Optometric SPF-290S In-Vitro Sun Protection Factor Measurement System
| Brand | Optometric |
|---|---|
| Origin | USA |
| Model | SPF-290S |
| Wavelength Range | 190–2600 nm (UV-Vis-NIR) |
| Detector Type | Photomultiplier Tube (PMT) |
| Optical Configuration | Dual-Wavelength |
| Spectral Resolution | 1.66 nm |
| Effective SPF Measurement Range | 290–400 nm |
| Wavelength Accuracy | ±0.2% |
| Wavelength Repeatability | 0.25 nm |
| Measurement Mode | Manual Wavelength Selection |
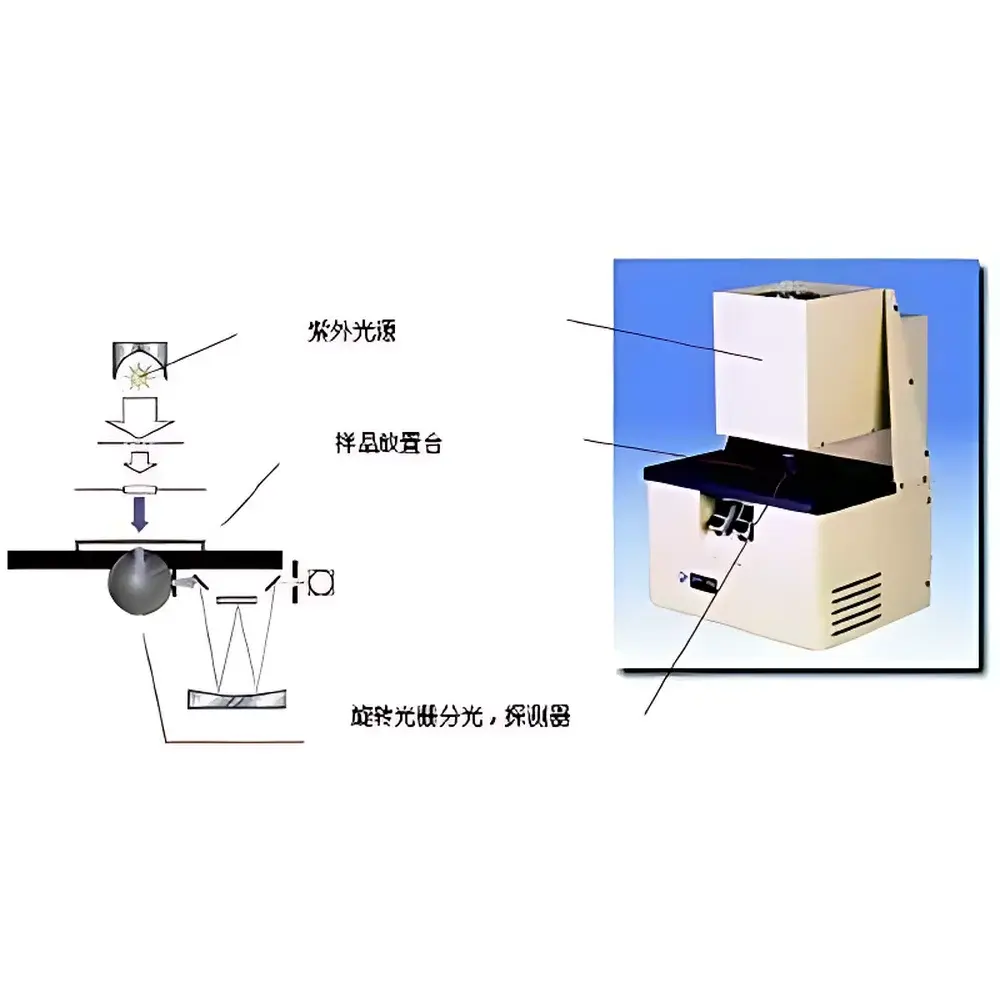
| Sample Illumination Source | Continuous Xenon Lamp with Solar-Spectrum-Matching Filters |
| Detection Geometry | Integrating Sphere |
| Measurement Points per Sample | Up to 12 Automatic Positions |
| Typical Measurement Time per Sample | ~20 s |
| Compliance Context | Aligned with FDA-recommended in-vitro methodology (21 CFR Part 352, ISO 24443:2021, COLIPA/ISO 24444:2019) |
Overview
The Optometric SPF-290S In-Vitro Sun Protection Factor Measurement System is a purpose-engineered spectrophotometric platform designed for standardized, reproducible quantification of ultraviolet (UV) radiation attenuation by topical sun protection products. Unlike in-vivo human testing—which faces ethical constraints, inter-subject variability, and regulatory complexity—the SPF-290S implements a validated in-vitro methodology based on spectral transmittance measurement across the biologically relevant UVB (290–320 nm) and UVA (320–400 nm) ranges. The system integrates a continuous-output xenon arc lamp with precision optical filtering to replicate the solar spectral irradiance profile mandated by FDA guidance documents and harmonized with ISO 24443:2021. UV transmittance is measured through a standardized substrate—typically a polymethyl methacrylate (PMMA) plate coated with a uniform, controlled thickness of sunscreen formulation—using a high-stability double-beam UV-Vis-NIR spectrophotometer (190–2600 nm range) equipped with a photomultiplier tube (PMT) detector and an integrating sphere for comprehensive collection of diffusely transmitted and scattered radiation. This architecture ensures traceable, operator-independent SPF calculation via the critical wavelength-weighted integral method defined in ISO 24444.
Key Features
- Continuous-spectrum xenon light source with solar-matched spectral output—identical in spectral fidelity to the reference source used in FDA-regulated in-vivo protocols
- Integrating sphere detection geometry enabling full capture of forward-scattered and diffusely transmitted UV photons, minimizing measurement bias from sample heterogeneity or surface scattering
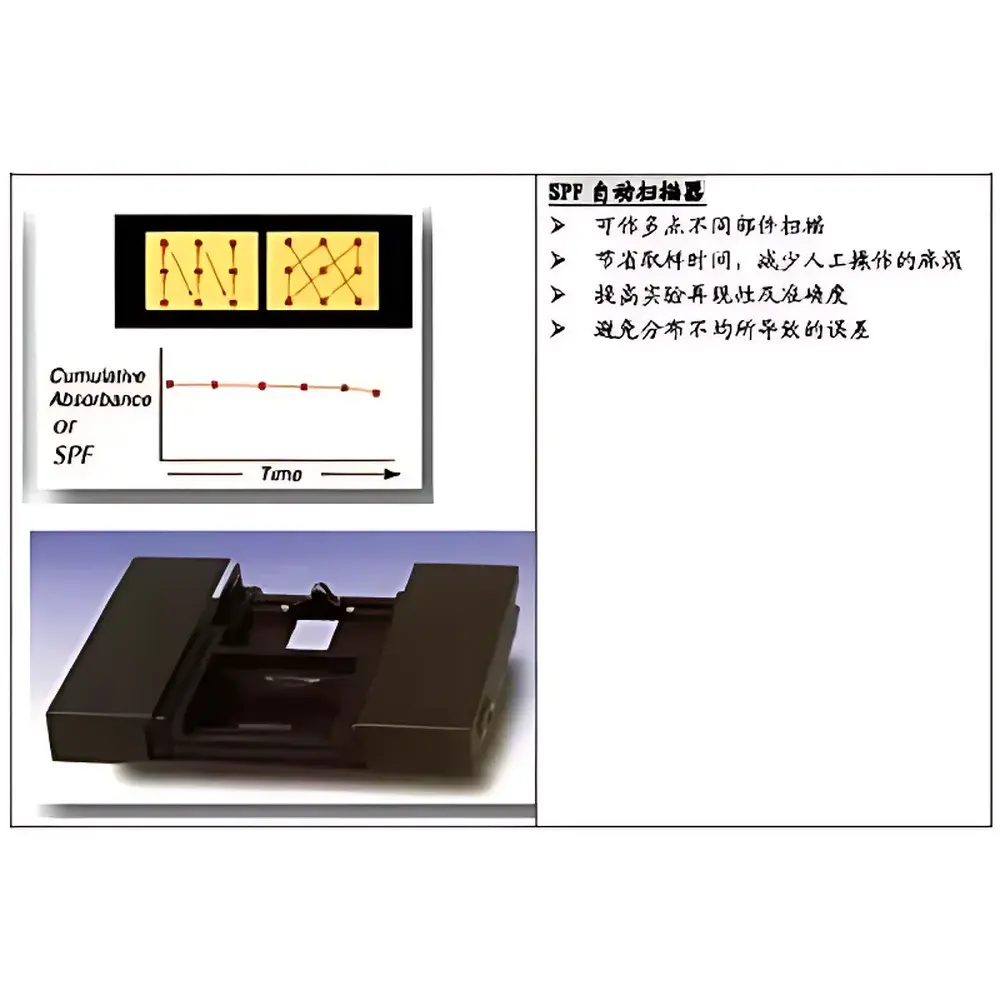
- Automated multi-point sampling across up to 12 predefined locations on the test substrate, generating statistically robust mean SPF values and standard deviation metrics
- Dual-wavelength optical path design supporting simultaneous reference and sample beam monitoring for enhanced baseline stability and drift compensation
- High-resolution spectral acquisition (1.66 nm) within the 290–400 nm SPF-critical band, with wavelength accuracy of ±0.2% and repeatability of 0.25 nm
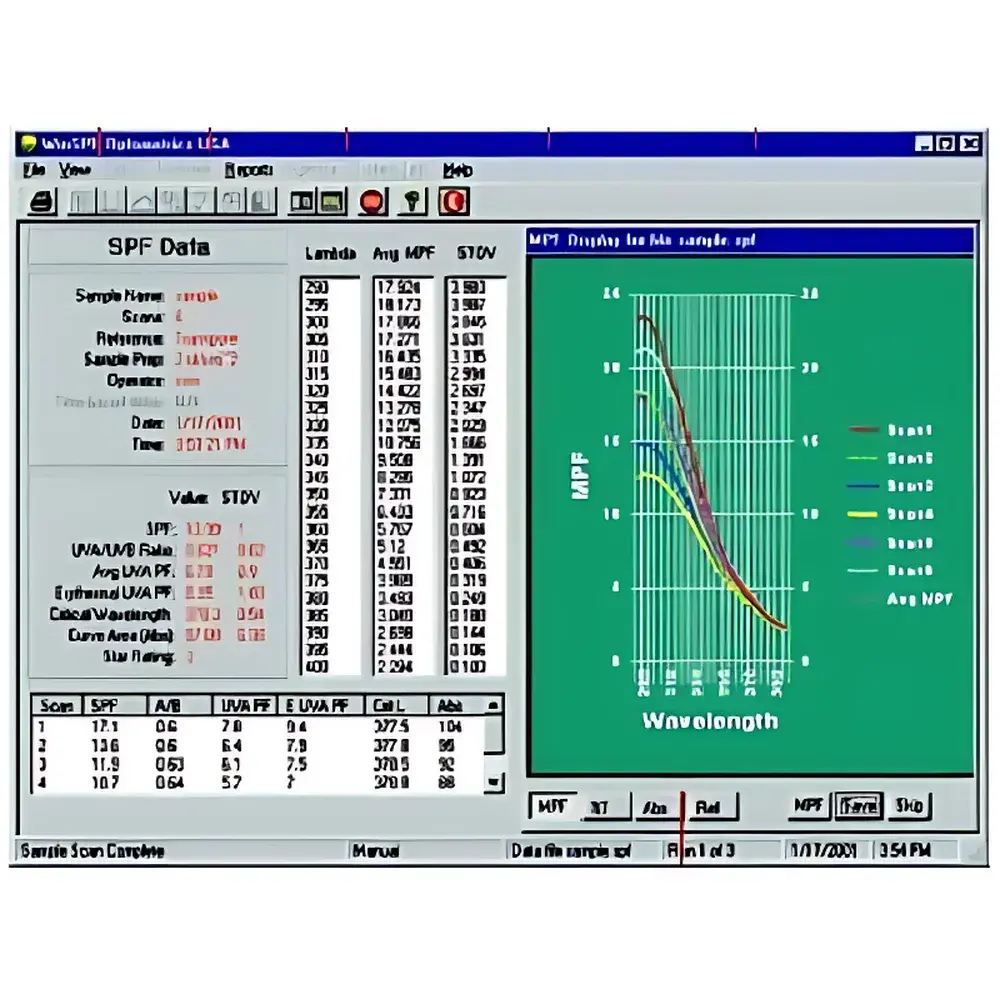
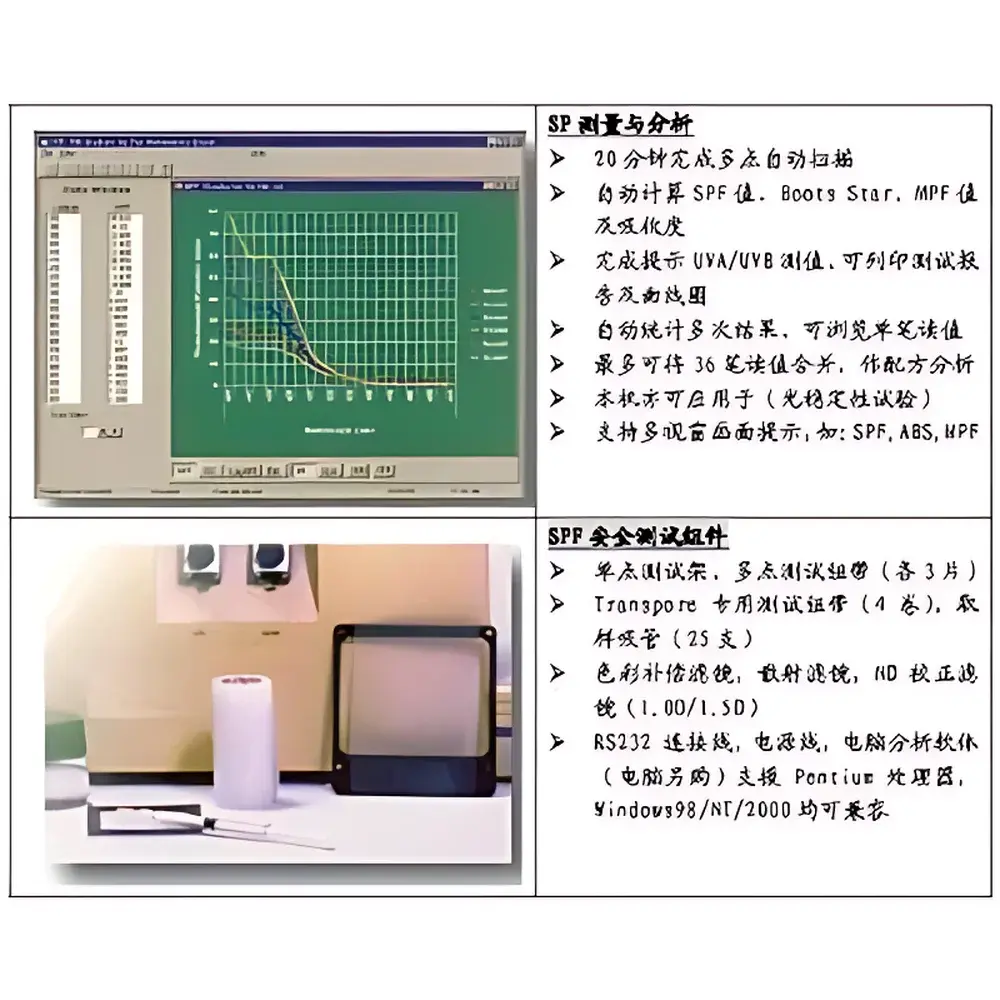
- Comprehensive software suite implementing ISO 24444:2019 and ISO 24443:2021 algorithms for SPF, UVA-PF, critical wavelength (λc), and UVA/UVB ratio calculations
Sample Compatibility & Compliance
The SPF-290S accommodates a broad range of UV-absorbing formulations applied to standardized PMMA substrates—including oil-in-water emulsions, water-in-oil systems, sprays, gels, and solid stick formats—provided coating uniformity meets ISO-defined thickness tolerances (±10%). Test accessories include Transpore™-compatible adhesive tapes, calibrated pipettes (25 µL), neutral density filters (1.2D and 1.5D), scattering correction filters, and color-compensation optics to address non-UV-absorbing chromophores. The system supports full audit trail generation, user access control, and electronic signature capability—enabling alignment with GLP-compliant laboratory practices and 21 CFR Part 11 requirements when deployed in regulated R&D or QC environments. Validation documentation includes traceable NIST-calibrated spectral irradiance data and inter-laboratory comparison reports published by Korean MFDS and European Cosmetics Europe consortiums.
Software & Data Management
The proprietary SPF Analysis Software provides a validated, menu-driven interface for instrument control, spectral acquisition, and regulatory-grade data reduction. All raw transmittance spectra are stored in vendor-neutral ASCII format with embedded metadata (timestamp, operator ID, sample ID, calibration status). The software implements automated baseline correction, stray-light compensation, and interpolation routines compliant with ISO 24444 Annex B. Quantitative outputs include: SPF (calculated per COLIPA algorithm), UVA Protection Factor (UVA-PF), critical wavelength (λc), UVA/UVB ratio, and spectral absorbance profiles. Export options include CSV, PDF analytical reports, and XML for LIMS integration. Audit logs record all parameter changes, measurement events, and software updates—supporting full traceability during internal audits or regulatory inspections.
Applications
- Regulatory-compliant in-vitro SPF screening during preclinical development of OTC sunscreen actives and finished products
- QC release testing of commercial sunscreen batches against internal specifications and ISO 24444 acceptance criteria
- Formulation optimization studies evaluating UV filter synergies, photostability degradation kinetics, and film integrity under simulated wear conditions
- Performance benchmarking of textile-based UV protective fabrics (UPF testing via modified substrate protocols)
- Photostability assessment of photosensitive pharmaceuticals and cosmetic actives exposed to controlled UV stress
- Method transfer and cross-validation between contract research organizations (CROs) and brand-owner laboratories
FAQ
Does the SPF-290S comply with current FDA or ISO sunscreen testing standards?
Yes—the system implements the core optical and computational principles specified in ISO 24443:2021 (in-vitro method) and aligns with FDA’s 2011 Final Rule (21 CFR Part 352) for sunscreen labeling, including spectral weighting functions and integration limits.
Can the SPF-290S measure UVA protection independently of SPF?
Yes—it calculates UVA Protection Factor (UVA-PF), critical wavelength (λc), and UVA/UVB ratio using the same spectral dataset, per ISO 24443 Annex A and COLIPA guidelines.
Is the system suitable for GLP or GMP-regulated environments?
With appropriate configuration (user access controls, electronic signatures, audit trail activation), the SPF-290S meets functional requirements for GLP-compliant sunscreen testing and supports 21 CFR Part 11 compliance when integrated into validated IT infrastructure.
What substrate materials are supported for sample application?
Standardized PMMA plates (e.g., 25 × 75 mm, 1 mm thick) are required; optional textured or roughened variants are available for mimicking skin topography per ISO 24443 Annex C.
How is instrument performance verified between measurements?
Daily verification uses NIST-traceable neutral density filters and scattering reference standards; full wavelength and photometric calibration is performed semi-annually using certified holmium oxide and potassium dichromate solutions.