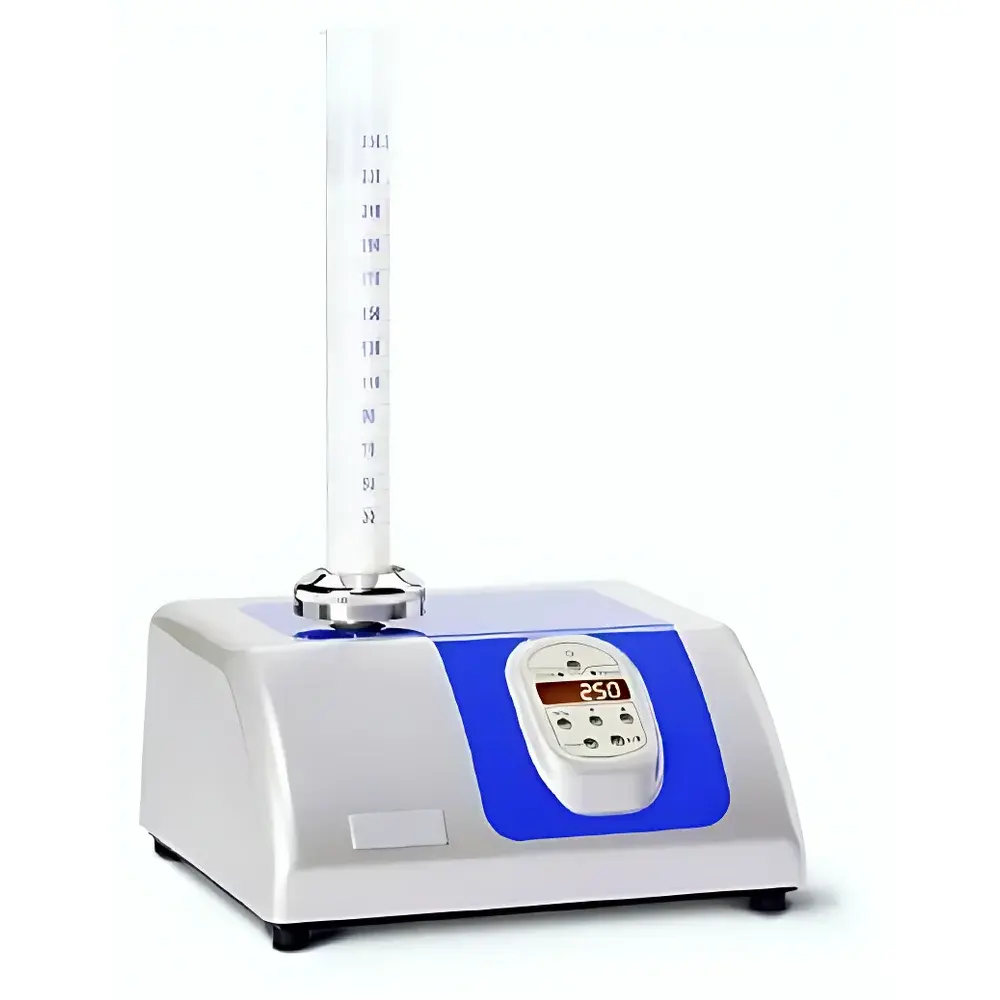
STAV II Powder Bulk Density Tester (Milk Powder Densitometer)
| Origin | Germany |
|---|---|
| Manufacturer | JEL GmbH |
| Type | STAV II |
| Power Supply | 230 V AC, 50 Hz |
| Vibration Frequency | 250 ± 15 cycles/min |
| Vibration Amplitude | 3.0 ± 0.1 mm |
| Available Cylinder Volumes | 50, 100, 250, 500, 1000 mL (DIN 53194 compliant) |
| Net Weight | 9 kg |
| Control Interface | Red illuminated panel with integrated counter (5-digit, programmable), conical locking mechanism for cylinder fixation, low-noise AC motor |
Overview
The STAV II Powder Bulk Density Tester is a precision-engineered instrument designed in accordance with DIN 53194 for the standardized determination of bulk density and tapped density of free-flowing and cohesive powdered materials—particularly critical in dairy powder processing, pharmaceutical excipient qualification, and food ingredient quality control. Based on the volumetric displacement principle, the STAV II measures the mass of a known volume of powder after controlled mechanical tapping, enabling calculation of bulk density (g/mL or g/cm³) and, when used with optional tap-count programming, tapped density and compressibility indices (e.g., Carr Index, Hausner Ratio). Its robust architecture integrates a calibrated electromagnetic vibration drive system, ensuring repeatable amplitude and frequency delivery independent of load variation or ambient temperature drift. The device operates exclusively under gravity-fed conditions without pneumatic or vacuum assistance, preserving material integrity and eliminating compaction artifacts induced by forced feeding.
Key Features
- High-precision mechanical tapping system delivering consistent 250 ± 15 cycles per minute at a fixed amplitude of 3.0 ± 0.1 mm—validated per DIN 53194 Annex A for reproducibility across operator shifts and laboratories.
- Interchangeable white-opaque graduated cylinders (50–1000 mL) manufactured to DIN 53194 dimensional and weight tolerances, with laser-etched calibration marks traceable to national metrology institutes.
- Conical locking collar mechanism ensures rigid, vibration-dampened cylinder fixation during tapping—eliminating lateral slippage or resonance-induced measurement scatter.
- Integrated 5-digit programmable counter with LED display enables precise tap count selection (1–99999), supporting multi-stage density protocols (e.g., initial bulk, intermediate taps, final tapped density).
- Dedicated red-illuminated control panel with tactile switch and status indicator lamp provides unambiguous operational feedback in low-light production or QC environments.
- Low-inertia AC motor (230 V / 50 Hz) engineered for continuous duty cycle operation with thermal overload protection and <25 dB(A) acoustic emission—suitable for shared laboratory spaces.
Sample Compatibility & Compliance
The STAV II accommodates powders ranging from highly aerated skim milk agglomerates to dense lactose monohydrate crystals, provided particle size distribution falls within the practical limits of DIN 53194 (typically d₉₀ < 1 mm). It is routinely deployed in compliance with ISO 787-11 (pigments), ASTM D1895 (plastics), USP <616> (pharmaceutical powders), and IDF Standard 103A (dairy powders). All cylinder volumes meet DIN 53194 requirements for wall thickness, base flatness, and graduation line width—ensuring traceability to German national standards (DKD/DAkkS). The instrument itself carries CE marking per Machinery Directive 2006/42/EC and EMC Directive 2014/30/EU.
Software & Data Management
While the STAV II operates as a stand-alone mechanical tester, its tap-count interface supports seamless integration with external digital workflows. Users may log results manually into LIMS or ELN systems using the programmable counter’s final value as an auditable endpoint. For automated data capture, optional RS-232 or USB-to-serial adapters enable real-time transmission of tap count and elapsed time to validated software platforms compliant with FDA 21 CFR Part 11 (electronic signatures, audit trails, user access controls). Calibration records—including cylinder verification certificates and amplitude/frequency validation reports—are maintained per GLP/GMP documentation requirements.
Applications
- Dairy industry: Routine QC of whole milk powder, whey protein concentrate, and infant formula for batch release against Codex Alimentarius density specifications.
- Pharmaceutical manufacturing: Excipient characterization (e.g., microcrystalline cellulose, starch) prior to direct compression tableting; assessment of flow aid efficacy.
- Food R&D: Correlation of bulk density with reconstitution behavior, solubility kinetics, and packaging fill-volume consistency.
- Chemical additives: Quality screening of fumed silica, precipitated calcium carbonate, and anti-caking agents for dispersion stability.
- Academic research: Fundamental studies on powder rheology, interparticle cohesion, and humidity-dependent density hysteresis.
FAQ
Does the STAV II comply with ISO or ASTM standards for bulk density measurement?
Yes—the instrument’s mechanical specification, cylinder geometry, and tapping parameters are aligned with DIN 53194, which forms the technical basis for ISO 787-11 and ASTM D1895. Full compliance documentation is supplied with each unit.
Can I use non-standard cylinder sizes or custom graduations?
No—only DIN 53194-certified cylinders (50, 100, 250, 500, and 1000 mL) are supported. Deviations invalidate traceability and void regulatory acceptance of results.
Is periodic recalibration required?
The vibration amplitude and frequency must be verified annually using a certified accelerometer and optical tachometer. Cylinder calibration requires annual re-verification per DKD-R 3-3 guidelines.
What maintenance does the STAV II require?
Routine cleaning of cylinder contact surfaces and inspection of the conical lock O-ring; motor lubrication is sealed-for-life. No user-serviceable electronics are present.
Is the device suitable for hygroscopic or electrostatic powders?
Yes—provided environmental RH is controlled (<40%) and grounding protocols are followed. Optional anti-static cylinder coatings are available upon request.