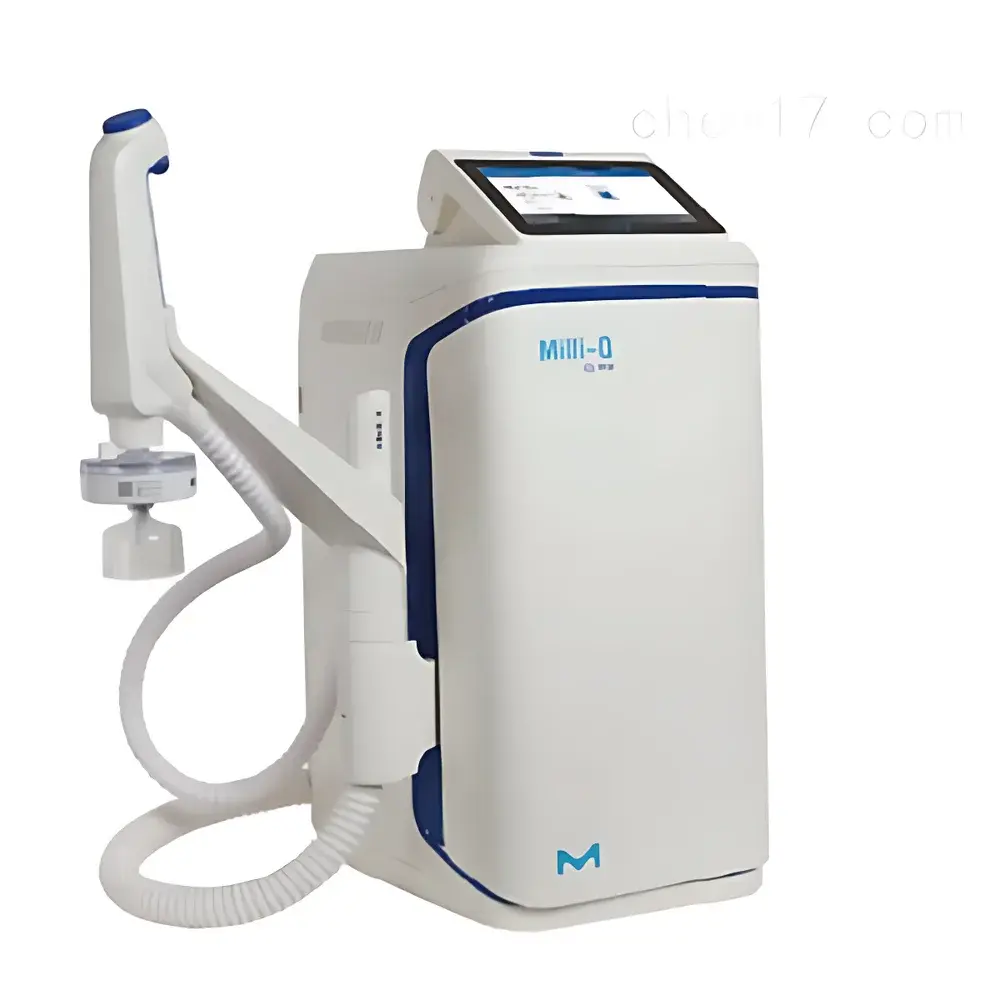
Milli-Q® EQ 7000 Ultra Pure Water System by Merck
| Brand | Milli-Q |
|---|---|
| Origin | France |
| Manufacturer | Merck KGaA |
| Product Type | Imported |
| Model | Milli-Q EQ 7000 |
| Price Range | USD 13,500 – 40,500 (est.) |
| Water Grade | Type I (ASTM D1193-20) |
| Resistivity | 18.2 MΩ·cm at 25 °C |
| TOC | ≤ 5 ppb |
| Endotoxins | < 0.001 EU/mL |
| Particles (>0.22 µm) | Non-detectable |
| Microbial Count | < 1 CFU/100 mL |
| Terminal Filtration | 0.22 µm sterile filtration (Millipak® Gold) |
| Compliance | ISO 9001, ISO 14001, FDA 21 CFR Part 11 ready (with optional audit trail module), GLP/GMP-supportive data management |
Overview
The Milli-Q® EQ 7000 Ultra Pure Water System by Merck is an integrated, benchtop-grade water purification platform engineered for laboratories requiring consistent, on-demand Type I ultrapure water compliant with ASTM D1193-20, ISO 3696, and CLSI EP21-A standards. It employs a multi-stage purification architecture comprising pre-treatment (activated carbon + dual-stage reverse osmosis), electrodeionization (EDI), dual-wavelength UV oxidation (185/254 nm), and final point-of-use polishing via replaceable POD-Pak® cartridges. The system continuously monitors resistivity, TOC, temperature, and flow rate in real time—ensuring full traceability of water quality parameters at the point of dispensing. Designed for scientific rigor rather than operational convenience alone, the EQ 7000 integrates seamlessly into regulated environments where data integrity, reproducibility, and compliance with GLP, GMP, and ISO/IEC 17025 frameworks are non-negotiable.
Key Features
- 7-inch high-resolution color touchscreen interface with intuitive icon-driven navigation and multilingual support (English, French, German, Chinese, Japanese, Korean)
- Q-POD® dispensing arm with three manually adjustable flow rates (low/medium/high), up to 2 L/min, and one-touch volumetric dispensing (preset volumes: 10 mL–5 L)
- Foot-switch activation option for hands-free operation—minimizing surface contact and supporting aseptic workflows
- Modular POD-Pak® terminal purification cartridges: Millipak® Gold (0.22 µm sterile filtration), Biopak® (ultrafiltration for endotoxin-, nuclease-, and protease-free water), EDS-Pak® (endocrine disruptor removal), LC-Pak® (trace organic scavenging), VOC-Pak® (volatile organic compound retention)
- e-Sure™ RFID-enabled cartridge identification for automatic usage tracking, remaining lifespan estimation, and audit-compliant consumables history
- Integrated online TOC sensor (UV-persulfate oxidation + NDIR detection) delivering real-time TOC values ≤ 5 ppb directly at the dispensing point
- Recirculating loop with periodic UV irradiation and programmable idle-mode sanitation to maintain microbial control during non-use periods
Sample Compatibility & Compliance
The Milli-Q® EQ 7000 delivers water suitable for critical analytical and life science applications—including HPLC, LC-MS, ICP-MS, cell culture, molecular biology (PCR, qRT-PCR, sequencing), and immunology assays. Its output meets or exceeds specifications defined in USP , EP 2.2.44, JP 17, and ISO 3696 Class 1. All POD-Pak® cartridges are manufactured under ISO 9001-certified conditions and validated per ASTM F838-20 for bacterial retention efficiency. System firmware supports 21 CFR Part 11-compliant electronic records when paired with Merck’s MyMilli-Q™ cloud service and optional audit trail license. Data logs—including resistivity, TOC, cartridge usage, maintenance events, and user actions—are exportable in CSV or PDF format with time-stamped digital signatures.
Software & Data Management
The EQ 7000 features embedded Ethernet connectivity (DHCP or static IP configuration) enabling integration into local laboratory networks. Remote monitoring, alarm notification, and historical data review are accessible via web browser or MyMilli-Q™ mobile application. All system events—including calibration checks, cartridge replacement alerts, and quality deviations—are timestamped and stored with immutable metadata. USB port allows direct export of raw logs to encrypted flash drives for offline archiving. Optional MyMilli-Q™ Digital Service subscription provides predictive maintenance scheduling, remote diagnostics, and automated firmware updates—all aligned with ISO/IEC 17025 traceability requirements.
Applications
- HPLC and UHPLC mobile phase preparation and instrument rinsing
- LC-MS and GC-MS solvent blanks and calibration standards
- Cell culture media formulation and reagent dilution requiring endotoxin-free water
- Nucleic acid isolation, transfection, and CRISPR-based workflows demanding nuclease-free conditions
- Trace metal analysis (ICP-OES, ICP-MS) where low background contamination is essential
- Pharmaceutical QC testing per USP and EP 2.2.44 water quality monographs
- Environmental analysis of persistent organic pollutants (POPs) and endocrine-disrupting compounds
FAQ
What is the guaranteed resistivity specification for Milli-Q® EQ 7000 water?
The system consistently delivers water with a resistivity of 18.2 MΩ·cm at 25 °C, verified continuously via in-line conductivity sensors calibrated against NIST-traceable standards.
How often must POD-Pak® cartridges be replaced?
Replacement intervals depend on usage volume and feed water quality; typical lifespans range from 3–12 months. The e-Sure™ system calculates remaining capacity based on actual flow and contaminant load, triggering alerts before performance degradation occurs.
Is the TOC sensor factory-calibrated and field-verifiable?
Yes—the integrated TOC analyzer undergoes factory calibration using certified potassium hydrogen phthalate standards and supports user-performed verification with traceable sucrose solutions per ASTM D5904-21.
Can the EQ 7000 be integrated into a centralized lab information management system (LIMS)?
Via its RESTful API and Modbus TCP protocol support, the system enables bidirectional data exchange with validated LIMS platforms for automated sample logging, audit trail synchronization, and electronic batch record generation.
Does Merck provide installation qualification (IQ) and operational qualification (OQ) documentation?
Yes—Merck-certified field engineers deliver full GMP-aligned IQ/OQ documentation packages upon installation, including as-built schematics, calibration certificates, and risk-assessed validation protocols compliant with Annex 15 and WHO TRS 986.