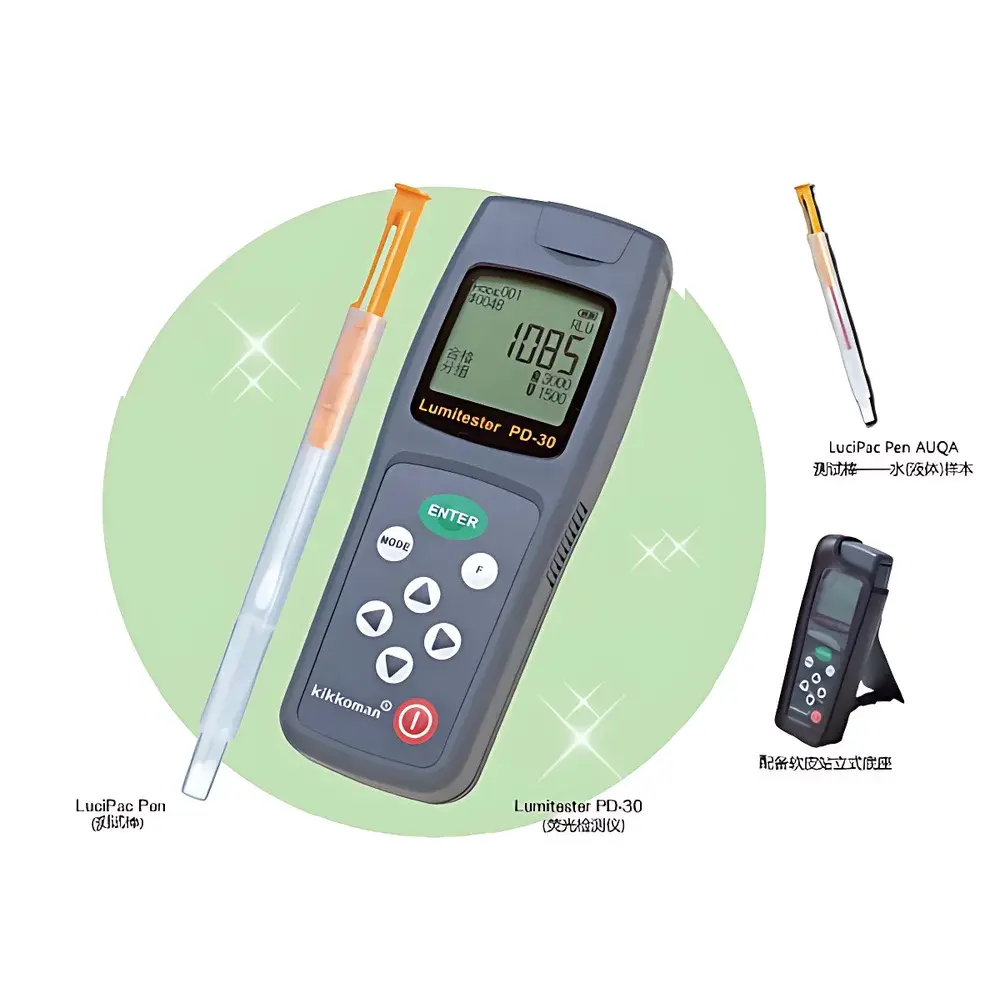
Kikkoman PD-30 Advanced ATP Bioluminescence Detector
| Origin | Japan |
|---|---|
| Manufacturer Type | Authorized Distributor |
| Origin Category | Imported |
| Model | PD-30 |
| Price Range | USD 1,400 – 2,800 |
Overview
The Kikkoman PD-30 Advanced ATP Bioluminescence Detector is a precision-engineered, handheld luminometer designed for rapid, quantitative hygiene monitoring in food production, healthcare, hospitality, and public infrastructure environments. It operates on the patented A³ Method (Adenylate Amplification & Amplified ATP Detection), a proprietary enzymatic cascade technology developed by Kikkoman Corporation. Unlike conventional ATP-only assays, the A³ Method simultaneously quantifies ATP, ADP, and AMP—three interconvertible adenylates present in all living cells and organic residues. By enzymatically converting ADP to ATP via pyruvate kinase (PK) and AMP to ATP via phosphoenolpyruvate carboxykinase (PEPCK), the system amplifies total adenylate content prior to luciferase-mediated luminescence detection. This approach significantly increases assay sensitivity and reduces false negatives caused by sample-dependent adenylate distribution shifts—particularly critical when evaluating heat-treated, fermented, or aged biological residues where ATP degrades preferentially to ADP/AMP. The PD-30 delivers results in ≤10 seconds with reproducible relative light unit (RLU) output calibrated against Kikkoman’s traceable reference standards.
Key Features
- Proprietary A³ Method enables simultaneous detection of ATP, ADP, and AMP—enhancing detection reliability across diverse residue types including thermally processed foods, biofilms, and desiccated microbial debris.
- Integrated solid-state photomultiplier tube (PMT) with auto-ranging gain control ensures linear response across 0–9,999,999 RLU, eliminating manual range selection errors.
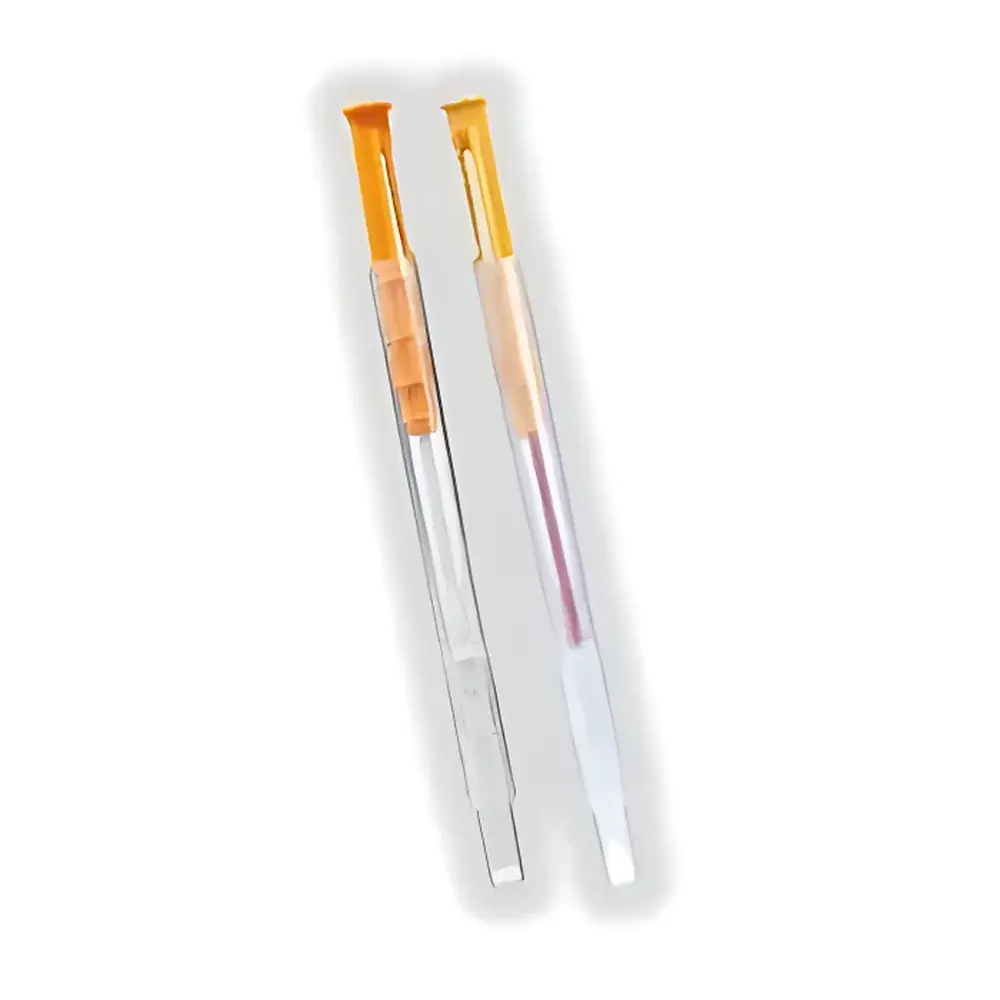
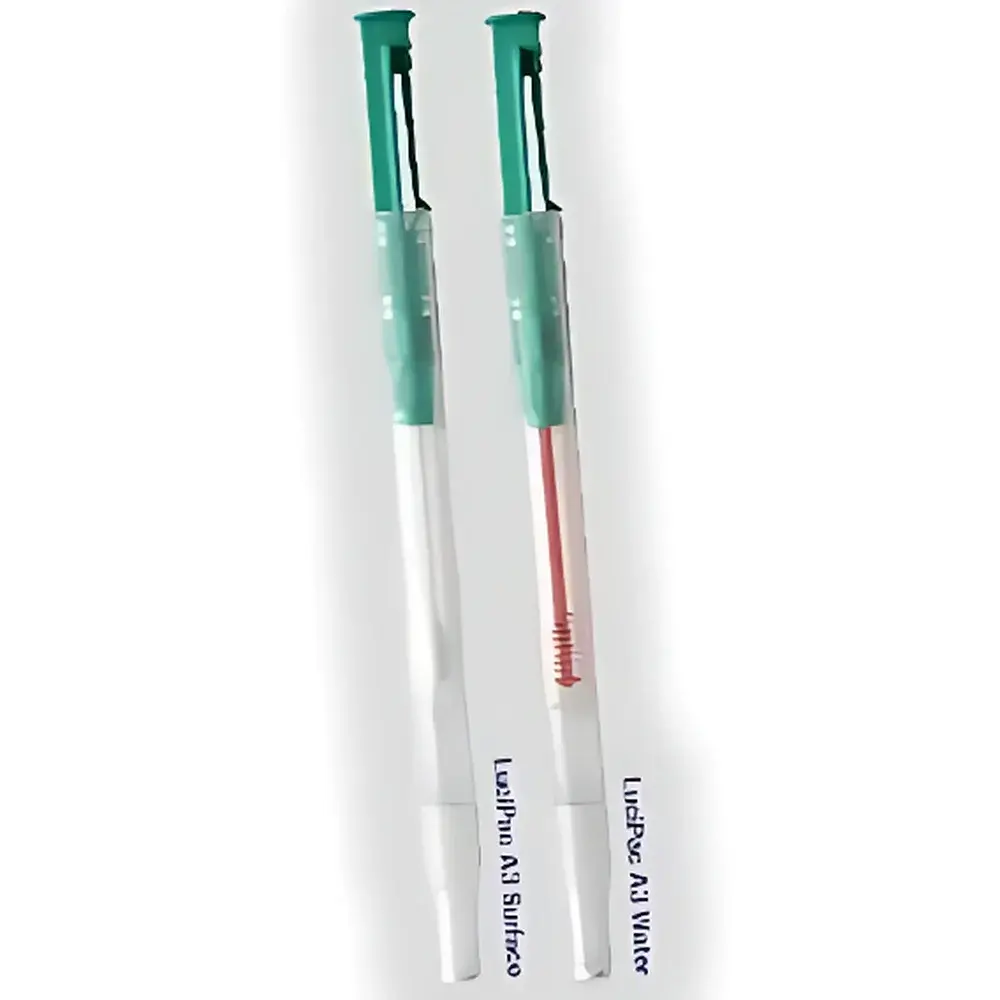
- Pre-calibrated, single-use, integrated swab-and-reagent detection sticks: cotton-tipped for surfaces, liquid-sampling format for rinse water or broth, and extended-length probes for narrow tubing or crevices.
- On-device data logging with timestamp, operator ID, location tag, and RLU value; supports up to 1,000 test records without external software.
- IP65-rated enclosure with chemical-resistant housing; operates continuously for ≥8 hours on rechargeable Li-ion battery.
- Compliant with ISO 22000, HACCP, and FDA Food Code Annex 3 guidance for environmental monitoring verification.
Sample Compatibility & Compliance
The PD-30 accommodates a broad spectrum of sampling matrices without dilution or pretreatment: stainless steel, glass, polypropylene, rubber gaskets, textile linens, human skin, and aqueous rinse solutions. Surface sampling follows standardized 10 cm × 10 cm (250 px × 250 px) protocols per ISO 14305 and AOAC Official Method 2015.01. For high-risk zones (e.g., food contact surfaces), recommended action limits are ≤200 RLU (smooth), ≤500 RLU (textured), and ≤2,000 RLU (hand surfaces). All reagents are manufactured under ISO 13485-certified conditions and supplied with lot-specific calibration certificates. Instrument performance validation includes daily ATP standard checks (10⁻¹⁵ mol ATP) and quarterly third-party verification per ASTM E2694-22 (Standard Guide for ATP Bioluminescence Assays in the Food Industry).
Software & Data Management
Raw RLU data export via USB-C or Bluetooth 5.0 to Kikkoman’s free PD-Link desktop application (Windows/macOS). PD-Link supports statistical process control (SPC) charting, trend analysis by zone/operator/time, automated alerting at user-defined thresholds, and PDF report generation compliant with GLP/GMP documentation requirements. Audit trail functionality meets FDA 21 CFR Part 11 criteria—including electronic signatures, immutable timestamps, and full change history—with optional integration into enterprise LIMS platforms via HL7 or CSV API. Data encryption conforms to AES-256 standards; no cloud storage is required unless explicitly enabled by the user.
Applications
- Food Manufacturing: Verification of CIP/SIP efficacy, post-sanitation validation, allergen residue screening, and root-cause analysis during non-conformance events.
- Healthcare Facilities: Environmental surface monitoring in operating rooms, patient rooms, and reusable medical device reprocessing areas per CDC/HICPAC guidelines.
- Hospitality & Public Infrastructure: Routine hygiene assessment of hotel linens, elevator buttons, spa filtration systems, and public transport interiors.
- Museum & Archival Conservation: Early detection of microbial colonization on parchment, wood, or textile artifacts using non-destructive surface swabbing.
- Education & Training: Real-time demonstration of hand hygiene efficacy, cleaning technique optimization, and cross-contamination risk awareness programs.
FAQ
What does “A³ Method” stand for?
A³ stands for Adenylate Amplification & Amplified ATP Detection—a proprietary enzymatic amplification protocol that converts ADP and AMP to ATP prior to bioluminescent quantification.
Can the PD-30 differentiate between microbial ATP and food-residue ATP?
No—it measures total adenylate load as a hygiene indicator. Differentiation requires complementary microbiological culture or PCR methods.
Is calibration required before each use?
No. The instrument performs automatic dark-current compensation and uses factory-calibrated, lot-matched reagents; only daily ATP standard verification is recommended.
How is RLU correlated to microbial concentration?
RLU values are not directly convertible to CFU/mL without empirical correlation studies specific to your environment, surface type, and recovery efficiency.
Does the PD-30 comply with regulatory audit requirements?
Yes—its data integrity architecture, audit trail, and documentation outputs support FDA, EFSA, and MHRA inspections under GMP/GLP frameworks.