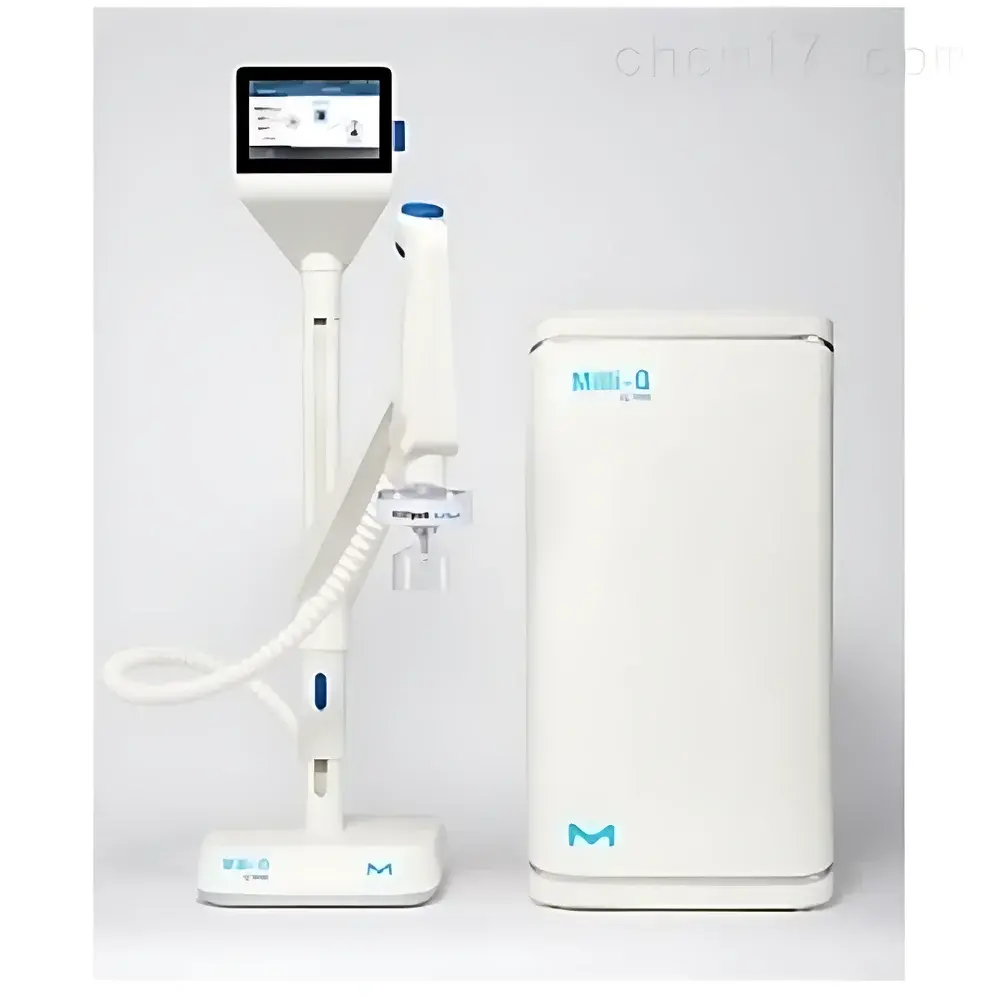
Milli-Q® IQ 7000 Ultra Pure Water System
| Brand | Milli-Q |
|---|---|
| Origin | France |
| Manufacturer | Merck KGaA |
| Product Type | Imported Ultra-Pure Water System |
| Model | Milli-Q IQ 7000 |
| Grade | Type I (ASTM D1193-20) |
| Resistivity | 18.2 MΩ·cm at 25 °C |
| TOC | ≤2 ppb (µg/L), typical ≤5 ppb |
| Endotoxins | <0.001 EU/mL |
| Microbial Count | <1 CFU/100 mL |
| Particles (>0.1 µm) | <1 particle/mL |
| Final Filtration | 0.22 µm PES membrane |
| UV Wavelength | 172 nm (Xe-excimer, mercury-free) |
| Compliance | ASTM D1193, ISO 3696, CLSI EP28-A3c, USP <1231>, EP 2.2.44, FDA 21 CFR Part 11 (audit trail enabled) |
Overview
The Milli-Q® IQ 7000 Ultra Pure Water System is a fully integrated, intelligent water purification platform engineered for laboratories requiring continuous, trace-level purity in Type I ultrapure water. It employs a multi-stage purification architecture combining pretreatment (Elix® or equivalent feedwater), dual-wavelength UV oxidation (172 nm Xe-excimer lamp), advanced mixed-bed ion exchange (IQ o™ and IPAK Meta/Quanta cartridges), and final 0.22 µm sterile filtration. Real-time, on-line monitoring of resistivity (cell constant 0.01 cm⁻¹, ±0.1 °C temp compensation), TOC (via A10 online sensor), temperature, and flow ensures compliance with stringent analytical and life science standards—including USP , EP 2.2.44, and ISO 3696 Grade 1. Designed and manufactured in France under ISO 9001 and ISO 14001 certified facilities, the system delivers stable 18.2 MΩ·cm resistivity with sub-ppb TOC and endotoxin levels below 0.001 EU/mL—enabling reproducible performance across high-sensitivity applications such as LC-MS, single-cell genomics, and low-background elemental analysis.
Key Features
- Q-POD® Touch Interface: Intuitive, tablet-style graphical user interface with real-time water quality display, programmable dispensing modes (drop-by-drop to 2 L/min), and guided maintenance workflows.
- Mercury-Free 172 nm UV Oxidation: Xenon-excimer lamp technology eliminates mercury use while delivering superior organic oxidation efficiency; validated for consistent TOC reduction to ≤2 ppb (typical).
- IQ o™ Ion Exchange Resin: Proprietary fine-particle mixed-bed resin with enhanced kinetics—reducing ion exchange media volume by 33% versus prior generations without compromising deionization capacity or lifespan.
- e-Sure™ Smart Cartridge Technology: RFID-enabled POD-Pak® cartridges with auto-recognition, usage tracking, and full audit trail integration; all consumables logged with timestamps, batch IDs, and expiration data.
- Modular Installation Flexibility: Compact Q-POD arm mounts on benchtop, wall, or cabinet; customizable tubing lengths with integrated cable management; system base unit accommodates under-bench or remote placement.
- Lab-Closure Mode: Energy- and water-conserving standby protocol for extended unattended operation; maintains microbial and chemical stability with minimal flush volume and power draw.
Sample Compatibility & Compliance
The Milli-Q® IQ 7000 is validated for compatibility with a broad spectrum of analytical and biological workflows requiring ultrapure water input. Its output meets or exceeds specifications for HPLC, UHPLC, GC, LC-MS, ICP-MS, AA, CE, MALDI-TOF, and IC. For life science applications—including cell culture, qPCR, NGS library prep, ELISA, western blotting, and monoclonal antibody formulation—the system supports optional terminal polisher modules (e.g., BioPak®, ViruPak®, and IsoPak®) to remove nucleases, DNase/RNase, viruses, or volatile organics. All hardware and firmware comply with GLP and cGMP requirements: built-in audit trail (21 CFR Part 11 compliant), electronic signatures, user access control with password-protected configuration tiers, and full traceability of every dispense event—including operator ID, volume, time/date, and real-time resistivity/TOC values.
Software & Data Management
The IQ 7000 integrates a secure, embedded database capable of storing >10 years of operational history. Every water delivery event is timestamped and associated with contextual metadata—user ID, project code, application type, and custom annotations. Reports are exportable via QR code scan or USB drive in PDF or CSV format. Daily, weekly, and monthly quality summaries include average resistivity, TOC trends, total volume dispensed, and cartridge utilization metrics. The system supports cost-allocation reporting by user or department. Calibration logs for the resistivity sensor (certified cell constant), temperature probe, and A10 TOC module are retained with traceable certificates. Full validation documentation—including IQ/OQ protocols, PQ templates, MP (maintenance procedure) guidelines, and system suitability test procedures for USP/EP compliance—is provided with installation.
Applications
This system serves as the primary ultrapure water source for core laboratory functions where background interference must be eliminated. In analytical chemistry labs, it supplies mobile phases for UPLC and solvents for ICP-MS sample introduction—ensuring low baseline noise and minimizing spectral interferences. In biotechnology settings, it supports serum-free mammalian cell culture media preparation, CRISPR-Cas9 reagent dilution, and high-fidelity DNA sequencing workflows where nuclease contamination or ionic carryover would compromise assay integrity. Environmental testing labs rely on its reproducible TOC performance for EPA Method 5310B validation. Pharmaceutical QC units deploy it for dissolution media preparation per USP , while academic core facilities utilize its modular polishers for proteomics (removing keratin contaminants) and surface science (eliminating surfactants affecting contact angle measurements).
FAQ
What feedwater quality is required for optimal IQ 7000 performance?
The system requires pretreated feedwater meeting ASTM D1193 Type III or ISO 3696 Grade 3 specifications—typically supplied by an Elix® or similar reverse osmosis/deionization unit. Inlet conductivity should remain below 50 µS/cm; higher levels may reduce cartridge lifetime and increase TOC breakthrough risk.
How does the A10 TOC sensor differ from conventional UV–persulfate methods?
The A10 uses a patented dual-wavelength (185/254 nm) UV oxidation chamber coupled with non-dispersive infrared (NDIR) detection, enabling real-time, reagent-free TOC quantification with <0.5 ppb detection limit and <±5% accuracy across 0–100 ppb range.
Can the IQ 7000 be integrated into a centralized lab water network?
Yes—via optional distribution loop kits with recirculation pump, sanitary stainless-steel piping, and loop-mounted UV units. System firmware supports remote monitoring via Ethernet or optional Wi-Fi module with Merck’s LabManager™ cloud dashboard.
Is validation support available for regulated environments?
All IQ 7000 units ship with Factory Acceptance Test (FAT) reports and Certificate of Conformance. Merck-certified Field Service Engineers provide IQ/OQ execution, PQ support, and ongoing calibration services aligned with ISO/IEC 17025 and Annex 11 requirements.
What is the expected service life of IQ o™ cartridges under standard lab usage?
Cartridge lifespan is usage-dependent but typically ranges from 6–12 months when fed with Elix-grade water at average daily demand of 100–200 L. The e-Sure™ system continuously calculates remaining capacity based on actual ion load and TOC exposure—not just time-based estimation.