PULUODY PLD-MPCS2.0 Insoluble Particulate Microscopic Counting System
| Brand | PULUODY |
|---|---|
| Origin | Shaanxi, China |
| Model | PLD-MPCS2.0 |
| Dispersion Method | Wet Dispersion |
| Instrument Type | Laboratory Microscopy-Based Particle Counting System |
| Measurement Range | 1 µm – 500 µm |
| Repeatability | < 5% (excluding sample preparation variability) |
| Analysis Time | 60 seconds |
| Magnification | 40× – 1000× |
| Resolution | 0.1 µm |
| Microscope Accuracy | ±0.02 µm |
| CCD Camera | 3 MP |
| Calibration Standard | NIST-traceable stage micrometer (0.1 µm division) |
| Software Environment | Windows OS (XP through 11) |
| Interface | RS-232 or USB |
| Particle Recognition Accuracy | >95% (per ChP 2020), <10% misclassification (per USP <788> & ISO 21501-1) |
| Overlap Tolerance | ≤5% at 10,000 particles/mL |
| Image Stitching | Fully automated, seam-free membrane scanning |
| Segmentation Speed | <1 second per field |
| Segmentation Success Rate | >93% |
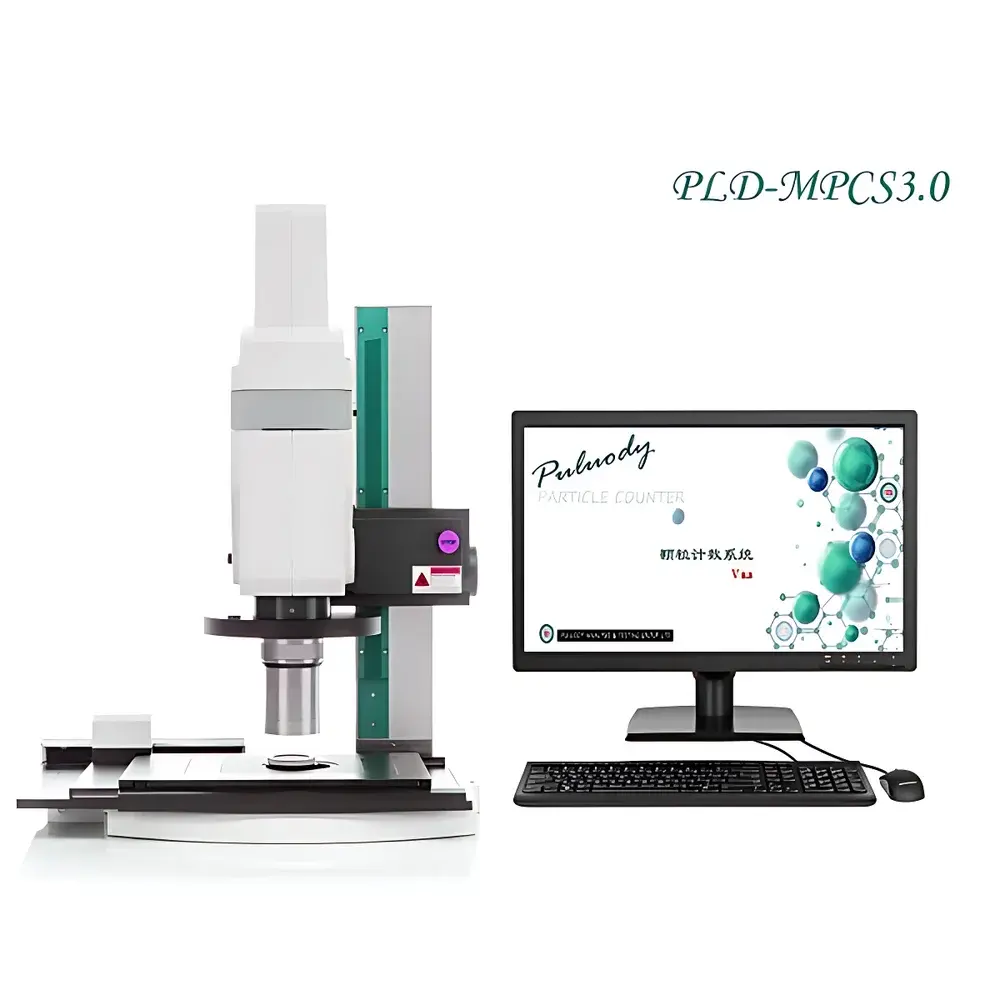
Overview
The PULUODY PLD-MPCS2.0 Insoluble Particulate Microscopic Counting System is a regulatory-compliant, image-based particle analysis platform engineered for quantitative morphological characterization and size distribution measurement of insoluble particulates in liquid pharmaceuticals, high-purity process fluids, and critical industrial suspensions. Unlike laser diffraction or light obscuration (LO) techniques, this system implements the pharmacopoeial microscopic counting method—specifically aligned with Chinese Pharmacopoeia (ChP) General Chapter 0903 Method 2—and delivers traceable, visually verifiable particle data grounded in direct optical imaging. At its core, the system integrates a precision upright metallurgical microscope, a high-sensitivity monochrome CCD camera, motorized XY-stage scanning, and deterministic image segmentation algorithms to acquire, stitch, and analyze particles across large-area membrane filters (e.g., 25 mm or 47 mm pore-track membranes). It measures not only equivalent circular diameter (ECD), but also aspect ratio, circularity, convexity, and Feret dimensions—parameters essential for root-cause investigation of particulate origin (e.g., fiber shedding, silicone oil droplets, crystalline precipitates, or metallic wear debris).
Key Features
- Full compliance with ChP 0903, USP , USP , EP 2.9.19, JP 6.06, and ISO 21501-1 Annex A for microscopic particle counting
- Automated, programmable membrane scanning with sub-micron positional repeatability and seamless digital tiling
- Calibration-traceable optical path: 0.1 µm stage micrometer, validated against NIST SRM 2806 and ISO 21501-3 reference standards
- Deterministic particle segmentation engine optimized for low-contrast, overlapping, and irregularly shaped particles—achieving >93% segmentation success rate
- Multi-parameter morphological profiling: ECD, length/width, aspect ratio distribution, circularity histogram, and convex hull analysis
- Role-based operator management with audit trail logging per FDA 21 CFR Part 11 requirements (electronic signature support optional)
- Pre-configured test templates for NAS 1638, ISO 4406, ISO 11171, and USP drug product categories (injectables, ophthalmics, vaccines, APIs)
- Manual focus and intensity control—required for compliant cleanroom-grade particulate assessment where automated exposure may mask subtle contaminants
Sample Compatibility & Compliance
The PLD-MPCS2.0 supports filtration-based analysis of aqueous and organic solvents across regulated and industrial applications. Compatible sample matrices include sterile injectables (e.g., monoclonal antibody formulations), ophthalmic solutions, vaccine suspensions, injection-grade water (WFI, PW), CMP slurries, electronic-grade ultrapure water (UPW), hydraulic oils, and semiconductor process chemicals. All analysis workflows adhere to Good Laboratory Practice (GLP) and Good Manufacturing Practice (GMP) documentation requirements. The system enables full traceability from raw image capture to final report generation—including metadata stamping (date/time, operator ID, filter lot, dilution factor, instrument calibration status). It satisfies validation criteria outlined in ASTM E2457, ISO 13322-1, and ICH Q5C for particle characterization in biopharmaceutical development.
Software & Data Management
The proprietary PULUODY ParticleVision™ software provides a secure, modular interface for method development, acquisition control, batch reporting, and long-term data archiving. Raw TIFF images are stored with embedded EXIF metadata; processed results export to CSV, PDF, and XML formats compatible with LIMS integration. The software enforces user-level permissions, version-controlled SOPs, and electronic signature workflows compliant with FDA 21 CFR Part 11 Subpart C. Audit trails record all parameter modifications, image re-analyses, and report revisions—supporting regulatory inspections under MHRA, PMDA, or EMA GLP audits. Optional cloud-sync modules enable remote review and cross-site method harmonization without compromising data sovereignty.
Applications
- Pharmaceutical Quality Control: Quantitative assessment of insoluble particles in parenteral drug products per ChP, USP, EP, and JP monographs
- Medical Device Evaluation: Testing of infusion sets, IV bags, syringes, and terminal filters for extractable/leachable particulates
- Water Purity Monitoring: Solid contaminant profiling in WFI, UPW, and process rinse waters per ASTM D5127 and SEMI F63
- Semiconductor Manufacturing: Slurry cleanliness verification (CMP, etchants) and wafer rinse analysis per SEMI F39
- Hydraulic & Lubrication Systems: Wear debris morphology analysis for predictive maintenance per ISO 4406 and ISO 11171
- Cosmetic & Food Safety: Foreign particulate identification in emulsions, suspensions, and sterile fill products
FAQ
Does the PLD-MPCS2.0 comply with USP for injectable testing?
Yes—it implements the microscopical particle count method specified in USP Chapter, including mandatory membrane filtration, defined field-of-view sampling, and manual focus verification per protocol.
Can it differentiate between silicone oil droplets and crystalline particles?
Yes—through multi-threshold grayscale segmentation, edge sharpness analysis, and circularity metrics, enabling classification of spherical vs. angular morphologies.
Is the system suitable for GMP-regulated environments?
Yes—when configured with Part 11-compliant software options, it meets ALCOA+ data integrity principles and supports full 21 CFR Part 11 validation packages.
What is the maximum particle concentration supported without coincidence error?
Up to 10,000 particles/mL with ≤5% coincidence error, verified using ISO 21501-1 reference suspensions.
Does it support custom reporting per internal quality standards?
Yes—users can define pass/fail thresholds, generate tiered reports (e.g., “Class 100” vs. “Class 10”), and embed company logos, QA stamps, and controlled document numbers.