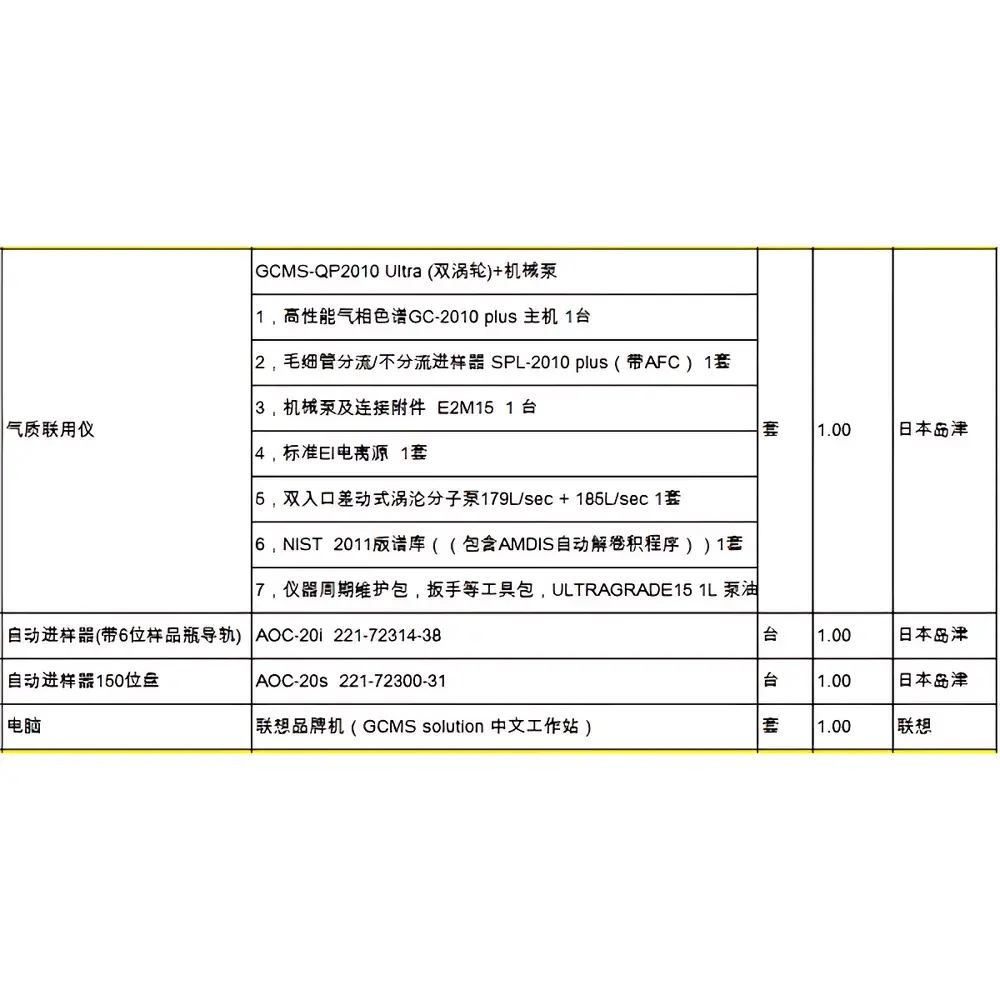
Shimadzu GCMS-QP2010 Plus Used Gas Chromatography-Mass Spectrometry System
| Brand | Shimadzu |
|---|---|
| Origin | Japan |
| Model | QP2010 Plus |
| Instrument Type | Benchtop GC-MS |
| Configuration | Single Quadrupole |
| Detection Mode | Electron Ionization (EI) |
| Mass Range | 1.5–1099 u |
| Scan Speed | Up to 10,000 u/sec |
| Interface Temperature | Up to 300 °C |
| Ion Source | Dual-mode (EI/CI optional with upgrade) |
| Vacuum System | Turbomolecular pump + backing pump |
| Compliance | Fully compatible with ASTM D5845, EPA Method 8270/8260, USP <621>, ISO 17025 workflows |
Overview
The Shimadzu GCMS-QP2010 Plus is a high-performance, single-quadrupole gas chromatography-mass spectrometry system originally engineered for routine qualitative and quantitative analysis in regulated and research laboratories. Designed and manufactured in Japan, this instrument integrates Shimadzu’s proprietary AART (Automatic Adjustment of Retention Time) algorithm, intelligent peak detection, and robust electron ionization (EI) source architecture to deliver reproducible spectral acquisition and precise retention time alignment across extended analytical sequences. Its core measurement principle relies on the separation of volatile and semi-volatile compounds via capillary gas chromatography, followed by mass-selective detection using a temperature-stabilized quadrupole mass analyzer operating under high vacuum (<1×10⁻⁵ Pa). The system is widely deployed in environmental testing (e.g., pesticide residue screening, PAHs, PCBs), pharmaceutical impurity profiling, food safety monitoring (mycotoxins, flavor volatiles), and forensic toxicology—where regulatory traceability and method transferability are critical.
Key Features
- High-sensitivity EI ion source with optimized electron beam current control and low-noise detector electronics
- Advanced AART functionality enabling automatic retention time correction without internal standards
- Real-time spectral deconvolution and library search (NIST/EPA/WM databases pre-installed)
- Integrated GC oven with programmable ramp rates up to 120 °C/min and temperature stability ±0.01 °C
- Mass axis calibration stability maintained over 72 hours without recalibration
- Front-panel touch interface with intuitive method setup wizard and status diagnostics
- Modular design supporting future upgrades including CI source, direct inlet probe, or GC auto-sampler integration
Sample Compatibility & Compliance
The GCMS-QP2010 Plus accommodates standard 0.25–0.53 mm ID fused-silica capillary columns (e.g., DB-5ms, HP-5MS, Rtx-5Sil MS) and accepts liquid, headspace, SPME, and thermal desorption injections. It supports split/splitless, PTV, and cold-on-column injection modes. All operational parameters—including method files, audit trails, and raw data—are stored in a structured, timestamped format compliant with GLP/GMP documentation requirements. When operated with Shimadzu’s LabSolutions GCMS software (ver. 5.9x or later), the system meets FDA 21 CFR Part 11 criteria for electronic records and signatures when configured with user authentication, role-based access control, and immutable audit logging. Routine performance verification follows ASTM D5845 (hydrocarbon analysis) and EPA Method 8270D (semivolatile organics), with built-in QC checklists and report templates.
Software & Data Management
LabSolutions GCMS provides a unified platform for instrument control, data acquisition, processing, and reporting. Key modules include Quantitative Analysis (QA), Qualitative Analysis (QA), and Batch Processing with customizable report generation (PDF, CSV, XML). Raw data (.qgd files) are stored in a vendor-neutral structure, enabling long-term archival and third-party reprocessing. Audit trail logs record all user actions—including method edits, integration parameter changes, and report exports—with SHA-256 hash integrity verification. Data backup routines support network drive mapping and scheduled archive compression. For laboratories undergoing ISO/IEC 17025 accreditation, the software includes calibration certificate tracking, instrument qualification (IQ/OQ/PQ) templates, and electronic signature workflows validated per ALCOA+ principles.
Applications
- Environmental: Screening and quantification of chlorinated pesticides, phthalates, brominated flame retardants, and emerging contaminants (e.g., PFAS precursors) in soil, water, and air matrices
- Pharmaceutical: Residual solvent analysis per ICH Q3C, genotoxic impurity detection at sub-ppm levels, and extractables/leachables profiling from packaging materials
- Food & Agriculture: Multi-residue pesticide analysis in fruits/vegetables, mycotoxin confirmation in cereals, and authenticity testing of essential oils and spices
- Forensics: Toxicological screening of drugs of abuse (benzodiazepines, opioids, stimulants) in biological fluids using selected ion monitoring (SIM) and retention index matching
- Materials Science: Outgassing analysis of polymers and composites under thermal stress, identification of volatile decomposition products
FAQ
Is this unit fully refurbished and certified prior to resale?
Yes. Each GCMS-QP2010 Plus undergoes full functional validation—including vacuum integrity test, mass calibration verification, sensitivity assessment (1 pg OFN in SIM mode), and GC oven thermal uniformity check—per Shimadzu’s original factory specifications. A comprehensive Certificate of Inspection and Performance Verification is provided.
What software version is installed, and is license transfer supported?
The system ships with LabSolutions GCMS v5.93 (or later), including full analytical license rights. License migration to the end-user’s domain is supported upon completion of Shimadzu’s authorized reseller transfer protocol.
Does the system include a GC auto-sampler?
Auto-sampler inclusion varies by unit configuration. Standard configurations include the AOC-20i or AOC-5000+; availability is confirmed during pre-purchase technical review and documented in the asset specification sheet.
Can this instrument be integrated into an existing LIMS environment?
Yes. LabSolutions GCMS supports ASTM E1394-compliant data export and offers optional LIMS connectivity via ODBC/JDBC drivers or HL7-compliant middleware adapters for seamless sample tracking and result ingestion.