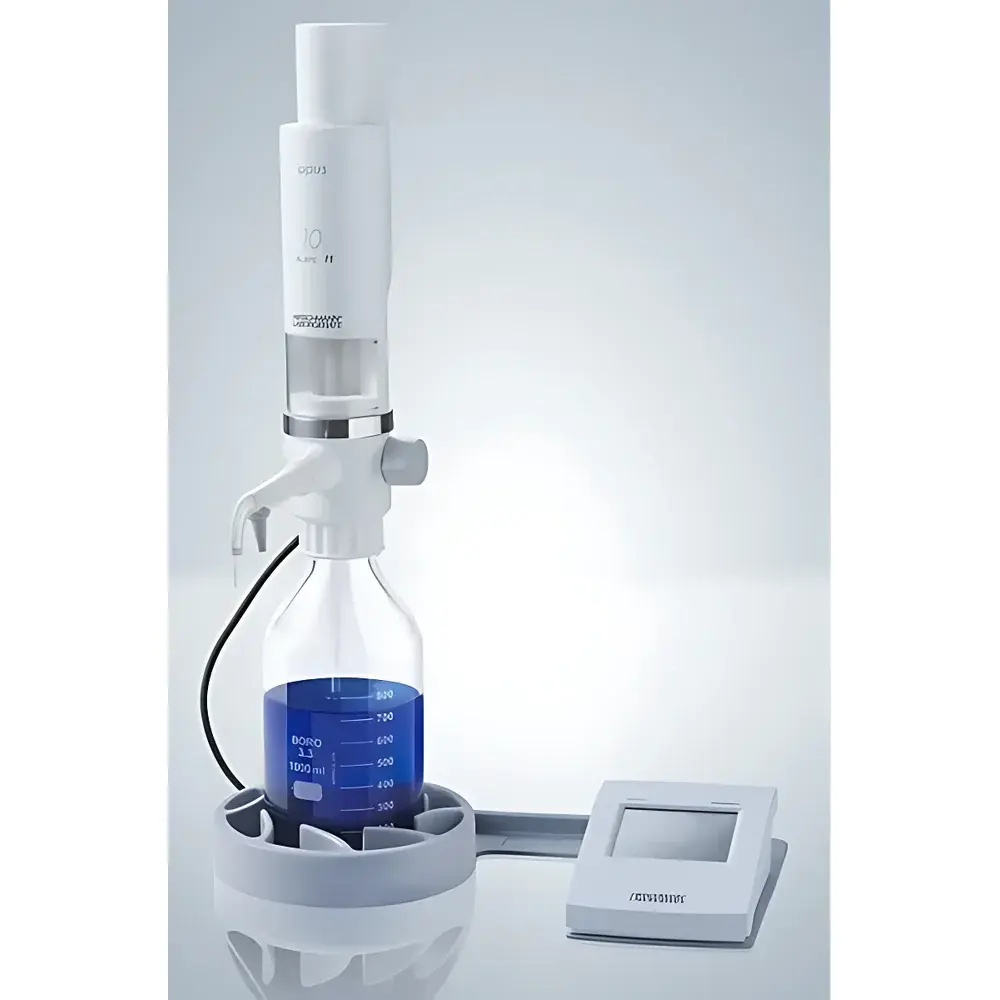
Hirschmann Opus Electronic Burette
| Brand | Hirschmann (Germany) |
|---|---|
| Origin | Germany |
| Model | Opus Titration |
| Volume Range Options | 10 mL / 20 mL / 50 mL |
| Minimum Dispensing Increment | 0.001 mL (10 mL), 0.01 mL (20 mL & 50 mL) |
| Accuracy (A) | ≤0.2% |
| Coefficient of Variation (CV) | ≤0.07% (10 mL & 20 mL), ≤0.05% (50 mL) |
| Wetted Materials | ECTFE, FEP, PTFE, borosilicate glass |
| Compliance | ISO 8655-3, DIN EN ISO 9001, GLP/GMP-ready, FDA 21 CFR Part 11–compatible software architecture |
Overview
The Hirschmann Opus Electronic Burette is a precision-engineered, motor-driven liquid dispensing instrument designed for volumetric titration in regulated laboratory and production environments. Unlike manual burettes or semi-automated alternatives, the Opus operates on closed-loop stepper motor control with optical position feedback, enabling reproducible, operator-independent delivery of titrant with traceable volume resolution down to 0.001 mL (for the 10 mL version). Its measurement principle adheres to the gravimetric and volumetric calibration framework defined in ISO 8655-3 for piston burettes, ensuring metrological traceability to national standards. The device is not a standalone analytical instrument but a critical component in quantitative chemical analysis workflows—particularly acid-base, redox, complexometric, and precipitation titrations—where repeatability, audit readiness, and reagent integrity are non-negotiable.
Key Features
- Motor-driven precision dispensing with real-time positional feedback and automatic endpoint detection via programmable stop conditions.
- Multi-mode operation: touch-screen TFT interface (800 × 480 px), external footswitch/mouse control, and RS-232/USB host integration for PC-based automation (e.g., via LabVIEW or custom LIMS scripts).
- Nine user-definable titration methods stored locally—including pre-titration, fast/slow approach modes, and pause-on-equivalence-point logic—enabling method standardization across shifts and operators.
- Chemically inert fluid path constructed entirely from fluoropolymers (ECTFE, FEP, PTFE) and Class A borosilicate glass; compatible with aggressive solvents, strong acids (excluding HF), and oxidizing agents.
- Integrated safety features: factory-calibrated pressure-relief valve, optional desiccant drying tube for hygroscopic reagents, and brown-window variant for light-sensitive titrants (e.g., iodine, cerium(IV) sulfate).
- Self-priming pump mechanism with automated air removal—eliminating manual rinsing cycles and reducing procedural variability.
Sample Compatibility & Compliance
The Opus supports aqueous, organic, and mixed-phase titrants within its specified viscosity and vapor pressure limits (up to 500 mPa·s, <10 kPa at 20 °C). All wetted components comply with USP , EU Pharmacopoeia 3.1.1, and ISO 10993-5 for material biocompatibility. Each unit undergoes individual gravimetric verification per ISO 8655-3 Annex C and ships with a unique serial-numbered Certificate of Conformance—including full-volume error curves, CV data, and temperature-compensated calibration records. These documents are structured to integrate directly into ISO 9001:2015 quality management systems and satisfy GLP (21 CFR Part 58) and GMP (EU Annex 11, FDA Guidance for Industry) documentation requirements. Audit trails, electronic signatures, and version-controlled method storage are supported through optional Hirschmann TitraSoft software.
Software & Data Management
TitraSoft (v4.x or later) provides FDA 21 CFR Part 11–compliant data acquisition, including electronic signatures, role-based access control, and immutable audit logs for all dispensing events, parameter changes, and calibration actions. Raw volume/time stamps, endpoint recognition timestamps, and environmental metadata (ambient temperature, humidity if sensor-equipped) are embedded in .CSV and .XML exports. The software supports direct import into ELN platforms (e.g., LabArchives, Benchling) via standardized API endpoints and enables batch reporting aligned with ISO/IEC 17025 clause 7.8. Local device memory retains ≥1,000 titration records with timestamp, operator ID, method name, and result summary—even during network outages.
Applications
- Quality control laboratories performing pharmacopeial titrations (USP , EP 2.2.20, JP 6.05) for raw material assay and finished product release.
- Environmental testing labs conducting COD, alkalinity, chloride, and hardness determinations per ASTM D1252, ISO 9963-1, and EPA 130.1.
- Food & beverage QA/QC for acidity (titratable acidity), SO₂, and preservative quantification under AOAC 947.05 and ISO 750.
- Research settings requiring high-reproducibility reagent addition in kinetic studies, catalyst screening, or pH-stat experiments.
- In-process control in fine chemical synthesis where stoichiometric addition must be documented for regulatory submissions.
FAQ
Does the Opus require periodic recalibration by the user?
No—each unit is factory-calibrated with NIST-traceable weights and certified per ISO 8655-3. Users perform only performance verification (e.g., gravimetric check) at defined intervals per their internal SOPs; full recalibration is performed exclusively by authorized Hirschmann service centers.
Can the Opus be integrated into an existing LIMS or MES system?
Yes—via RS-232 or USB CDC virtual COM port using ASCII command protocol (documented in Hirschmann’s “Opus Communication Interface Manual”). Custom drivers are available for Siemens SIMATIC, Rockwell FactoryTalk, and Thermo Fisher SampleManager.
Is the 10 mL version suitable for micro-titrations requiring sub-microliter resolution?
No—the minimum increment is 1 µL (0.001 mL), but volumetric accuracy specifications apply only down to 10% of nominal capacity (i.e., ≥1 mL for the 10 mL model). For true sub-100 µL work, a dedicated micro-burette or syringe pump is recommended.
How is reagent evaporation prevented during extended idle periods?
The optional drying tube (Cat. No. 958209020) connects inline between reagent bottle and burette inlet, maintaining inert gas blanket (N₂ or Ar) over the titrant reservoir—critical for volatile bases like NaOH in ethanol or thiosulfate solutions.
What validation documentation is provided upon delivery?
Each unit includes: (1) Individual Certificate of Conformance with full calibration report, (2) ISO/IEC 17025-accredited test certificate (optional add-on), (3) Material Declarations per REACH/ROHS, and (4) Installation Qualification (IQ) checklist compliant with GAMP5.